Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which enhance metabolic stability and lipophilicity. Patent CN111423381A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing critical challenges in modern drug synthesis. This technology leverages a transition metal palladium-catalyzed carbonylation cascade reaction, utilizing readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts. Unlike conventional approaches that often require hazardous reagents or extreme conditions, this innovation operates under remarkably mild parameters, specifically at 30°C, ensuring high reaction efficiency and exceptional substrate compatibility. For R&D directors and procurement specialists, this represents a significant opportunity to streamline the production of high-value intermediates used in bioactive molecules and functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles with trifluoromethyl functional groups has relied heavily on synthons like trifluorodiazoethane. While effective in certain contexts, these traditional reagents pose substantial safety risks due to their explosive nature and instability, complicating storage and handling in large-scale facilities. Furthermore, alternative methods using trifluoroethylimide acid halides have not been widely explored or optimized, leaving a gap in practical, safe synthetic routes. Conventional processes often demand high temperatures, pressurized carbon monoxide cylinders, or complex multi-step sequences that drive up operational costs and extend lead times. These limitations create bottlenecks in the supply chain for reliable pharmaceutical intermediate suppliers, as the inherent dangers and inefficiencies restrict the ability to scale production reliably while maintaining stringent purity specifications required for API manufacturing.
The Novel Approach
The patented methodology revolutionizes this landscape by employing a palladium-catalyzed carbonylation series reaction that bypasses the need for gaseous carbon monoxide and unstable diazo compounds. By utilizing a mixture of formic acid and acetic anhydride as a safe carbon monoxide surrogate, the process generates the necessary carbonyl species in situ under ambient pressure. The reaction integrates trifluoroethylimidoyl chloride and propargylamine with diaryliodonium salts to efficiently construct the imidazole core. As illustrated in the general reaction scheme below, this one-pot transformation is not only operationally simple but also exhibits remarkable versatility.
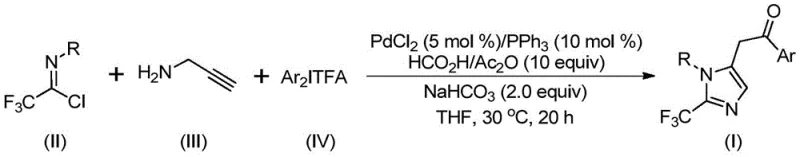
This novel approach allows for the design and synthesis of diversified substituted imidazole compounds simply by modifying the substrates, thereby broadening the utility of the method for creating diverse chemical libraries. The mild reaction temperature of 30°C significantly reduces energy consumption compared to traditional high-heat protocols, aligning with green chemistry principles while delivering high yields.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
Understanding the mechanistic pathway is crucial for R&D teams aiming to optimize this process for specific derivatives. The reaction likely initiates with a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine, yielding a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization, setting the stage for the palladium catalytic cycle. The palladium catalyst, typically palladium chloride coordinated with triphenylphosphine, facilitates the aminopalladation of the alkyne moiety, generating a key alkenyl palladium intermediate. Through further isomerization, this transforms into an alkyl palladium species, which is then subjected to carbonylation driven by the carbon monoxide released from the formic acid and acetic anhydride mixture.
Following carbonylation, an acyl palladium intermediate is formed, which serves as the precursor for the final ring closure. The diaryliodonium salt then participates in an oxidative addition step, creating a high-valent tetravalent palladium intermediate. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted imidazole compound and regenerates the active palladium catalyst. This intricate cascade ensures high atom economy and minimizes the formation of unwanted byproducts. The compatibility with various functional groups, as evidenced by the successful synthesis of derivatives shown below, confirms the robustness of this catalytic system against electronic variations in the substrate.
