Advanced NTFTS-Mediated Trifluoroacetylation for Scalable Pharmaceutical Intermediate Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to introduce fluorine-containing motifs into complex molecular architectures, driven by the profound impact of fluorination on metabolic stability and lipophilicity. Patent CN110041235B introduces a significant technological breakthrough in this domain by disclosing N-phenyl-N-p-toluenesulfonyl trifluoroacetamide, abbreviated as NTFTS, as a superior trifluoroacetylating reagent. This novel reagent facilitates the efficient and highly selective conversion of arylboronic acid derivatives into trifluoroacetophenone compounds under mild transition metal catalysis. Unlike traditional methods that rely on hazardous gases or harsh Lewis acids, this innovation leverages a stable solid reagent that simplifies operational protocols while maintaining exceptional yield profiles. For R&D directors and process chemists, this represents a pivotal shift towards safer, more controllable synthetic routes for high-value intermediates used in drug discovery and agrochemical development.
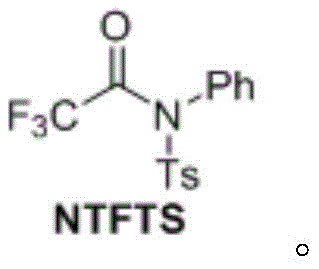
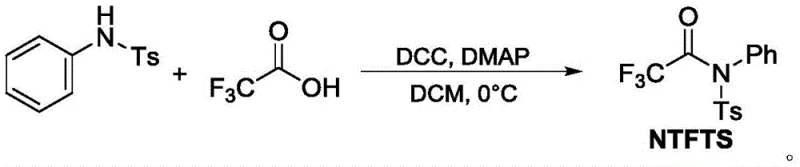
The structural integrity and stability of the NTFTS reagent are central to its utility, offering a distinct advantage over volatile liquid or gaseous alternatives. By incorporating a tosyl group and a phenyl ring, the reagent achieves a balance of reactivity and stability that allows it to be stored and handled with standard laboratory precautions. This stability translates directly into supply chain reliability, as the reagent does not require specialized cryogenic storage or gas handling infrastructure. Furthermore, the synthesis of NTFTS itself is straightforward, involving the condensation of N-phenyl-p-toluenesulfonamide with trifluoroacetic acid using DCC and DMAP, ensuring that the precursor supply remains robust and cost-effective for large-scale manufacturing needs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoroacetophenones has been plagued by significant operational and safety challenges inherent to classical organic transformations. The Friedel-Crafts acylation, a staple in aromatic substitution, typically necessitates the use of corrosive and hazardous reagents such as trifluoroacetyl chloride gas or trifluoroacetic anhydride in the presence of strong Lewis acids like aluminum chloride or sulfuric acid. These conditions are not only environmentally detrimental due to the generation of substantial acidic waste streams but also pose severe safety risks regarding equipment corrosion and operator exposure. Additionally, the Grignard reaction pathway requires the preparation of air- and moisture-sensitive organometallic reagents, demanding rigorous anhydrous conditions and limiting functional group tolerance. Oxidation methods, while viable, often rely on stoichiometric amounts of heavy metal oxidants, leading to complex purification processes and significant environmental burdens that contradict modern green chemistry principles.
The Novel Approach
In stark contrast, the methodology disclosed in CN110041235B utilizes the NTFTS reagent to effect trifluoroacetylation through a palladium-catalyzed cross-coupling mechanism that operates under remarkably mild conditions. This novel approach eliminates the need for gaseous reagents and strong acids, replacing them with a stable solid amide that reacts efficiently at temperatures ranging from 25°C to 50°C. The reaction proceeds in common anhydrous organic solvents such as toluene, tetrahydrofuran, or 1,4-dioxane, utilizing readily available bases like potassium carbonate or cesium carbonate. This shift from harsh electrophilic substitution to catalytic cross-coupling dramatically improves the functional group compatibility, allowing for the preservation of sensitive moieties such as esters, nitriles, and halides that would otherwise be compromised. The result is a streamlined process that reduces waste, enhances safety, and delivers high-purity products with minimal downstream processing requirements.
Mechanistic Insights into Palladium-Catalyzed Trifluoroacetylation
The core of this technological advancement lies in the palladium-catalyzed activation of the NTFTS reagent, which serves as an electrophilic source of the trifluoroacetyl group. The catalytic cycle likely initiates with the oxidative addition of the palladium(0) species into the N-C(O) bond of the NTFTS, or alternatively, involves a transmetallation sequence where the arylboronic acid transfers the aryl group to the palladium center first. Subsequent coordination and insertion steps facilitate the formation of the carbon-carbon bond between the aryl ring and the trifluoroacetyl moiety. The presence of bulky phosphine ligands, such as tri-tert-butylphosphine or tricyclohexylphosphine, plays a critical role in stabilizing the active palladium species and promoting the reductive elimination step that releases the final trifluoroacetophenone product. This mechanistic pathway ensures high turnover numbers and minimizes catalyst deactivation, which is crucial for maintaining economic viability in industrial settings.

