Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Intermediates
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Intermediates
The pharmaceutical and agrochemical industries continuously seek robust synthetic routes for nitrogen-containing heterocycles due to their pervasive presence in bioactive molecules. Quinazolinone derivatives, in particular, represent a privileged scaffold found in numerous therapeutic agents ranging from anticancer drugs to antimalarials. As illustrated in the structural diversity of known bioactive compounds such as Afloqualone, Luotonin F, and Sildenafil analogs, the quinazolinone core is indispensable for modern drug discovery. 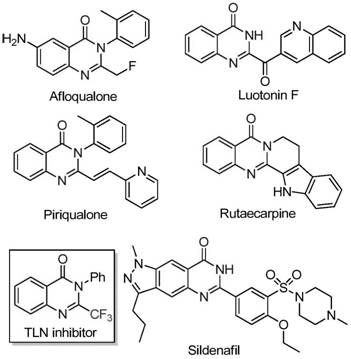 . The introduction of a trifluoromethyl group into this heterocyclic system further enhances metabolic stability, lipophilicity, and bioavailability, making 2-trifluoromethyl substituted quinazolinones highly coveted targets. Patent CN111675662B discloses a groundbreaking preparation method that addresses long-standing synthetic challenges, offering a pathway that is both economically viable and chemically efficient for producing these valuable intermediates.
. The introduction of a trifluoromethyl group into this heterocyclic system further enhances metabolic stability, lipophilicity, and bioavailability, making 2-trifluoromethyl substituted quinazolinones highly coveted targets. Patent CN111675662B discloses a groundbreaking preparation method that addresses long-standing synthetic challenges, offering a pathway that is both economically viable and chemically efficient for producing these valuable intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted quinazolinones has relied heavily on the cyclization of synthons bearing the trifluoromethyl group with substrates like anthranilamide or isatoic anhydride. While effective in specific contexts, these traditional methodologies suffer from significant drawbacks that hinder their widespread industrial adoption. Common trifluoromethylating agents such as trifluoroacetic anhydride or ethyl trifluoroacetate often necessitate severe reaction conditions, including extreme temperatures or highly corrosive environments, which pose safety risks and increase operational costs. Furthermore, these legacy routes frequently exhibit narrow substrate scope, failing to tolerate sensitive functional groups, and often result in mediocre yields that complicate downstream purification. The reliance on expensive or difficult-to-handle reagents creates a bottleneck for procurement teams aiming to secure reliable supply chains for complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to these conventional limitations, the novel method described in patent CN111675662B utilizes a strategy centered on the reaction between readily available trifluoroethylimidoyl chloride and isatin derivatives. This approach leverages a cheap metal iron catalyst, specifically ferric chloride, to drive the transformation under relatively mild conditions. By shifting the synthetic logic to utilize isatin as a key building block, the method bypasses the need for harsh cyclization promoters. The reaction proceeds through a tandem sequence involving alkali-promoted carbon-nitrogen bond formation followed by iron-catalyzed decarbonylation and cyclization. This mechanistic shift not only simplifies the operational procedure but also significantly broadens the functional group tolerance, allowing for the synthesis of diverse derivatives that were previously difficult to access. The use of abundant iron salts instead of precious metals represents a paradigm shift towards more sustainable and cost-effective manufacturing processes.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this innovative synthesis lies in the intricate interplay between the iron catalyst and the base-mediated activation of the substrates. The reaction initiates with the interaction between trifluoroethylimidoyl chloride and isatin in the presence of sodium hydride, which acts as a strong base to facilitate the initial nucleophilic attack. This step generates a transient trifluoroacetamidine intermediate, setting the stage for the subsequent cyclization event. The addition of ferric chloride (FeCl3) is critical, as it likely coordinates with the carbonyl oxygen or the nitrogen centers to lower the activation energy for the decarbonylation step. 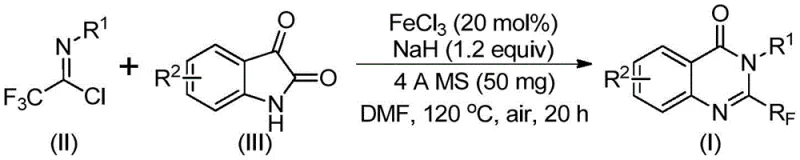 . The presence of 4A molecular sieves in the reaction mixture plays a pivotal role in sequestering moisture, thereby preventing the hydrolysis of the sensitive imidoyl chloride starting material and ensuring high conversion rates. The heating profile, typically ramping from 40°C to 120°C, allows for controlled reaction kinetics, minimizing side reactions while driving the thermodynamic equilibrium towards the desired quinazolinone product.
