Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for High-Purity Pharmaceutical Intermediates
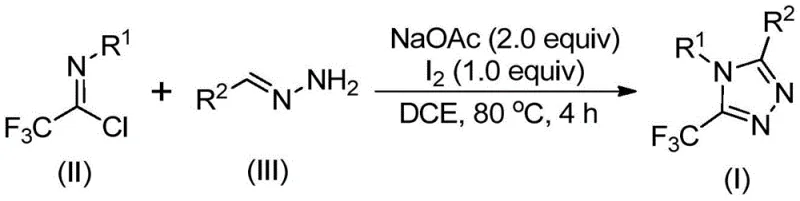
The pharmaceutical and agrochemical industries are constantly seeking efficient routes to fluorinated heterocycles, driven by the unique ability of the trifluoromethyl group to enhance metabolic stability, lipophilicity, and bioavailability in drug candidates. Patent CN110467579B introduces a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in current synthetic methodologies. This innovation leverages a non-metallic iodine-promoted cyclization strategy that transforms inexpensive hydrazones and trifluoroethylimidoyl chlorides into high-value scaffolds under remarkably mild conditions. By eliminating the need for complex catalytic systems and harsh reaction environments, this technology offers a streamlined pathway for producing key intermediates used in the development of advanced therapeutics and functional materials. The robustness of this protocol suggests immediate applicability for reliable pharmaceutical intermediate supplier networks aiming to optimize their production portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of trifluoromethyl-substituted nitrogen-containing heterocycles has relied heavily on two primary strategies, both of which present significant logistical and economic challenges for large-scale manufacturing. The first approach involves the direct trifluoromethylation of pre-synthesized heterocyclic cores, which necessitates the use of specialized and often hazardous trifluoromethylating reagents that are costly to procure and handle safely. The second mainstream method utilizes synthons bearing trifluoromethyl groups, such as trifluorodiazoethane, which poses severe safety risks due to its explosive nature and instability, requiring specialized infrastructure to manage. Furthermore, many existing protocols depend on transition metal catalysts that leave behind toxic residues, necessitating rigorous and expensive purification steps to meet the stringent purity specifications required for active pharmaceutical ingredients (APIs). These factors collectively inflate the cost of goods sold and complicate the supply chain continuity for essential chemical building blocks.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a direct condensation and oxidative cyclization strategy that bypasses these historical hurdles entirely. By employing readily available hydrazones and trifluoroethylimidoyl chlorides as starting materials, the process accesses the 1,2,4-triazole core through a straightforward one-pot sequence. The use of elemental iodine as a promoter, rather than a stoichiometric oxidant or heavy metal catalyst, represents a paradigm shift towards greener chemistry. This novel approach operates effectively at moderate temperatures around 80°C in common organic solvents like dichloroethane, without the need for rigorous exclusion of air or moisture. Such operational simplicity not only reduces the capital expenditure required for reactor setup but also drastically simplifies the post-reaction workup, allowing for rapid isolation of the target compounds via standard filtration and chromatography techniques.
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic pathway of this transformation is elegantly designed to maximize atom economy while minimizing side reactions. The reaction initiates with a base-promoted intermolecular carbon-nitrogen bond formation between the hydrazone and the trifluoroethylimidoyl chloride, generating a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization to align the reactive centers for cyclization. The introduction of elemental iodine then triggers a base-promoted oxidative iodination, creating a reactive iodine-bearing intermediate that is primed for ring closure. The final stage involves an intramolecular electrophilic substitution followed by aromatization, which drives the equilibrium towards the formation of the stable 5-trifluoromethyl substituted 1,2,4-triazole product. This cascade ensures high conversion rates and minimizes the accumulation of partially reacted intermediates that often plague multi-step syntheses.
From an impurity control perspective, the mild nature of the iodine promotion is particularly advantageous. Unlike aggressive oxidants that might degrade sensitive functional groups on the aromatic rings, elemental iodine provides a controlled oxidation potential that preserves the integrity of substituents such as halogens, ethers, and nitro groups. This high level of chemoselectivity is crucial for R&D teams focusing on cost reduction in API manufacturing, as it reduces the burden on downstream purification processes. The ability to tolerate a wide range of electronic environments on both the N1 and C5 positions of the triazole ring ensures that the process is versatile enough to generate diverse libraries of analogs for structure-activity relationship (SAR) studies without needing to re-optimize reaction conditions for each new substrate.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
The synthesis protocol outlined in the patent is designed for ease of execution, making it accessible for both laboratory-scale discovery and pilot-plant operations. The procedure involves charging a reaction vessel with sodium acetate, the specific trifluoroethylimidoyl chloride, and the chosen hydrazone derivative in an organic solvent. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and timing required to achieve optimal yields. The reaction is heated to facilitate the initial coupling, followed by the timed addition of iodine to drive the oxidative cyclization to completion. This sequential addition is key to controlling the reaction exotherm and ensuring the formation of the desired regioisomer.