From an impurity control perspective, this catalytic system offers superior selectivity compared to non-catalytic alternatives. The mild reaction conditions prevent the occurrence of side reactions such as poly-acylation or decomposition of the trifluoroacetyl group, which are common pitfalls in Friedel-Crafts reactions. Furthermore, the byproduct generated from the NTFTS reagent, N-phenyl-p-toluenesulfonamide, is a stable solid that can be easily separated from the desired ketone product via standard aqueous workup or crystallization techniques. This clean reaction profile significantly reduces the burden on purification teams, allowing for the isolation of high-purity intermediates suitable for direct use in subsequent synthetic steps without extensive chromatographic purification. The ability to tolerate a wide range of substituents on the arylboronic acid, including electron-withdrawing groups like nitro and cyano, further underscores the robustness of this method for diverse medicinal chemistry applications.
How to Synthesize Trifluoroacetophenones Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant environments. The protocol involves charging a reactor with the palladium catalyst, ligand, base, and NTFTS reagent under an inert nitrogen atmosphere to prevent catalyst oxidation. Following the addition of the anhydrous solvent and the arylboronic acid substrate, the mixture is stirred at moderate temperatures for 16 to 24 hours. The detailed standardized synthesis steps, including specific molar ratios and workup procedures validated by patent examples, are outlined below to ensure reproducibility and optimal yield.
- Prepare the reaction vessel by charging palladium catalyst (e.g., Pd(OAc)2), ligand (e.g., P(t-Bu)3), base (e.g., K2CO3), and the NTFTS reagent under inert atmosphere.
- Add anhydrous organic solvent such as toluene or 1,4-dioxane, followed by the arylboronic acid derivative substrate.
- Stir the mixture at mild temperatures between 25°C and 50°C for 16 to 24 hours, then purify the resulting trifluoroacetophenone via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the NTFTS-mediated trifluoroacetylation process offers tangible strategic benefits that extend beyond mere chemical efficiency. The transition from hazardous gaseous reagents to a stable solid reagent fundamentally alters the logistics of raw material management, eliminating the need for specialized gas cylinders and reducing regulatory compliance costs associated with toxic inhalants. Moreover, the use of commodity chemicals such as arylboronic acids and common palladium catalysts ensures a resilient supply chain that is less susceptible to the volatility often seen with exotic or proprietary reagents. This stability in sourcing translates to more predictable lead times and reduced risk of production stoppages due to material shortages.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the elimination of expensive and hazardous reagents like trifluoroacetyl chloride, which often incur high handling and disposal costs. By utilizing NTFTS, manufacturers can avoid the capital expenditure associated with corrosion-resistant reactors and scrubbing systems required for acidic gas handling. Additionally, the high yields reported in the patent examples, reaching up to 98% in certain substrates, minimize raw material waste and maximize the throughput of valuable intermediates. The simplified workup procedure, which avoids complex quenching of strong acids, further reduces labor costs and solvent consumption, contributing to a lower overall cost of goods sold (COGS) for the final API or intermediate.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents like NTFTS significantly mitigates the risks associated with reagent degradation during storage and transport. Unlike moisture-sensitive anhydrides or air-sensitive organometallics, NTFTS can be stocked in bulk without stringent environmental controls, providing a buffer against supply disruptions. The broad availability of arylboronic acids from multiple global suppliers further diversifies the supply base, preventing single-source dependency. This robustness ensures continuous production capability, which is critical for meeting the demanding delivery schedules of pharmaceutical clients and maintaining inventory levels for key therapeutic programs.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage quantities is facilitated by the mild thermal profile and the absence of exothermic hazards typical of Friedel-Crafts reactions. The operation at near-ambient temperatures (25-50°C) reduces energy consumption for heating or cooling, aligning with sustainability goals and lowering utility costs. Furthermore, the reduction in chemical waste, particularly acidic effluents and heavy metal oxidants, simplifies wastewater treatment and helps facilities maintain strict environmental compliance standards. This eco-friendly profile not only reduces disposal fees but also enhances the corporate sustainability image, which is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel trifluoroacetylation technology. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on process capabilities and limitations. Understanding these details is essential for project managers evaluating the feasibility of integrating this route into existing manufacturing pipelines.
Q: What are the advantages of NTFTS over traditional trifluoroacetylating agents?
A: Unlike gaseous trifluoroacetyl chloride or moisture-sensitive anhydrides, NTFTS is a stable, solid reagent that allows for safer handling, easier storage, and milder reaction conditions without generating excessive acidic waste.
Q: What is the substrate scope for this trifluoroacetylation method?
A: The method exhibits broad compatibility with various arylboronic acids, including those with electron-donating groups (alkyl, alkoxy), electron-withdrawing groups (esters, halides, nitro), and sterically hindered biaryl or naphthyl systems.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes commercially available catalysts and operates at low temperatures (25-50°C) in common solvents, making it highly scalable with simplified post-treatment procedures compared to Friedel-Crafts acylation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoroacetophenone Supplier
The technological potential of NTFTS-mediated trifluoroacetylation is immense, offering a pathway to high-value fluorinated intermediates that are critical for modern drug discovery. NINGBO INNO PHARMCHEM stands ready to leverage this advanced chemistry, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of trifluoroacetophenone intermediate meets the exacting standards required for GMP manufacturing. We understand the complexities of fluorine chemistry and have the expertise to optimize these reactions for maximum efficiency and safety.
We invite you to collaborate with us to explore how this innovative process can enhance your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can support your development timelines and commercial goals with reliable, high-quality chemical solutions.
