Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for High-Value Pharmaceutical Intermediates
Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for High-Value Pharmaceutical Intermediates
The strategic incorporation of trifluoromethyl groups into heterocyclic scaffolds represents a cornerstone of modern medicinal chemistry, driven by the profound impact these motifs have on the pharmacokinetic profiles of drug candidates. As detailed in patent CN110467579B, a novel and efficient preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds has been developed, addressing critical bottlenecks in the supply chain for high-performance active pharmaceutical ingredients (APIs). The 1,2,4-triazole ring is a privileged structure found in a vast array of bioactive molecules, ranging from antifungal agents like fluconazole to iron chelators such as deferasirox, as well as functional materials used in organic light-emitting diodes (OLEDs). The introduction of the trifluoromethyl group specifically enhances electronegativity, metabolic stability, and lipophilicity, thereby improving membrane permeability and oral bioavailability. This technological breakthrough offers a streamlined pathway for producing these valuable intermediates, positioning it as a vital asset for any organization seeking a reliable pharmaceutical intermediate supplier capable of delivering complex fluorinated heterocycles.
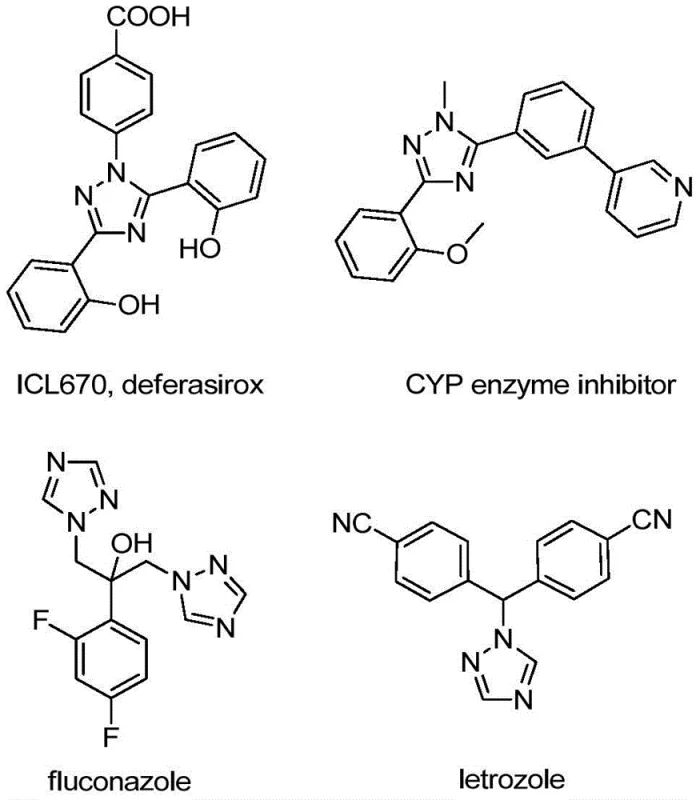
The significance of this innovation extends beyond mere academic interest; it directly impacts the commercial viability of synthesizing next-generation therapeutics. Traditional routes to these fluorinated heterocycles often suffer from harsh reaction conditions, limited substrate scope, or the reliance on hazardous reagents that complicate scale-up. By contrast, the methodology disclosed in the patent utilizes readily available starting materials—specifically trifluoroethylimidoyl chlorides and hydrazones—under mild conditions promoted by non-metallic iodine. This approach not only simplifies the operational workflow but also aligns with the growing industry demand for greener, more sustainable chemical manufacturing processes. For R&D directors and procurement managers alike, understanding the nuances of this synthesis is key to optimizing the cost structure and supply reliability of their pipeline projects involving fluorinated triazole derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted nitrogen-containing heterocycles has been fraught with synthetic challenges that hinder efficient large-scale production. One predominant strategy involves the direct trifluoromethylation of pre-synthesized 1,2,4-triazole rings. While conceptually straightforward, this route typically necessitates the use of specialized and often expensive trifluoromethylating reagents, such as Togni reagents or Umemoto reagents, which significantly inflate the raw material costs. Furthermore, these reactions frequently require stringent control over reaction parameters, including low temperatures and strictly anhydrous environments, to prevent reagent decomposition and ensure selectivity. Another common approach utilizes trifluorodiazoethane as a synthon; however, this reagent is inherently unstable and potentially explosive, posing severe safety risks that are unacceptable in a commercial manufacturing setting. These limitations create substantial barriers to entry for cost reduction in API manufacturing, forcing companies to rely on multi-step sequences with poor atom economy and difficult purification protocols.
