Revolutionizing Pharmaceutical Intermediate Production: Scalable Synthesis of High-Purity 3-Arylquinolinone Derivatives for Global Supply Chains
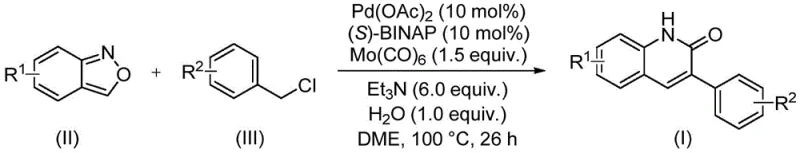
The patent CN113045489B represents a significant advancement in heterocyclic compound synthesis, introducing an innovative palladium-catalyzed aminocarbonylation methodology specifically designed for manufacturing 3-arylquinoline-2(1H) ketone derivatives. This breakthrough addresses longstanding challenges in pharmaceutical intermediate production by leveraging benzisoxazole's unique dual functionality as both nitrogen source and formyl donor within a single reaction vessel. The process demonstrates remarkable operational simplicity while delivering high-yielding results across diverse substrate combinations, establishing a new benchmark for efficiency in quinolinone derivative synthesis. By eliminating multi-step sequences required in conventional approaches, this method significantly reduces manufacturing complexity while maintaining exceptional product purity essential for pharmaceutical applications. The strategic integration of commercially available catalysts and ligands further enhances its industrial viability without compromising on quality standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for quinolinone derivatives, including Vilsmeier-Haack, Knorr, and Friedlander reactions, suffer from multiple critical limitations that hinder their industrial scalability. These methods typically require harsh reaction conditions involving strong acids or high temperatures that promote unwanted side reactions and decomposition pathways, particularly with sensitive functional groups commonly found in pharmaceutical intermediates. The multi-step nature of conventional approaches necessitates extensive purification procedures between stages, significantly increasing production timelines while introducing cumulative yield losses that impact overall process economics. Furthermore, transition metal-catalyzed variants often demand complex catalyst removal protocols to meet stringent pharmaceutical purity requirements, adding substantial cost burdens through specialized equipment and additional processing steps that compromise supply chain efficiency for global manufacturers.
The Novel Approach
The patented methodology overcomes these limitations through an elegant single-step palladium-catalyzed aminocarbonylation process that operates under remarkably mild conditions at precisely controlled temperatures. By utilizing benzisoxazole as a dual-functionality reagent serving simultaneously as nitrogen source and formyl donor, the reaction eliminates the need for separate reagents while maintaining exceptional substrate compatibility across diverse functional groups including halogens, alkoxy moieties, and cyano substituents. The carefully optimized catalyst system featuring palladium acetate with (S)-BINAP ligand ensures high conversion rates without requiring extensive post-reaction metal removal procedures, directly addressing critical pain points in pharmaceutical intermediate manufacturing. This streamlined approach significantly reduces both processing time and operational complexity while delivering consistent high-purity products suitable for immediate incorporation into downstream pharmaceutical synthesis pathways.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The reaction mechanism proceeds through a sophisticated catalytic cycle initiated by oxidative addition of benzyl chloride to the palladium(0) species generated in situ from palladium acetate and (S)-BINAP ligand. This key step forms an aryl-palladium intermediate that subsequently undergoes carbonyl insertion facilitated by molybdenum carbonyl as the carbon monoxide source, creating an acyl-palladium complex essential for the transformation. The benzisoxazole then participates through nucleophilic attack on this intermediate, with its unique structure enabling simultaneous nitrogen incorporation and ring formation through intramolecular cyclization. The final reductive elimination step releases the desired quinolinone product while regenerating the active palladium catalyst, completing the catalytic cycle with exceptional atom economy that minimizes waste generation throughout the process.
Impurity control is achieved through the precise stoichiometric balance between reactants and the selective nature of the catalytic pathway, which inherently suppresses common side reactions observed in traditional methods. The dual role of benzisoxazole prevents competing pathways that typically generate regioisomeric impurities, while the mild reaction conditions (100°C in DME solvent) minimize thermal degradation products that compromise purity profiles in conventional syntheses. The water co-solvent plays a critical role in hydrolyzing potential intermediates that could lead to impurities, ensuring clean conversion to the target quinolinone structure with minimal byproduct formation. This inherent selectivity eliminates the need for extensive purification steps typically required to achieve pharmaceutical-grade purity standards.