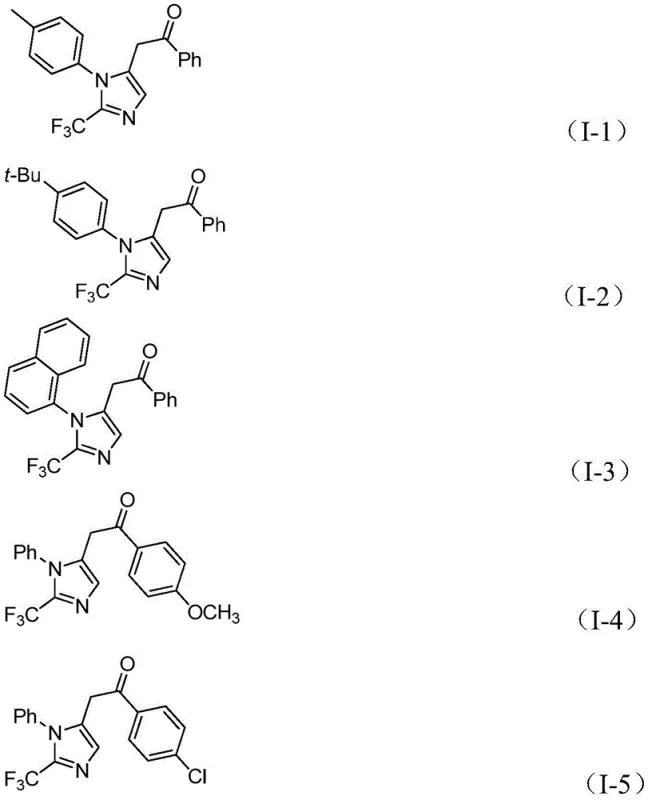
Impurity control is inherently managed by the specificity of the palladium cycle and the mild conditions, which prevent thermal degradation of sensitive functional groups. The use of sodium bicarbonate as a mild base further mitigates side reactions, ensuring a clean crude profile that simplifies downstream purification efforts.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Executing this synthesis requires precise adherence to the molar ratios and reaction conditions outlined in the patent to achieve optimal yields ranging from 65% to 97%. The process involves dissolving the catalyst system and reagents in an aprotic solvent such as tetrahydrofuran (THF), which has been identified as the preferred medium for maximizing conversion rates. The reaction mixture is stirred at a controlled temperature of 30°C for a duration of 16 to 24 hours, allowing sufficient time for the cascade sequence to reach completion without excessive energy input. Detailed standardized synthesis steps follow below.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, and a formic acid/acetic anhydride mixture in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt to the reaction vessel under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, then filter and purify via column chromatography to isolate the target imidazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this patented technology are profound. The shift from hazardous, specialized reagents to commodity chemicals like trifluoroethylimidoyl chloride and propargylamine drastically simplifies the sourcing strategy. These starting materials are not only cheap and readily available in the global market but also possess long shelf lives, reducing inventory waste and storage costs associated with unstable precursors. The elimination of high-pressure equipment and the ability to run reactions at near-room temperature significantly lower capital expenditure (CAPEX) and operational expenditure (OPEX) for manufacturing facilities.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by replacing expensive and dangerous trifluorodiazoethane with stable, low-cost imidoyl chlorides. Additionally, the use of inexpensive palladium chloride instead of more exotic catalysts, combined with the avoidance of high-energy heating protocols, drives down the overall cost of goods sold (COGS). The high reaction efficiency and yields observed across a broad range of substrates mean less raw material is wasted, directly improving the bottom line for cost reduction in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for supply chain continuity. Since the key reagents—aromatic amines, alkynyl amines, and diaryliodonium salts—are commercially available and can be synthesized from abundant precursors like aryl boronic acids, the risk of supply disruption is minimized. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed runs or complex troubleshooting, thereby securing a reliable supply of high-purity intermediates.
- Scalability and Environmental Compliance: The patent explicitly notes the method's potential for expansion to gram-level and industrial-scale production. The simplified post-processing, involving basic filtration and standard silica gel chromatography, avoids complex separation techniques that often hinder scale-up. Furthermore, the avoidance of toxic gases and the use of milder reagents contribute to a safer environmental footprint, facilitating easier compliance with increasingly strict environmental regulations regarding waste disposal and worker safety in chemical plants.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on its practical application in an industrial setting.
Q: What are the advantages of this palladium-catalyzed method over traditional trifluoromethylation?
A: Unlike traditional methods that often rely on unstable or hazardous trifluorodiazoethane, this patented process utilizes stable and commercially available trifluoroethylimidoyl chlorides. It operates under mild conditions (30°C) without the need for high-pressure carbon monoxide gas, significantly enhancing operational safety and scalability for industrial production.
Q: What is the substrate compatibility of this synthesis route?
A: The method demonstrates excellent functional group tolerance. It successfully accommodates various substituents on both the aryl ring of the imidoyl chloride and the diaryliodonium salt, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens, nitro, and trifluoromethyl groups, yielding products with high efficiency.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly states that the method is scalable to the gram level and potentially beyond. The use of inexpensive catalysts like palladium chloride, combined with simple post-processing steps involving filtration and standard column chromatography, makes it highly viable for cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
As the demand for fluorinated heterocycles continues to surge in the development of next-generation therapeutics, partnering with an experienced CDMO is essential for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of 2-trifluoromethyl imidazole meets the highest international standards, supporting your regulatory filings and clinical trials with confidence.
We invite you to leverage our technical expertise to optimize your supply chain and reduce time-to-market. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced palladium-catalyzed processes can deliver superior value and reliability for your organization.