The Novel Approach
The methodology presented in patent CN110467579B offers a transformative alternative by leveraging a condensation-cyclization strategy between trifluoroethylimidoyl chlorides and hydrazones. This route bypasses the need for dangerous diazo compounds and expensive late-stage trifluoromethylation agents. Instead, it employs a tandem reaction sequence where the carbon-nitrogen bond formation and ring closure are facilitated by a simple base, sodium acetate, and promoted by elemental iodine. The reaction proceeds efficiently at moderate temperatures, typically around 80°C, in common organic solvents like 1,2-dichloroethane (DCE). Crucially, this method eliminates the requirement for inert atmosphere techniques, allowing the reaction to be conducted under ambient air without significant loss in yield or purity. The versatility of this approach is demonstrated by its ability to tolerate a wide range of substituents on both the imidoyl chloride and the hydrazone components, enabling the rapid generation of diverse libraries of 5-trifluoromethyl-1,2,4-triazoles for structure-activity relationship (SAR) studies.
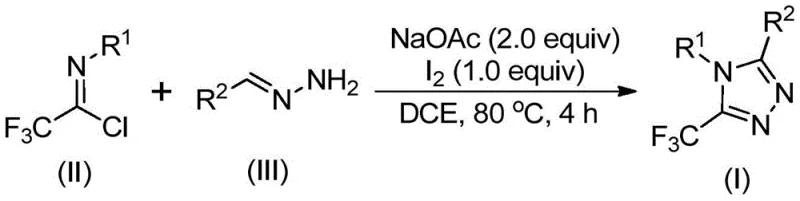
Mechanistic Insights into Iodine-Promoted Cyclization
A deep understanding of the reaction mechanism is essential for R&D teams aiming to optimize this process for specific substrates or troubleshoot potential impurities. The proposed mechanism begins with a base-promoted intermolecular nucleophilic attack, where the amino group of the hydrazone attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This initial step forms a trifluoroacetamidine intermediate, releasing hydrochloric acid which is scavenged by the sodium acetate base. Following this condensation, the intermediate undergoes an isomerization to align the reactive centers for cyclization. The addition of elemental iodine then triggers an oxidative iodination process, generating an iodinated species that acts as a potent electrophile. This activation is critical, as it facilitates the subsequent intramolecular electrophilic substitution where the nitrogen atom attacks the activated carbon center, closing the five-membered triazole ring. The final step involves aromatization and elimination of hydrogen iodide to yield the stable 5-trifluoromethyl-1,2,4-triazole product. This mechanistic pathway highlights the dual role of iodine as both an oxidant and a leaving group facilitator, driving the reaction to completion with high efficiency.
From an impurity control perspective, the mildness of this iodine-promoted system offers distinct advantages over transition metal-catalyzed alternatives. Heavy metal catalysts often leave behind trace residues that are difficult to remove and can catalyze unwanted side reactions during storage or downstream processing. By utilizing non-metallic iodine, the impurity profile is significantly simplified, primarily consisting of unreacted starting materials or simple halogenated byproducts that are easily removed via standard aqueous workups or silica gel chromatography. Furthermore, the absence of strong oxidants or radical initiators minimizes the risk of over-oxidation or polymerization of sensitive functional groups on the aromatic rings. This clean reaction profile ensures that the resulting high-purity pharmaceutical intermediates meet the rigorous quality standards required for clinical development, reducing the burden on analytical teams to characterize complex degradation products.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazoles Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and addition sequences to maximize yield and safety. The protocol generally involves dissolving the trifluoroethylimidoyl chloride and hydrazone in a suitable aprotic solvent, followed by the addition of sodium acetate as the base. The mixture is heated to facilitate the initial condensation before the iodine promoter is introduced to drive the cyclization. Detailed standardized operating procedures are critical for ensuring batch-to-batch consistency, particularly when scaling up from gram to kilogram quantities. For a comprehensive guide on the specific molar ratios, temperature ramps, and workup procedures validated in the patent examples, please refer to the technical steps outlined below.