How to Synthesize 3-Arylquinolinone Efficiently
This patented methodology represents a significant advancement in the manufacturing of high-value quinolinone intermediates through its innovative approach to heterocyclic ring formation. The process leverages commercially available starting materials and catalysts to achieve exceptional yields while maintaining operational simplicity suitable for large-scale production environments. By eliminating multi-step sequences required in conventional syntheses, this method dramatically reduces both processing time and complexity while delivering consistent high-purity products essential for pharmaceutical applications. Detailed standardized synthesis procedures are provided below to ensure seamless implementation across diverse manufacturing facilities.
- Prepare reaction mixture by combining benzisoxazole compound (II), benzyl chloride derivative (III), palladium acetate catalyst (0.1 mol%), (S)-BINAP ligand (0.1 mol%), molybdenum carbonyl (1.5 equiv.), triethylamine (6.0 equiv.), and water (1.0 equiv.) in ethylene glycol dimethyl ether solvent
- Conduct the reaction under nitrogen atmosphere at precisely 100°C for 26 hours in sealed vessel, maintaining strict temperature control to ensure complete conversion while preventing side reactions
- Perform post-processing through filtration to remove catalyst residues, followed by silica gel sample preparation and column chromatography purification to obtain high-purity 3-arylquinolinone derivative with stringent quality control
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach delivers substantial strategic advantages for procurement and supply chain operations by addressing critical pain points in pharmaceutical intermediate sourcing. The elimination of complex multi-step processes significantly reduces production lead times while enhancing supply chain resilience through simplified material requirements and reduced dependency on specialized equipment. The use of readily available starting materials creates inherent flexibility in sourcing strategies, allowing manufacturers to maintain consistent production schedules even during market fluctuations or regional supply disruptions that commonly affect traditional synthetic routes.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal removal steps and simplified purification protocol directly reduces processing costs by minimizing specialized equipment requirements and associated operational expenses. The dual functionality of benzisoxazole eliminates the need for separate nitrogen and formyl sources, significantly reducing raw material costs while maintaining high atom economy that minimizes waste disposal expenses throughout the manufacturing process.
- Enhanced Supply Chain Reliability: The broad availability of all starting materials from multiple global suppliers creates inherent redundancy in sourcing strategies, substantially reducing vulnerability to single-source dependencies that frequently disrupt traditional supply chains. The robust nature of the reaction tolerates minor variations in raw material quality without requiring process reoptimization, ensuring consistent production output even when sourcing from different regional suppliers during market fluctuations.
- Scalability and Environmental Compliance: The straightforward reaction setup and mild operating conditions enable seamless scale-up from laboratory to commercial production without requiring specialized infrastructure modifications, significantly accelerating time-to-market for new pharmaceutical candidates. The reduced number of processing steps minimizes solvent usage and waste generation compared to conventional methods, aligning with increasingly stringent environmental regulations while lowering overall environmental impact through improved process efficiency.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement teams regarding implementation of this patented manufacturing technology across global supply chains. These insights are derived directly from the patent's experimental data and technical specifications to provide accurate guidance for operational planning and quality assurance protocols.
Q: How does benzisoxazole's dual role as nitrogen and formyl source improve process efficiency?
A: The innovative utilization of benzisoxazole as both nitrogen donor and formyl source eliminates the need for separate reagents, streamlining the synthetic pathway by reducing reaction steps and minimizing intermediate handling. This dual functionality significantly simplifies the manufacturing process while maintaining high functional group tolerance across diverse substrates.
Q: What advantages does this method offer over conventional Vilsmeier Haack or Knorr reactions?
A: Unlike traditional methods requiring harsh conditions or multiple steps, this palladium-catalyzed approach operates under milder parameters with broader substrate compatibility. The elimination of transition metal removal steps and simplified purification process directly enhances operational efficiency while improving product purity profiles for pharmaceutical applications.
Q: How does the broad functional group tolerance benefit pharmaceutical intermediate manufacturing?
A: The exceptional tolerance for various substituents including halogens, alkoxy groups, and cyano functionalities enables rapid production of diverse derivative libraries without process reoptimization. This flexibility supports accelerated drug discovery timelines while maintaining consistent quality standards across different molecular variants required by pharmaceutical clients.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolinone Derivatives Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities. As a trusted CDMO partner specializing in complex heterocyclic synthesis, we have successfully implemented this patented methodology across multiple client projects with consistent delivery of high-purity quinolinone intermediates meeting global regulatory standards. Our dedicated technical team ensures seamless technology transfer through comprehensive process validation protocols that guarantee reproducibility at any scale required by pharmaceutical manufacturers worldwide.
Leverage our expertise to accelerate your drug development timeline through Customized Cost-Saving Analysis tailored to your specific production requirements. Contact our technical procurement team today to request specific COA data and route feasibility assessments that demonstrate how this innovative synthesis can optimize your supply chain operations while maintaining uncompromised quality standards essential for pharmaceutical applications.