- Mix sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane (DCE).
- Heat the reaction mixture to 80°C and stir for 2 to 4 hours to allow initial condensation and cyclization.
- Add elemental iodine to the system and continue heating for an additional 1 to 2 hours to promote oxidative aromatization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond simple chemical efficiency. The elimination of exotic reagents and the reliance on commodity chemicals like hydrazones and imidoyl chlorides significantly de-risks the supply chain, ensuring that raw material availability remains stable even during market fluctuations. The absence of heavy metal catalysts removes the need for expensive scavenging resins or complex extraction protocols, directly translating to lower operational expenditures and reduced waste disposal costs. Furthermore, the robustness of the reaction conditions means that production can be scheduled with greater flexibility, as there is no need for extended purging cycles or specialized drying of solvents, thereby enhancing overall plant throughput and asset utilization.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive starting materials that are widely available in the global chemical market. By avoiding the use of precious metal catalysts such as palladium or copper, the method eliminates a major cost driver associated with catalyst recovery and metal residue testing. Additionally, the simplified workup procedure, which often requires only filtration and standard column chromatography, reduces the consumption of silica gel and solvents compared to more complex multi-step sequences. These factors combine to deliver substantial cost savings per kilogram of produced intermediate, improving the margin profile for downstream API synthesis.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents like elemental iodine and sodium acetate ensures that production schedules are not disrupted by the short shelf-life issues common with sensitive organometallic reagents. The tolerance for ambient atmospheric conditions means that the process can be run in standard glass-lined or stainless steel reactors without the need for specialized inert gas manifolds, increasing the number of available production suites within a facility. This operational flexibility allows for rapid scale-up from gram to kilogram quantities, ensuring that clinical trial material can be produced quickly to meet tight development timelines without compromising on quality or consistency.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the replacement of hazardous trifluorodiazoethane with safer imidoyl chlorides represents a significant improvement in process safety. The generation of waste is minimized due to the high atom efficiency of the cyclization and the lack of metal-contaminated waste streams, simplifying compliance with increasingly stringent environmental regulations. The ability to run the reaction in common solvents like dichloroethane, which can be efficiently recovered and recycled, further supports sustainability goals. This alignment with green chemistry principles makes the technology attractive for long-term commercial partnerships focused on sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on reaction parameters and substrate compatibility. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing workflows.
Q: Does this synthesis require expensive heavy metal catalysts?
A: No, the patented method utilizes elemental iodine as a promoter instead of toxic or expensive transition metal catalysts, significantly simplifying purification and reducing environmental impact.
Q: What are the reaction conditions regarding moisture and oxygen?
A: The process is robust and does not require strict anhydrous or anaerobic conditions, making it highly suitable for standard industrial reactor setups without specialized inert atmosphere equipment.
Q: Can this method accommodate diverse substrate substituents?
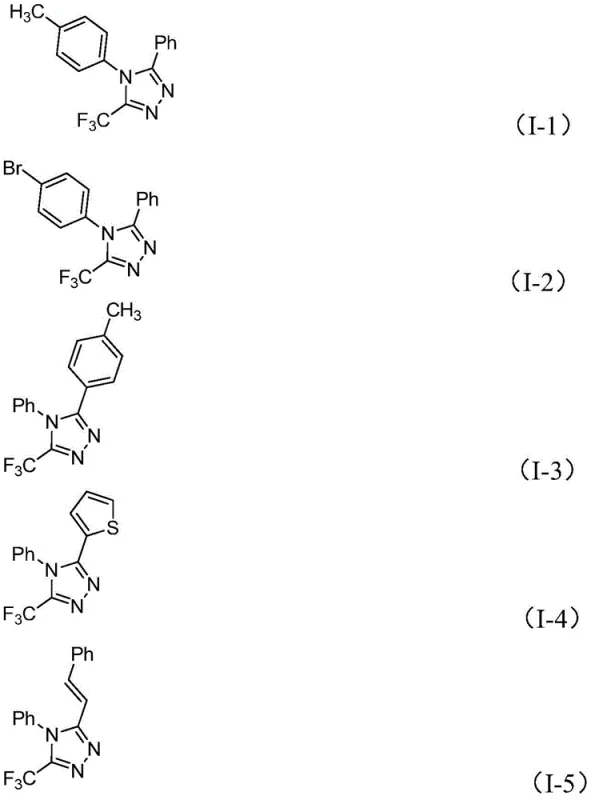
A: Yes, the method demonstrates excellent functional group tolerance, successfully synthesizing derivatives with methyl, methoxy, bromo, nitro, and heteroaryl groups on both the N1 and C5 positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify identity and assay. By leveraging advanced synthetic technologies like the iodine-promoted cyclization described in CN110467579B, we can offer competitive pricing and consistent supply for complex heterocyclic building blocks.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new targets, our experts are ready to provide tailored solutions. Contact us today to explore how our manufacturing capabilities can enhance your R&D productivity and streamline your path to market.