- Mix sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane.
- Heat the reaction mixture to 80°C and stir for 2 to 4 hours to allow initial condensation and isomerization.
- Add elemental iodine to the system and continue heating for 1 to 2 hours to promote oxidative cyclization and aromatization.
- Perform post-treatment including filtration and silica gel purification to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible operational efficiencies and risk mitigation strategies. The shift away from exotic reagents and hazardous conditions directly addresses the volatility often seen in the supply of specialized fine chemicals. By relying on commodity chemicals like hydrazones and imidoyl chlorides, which are widely produced for various industrial applications, manufacturers can secure a more stable and predictable supply chain. This stability is crucial for maintaining continuous production schedules and avoiding costly delays associated with sourcing niche reagents. Moreover, the simplified reaction conditions reduce the dependency on specialized equipment, such as high-pressure reactors or gloveboxes, thereby lowering the capital expenditure required for technology transfer and scale-up.
- Cost Reduction in Manufacturing: The economic benefits of this method are driven primarily by the elimination of expensive transition metal catalysts and the use of inexpensive promoters like elemental iodine. Transition metals such as palladium or rhodium not only carry high price tags but also incur significant costs related to recovery and disposal to meet environmental regulations. By removing these metals from the process, the overall cost of goods sold (COGS) is drastically reduced. Additionally, the high atom economy of the condensation-cyclization sequence minimizes waste generation, further lowering disposal costs. The ability to run the reaction without strict anhydrous conditions also reduces utility costs associated with solvent drying and nitrogen purging, contributing to substantial cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures that production is less susceptible to disruptions caused by environmental factors or equipment failures. Since the process does not require sensitive inert atmosphere techniques, it can be executed in standard glass-lined or stainless steel reactors available in most multipurpose chemical plants. This flexibility allows for greater agility in scheduling production runs across different facilities, enhancing the overall resilience of the supply network. Furthermore, the broad substrate scope means that a single platform technology can be used to produce a wide variety of derivatives, reducing the need for multiple distinct synthetic routes and simplifying inventory management for raw materials.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but the exothermic profile of this iodine-promoted reaction is manageable and well-suited for large-scale operations. The use of common solvents like dichloroethane, which can be recovered and recycled, aligns with green chemistry principles and helps facilities maintain compliance with increasingly stringent environmental regulations. The absence of heavy metal waste streams simplifies the wastewater treatment process, reducing the environmental footprint of the manufacturing site. This alignment with sustainability goals is increasingly important for multinational corporations aiming to meet their corporate social responsibility (CSR) targets while maintaining efficient commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common questions regarding the reaction scope, safety, and scalability. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable foundation for decision-making. Understanding these details is essential for assessing the potential integration of this method into existing production workflows.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the method utilizes elemental iodine as a promoter instead of costly heavy metal catalysts like palladium or copper, significantly reducing raw material costs and simplifying metal residue removal.
Q: What are the reaction conditions regarding moisture and oxygen sensitivity?
A: The process is robust and does not require strict anhydrous or anaerobic conditions, making it highly suitable for standard industrial reactor setups without specialized inert atmosphere equipment.
Q: Can this method accommodate diverse functional groups on the substrate?
A: Yes, the method demonstrates excellent functional group tolerance, successfully synthesizing derivatives with methyl, methoxy, bromo, nitro, and heteroaryl substituents with high yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new drugs and advanced materials. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify identity and assay. Whether you require custom synthesis of novel triazole derivatives or reliable supply of established intermediates, our infrastructure is designed to support your long-term growth and innovation goals.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits of switching to this iodine-promoted route for your supply chain. We encourage potential partners to contact us to obtain specific COA data for our available stock or to initiate route feasibility assessments for your proprietary molecules. Let us collaborate to accelerate your development timelines and secure a competitive advantage in the global marketplace.