. The presence of 4A molecular sieves in the reaction mixture plays a pivotal role in sequestering moisture, thereby preventing the hydrolysis of the sensitive imidoyl chloride starting material and ensuring high conversion rates. The heating profile, typically ramping from 40°C to 120°C, allows for controlled reaction kinetics, minimizing side reactions while driving the thermodynamic equilibrium towards the desired quinazolinone product.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed alternatives. The use of a Lewis acidic iron catalyst in conjunction with a strong base creates a unique chemical environment that suppresses the formation of polymeric byproducts often seen in harsh acidic conditions. The decarbonylation step, which releases carbon monoxide, is efficiently managed within the closed system, and the resulting aromatic stabilization of the quinazolinone ring drives the reaction to completion. The high yields reported across various substituted isatins suggest that the catalytic cycle is robust against electronic variations on the aromatic rings. Whether the substrate contains electron-donating groups like methoxy or electron-withdrawing groups like nitro or halogens, the iron catalyst maintains its efficacy, ensuring a consistent impurity profile that simplifies the final purification steps required for pharmaceutical grade materials.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of handling in a standard laboratory or pilot plant setting. The protocol involves charging a reaction vessel with the catalyst system, drying agents, and substrates in a polar aprotic solvent such as DMF. The specific stoichiometry and temperature gradients are optimized to maximize the turnover number of the iron catalyst while minimizing decomposition of the sensitive imidoyl chloride. Detailed standardized operating procedures regarding the addition order of reagents and the specific workup protocols are essential for achieving the high purity levels demanded by regulatory standards. For a comprehensive breakdown of the exact molar ratios, solvent volumes, and temperature profiles validated in the patent examples, please refer to the technical guide below.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then heat to 120°C for an additional 18-20 hours under air.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this iron-catalyzed methodology presents a compelling value proposition centered on cost stability and supply security. The primary driver of cost reduction in this manufacturing process is the substitution of expensive noble metal catalysts or specialized fluorinating reagents with commodity chemicals like ferric chloride and sodium hydride. These reagents are globally sourced, widely available, and subject to minimal price volatility compared to proprietary catalysts. Additionally, the use of isatin and trifluoroethylimidoyl chloride as starting materials leverages established supply chains for bulk fine chemicals, reducing the risk of raw material shortages. The simplified post-treatment process, which relies on standard filtration and chromatography rather than complex extraction or distillation sequences, further reduces the operational expenditure associated with labor and energy consumption during the manufacturing cycle.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes the need for expensive metal scavenging steps, which are often required to meet strict residual metal limits in pharmaceutical ingredients. By utilizing iron, a non-toxic and abundant metal, the process inherently lowers the cost of goods sold (COGS). Furthermore, the high atom economy of the cyclization reaction minimizes waste generation, leading to substantial savings in waste disposal fees. The ability to run the reaction in common solvents like DMF, which can be recovered and recycled in large-scale operations, adds another layer of economic efficiency that directly impacts the bottom line for large volume production campaigns.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against varying substrate electronics ensures that supply continuity is maintained even when specific substituted isatins are required. Unlike sensitive organometallic reactions that may fail with slight variations in raw material quality, this iron-catalyzed system demonstrates high tolerance, reducing the rejection rate of incoming raw materials. The scalability of the process from gram to multi-kilogram levels without significant re-optimization means that suppliers can rapidly respond to increased demand from clinical trials or commercial launches. This flexibility is crucial for maintaining just-in-time inventory strategies and avoiding costly production delays.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the process aligns well with green chemistry principles. The use of air as the oxidant (implied by the open vessel or air conditions in some steps) eliminates the need for hazardous stoichiometric oxidants. The reaction conditions, while requiring elevated temperatures, do not demand extreme pressures or cryogenic cooling, simplifying the engineering requirements for reactor vessels. This ease of scale-up facilitates the transition from R&D to commercial manufacturing, ensuring that the supply of high-purity intermediates can grow in lockstep with the development pipeline of the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-trifluoromethyl quinazolinones using this patented methodology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a transparent view of the technology's capabilities. Understanding these nuances helps stakeholders make informed decisions regarding process adoption and vendor selection for their specific project needs.
Q: What are the advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) offers significant cost advantages as it is an inexpensive, earth-abundant metal compared to precious metal catalysts. Furthermore, the patent data indicates high functional group tolerance and efficient conversion rates under relatively mild oxidative conditions, reducing the need for complex inert atmosphere setups often required by other transition metals.
Q: Can this synthesis method accommodate diverse substituents on the aromatic ring?
A: Yes, the method demonstrates excellent substrate scope. The patent explicitly details successful synthesis with various substituents including methyl, fluoro, bromo, chloro, methoxy, and nitro groups at ortho-, meta-, and para-positions, confirming its versatility for generating diverse chemical libraries.
Q: Is this process suitable for large-scale industrial production?
A: The process is designed with scalability in mind. It utilizes readily available starting materials like isatin and trifluoroethylimidoyl chloride, operates in common solvents like DMF, and avoids extremely sensitive reagents. The simplified post-treatment involving filtration and standard column chromatography supports potential scale-up from gram to kilogram levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our team of expert chemists has extensively evaluated the iron-catalyzed synthesis route described in CN111675662B and confirmed its potential for delivering high-purity 2-trifluoromethyl quinazolinones. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed impurity profiling to support regulatory filings. We are committed to being a strategic partner who not only supplies chemicals but also provides technical solutions to optimize your manufacturing footprint.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us to obtain specific COA data for our catalog compounds or to initiate route feasibility assessments for custom derivatives. Let us help you accelerate your project timelines with reliable, cost-effective, and scalable chemical solutions tailored to your unique challenges.
