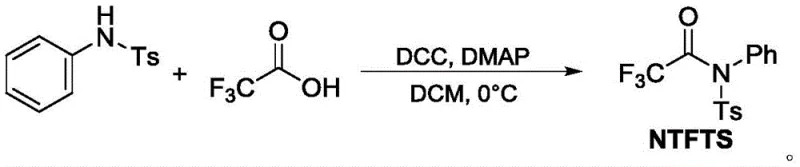
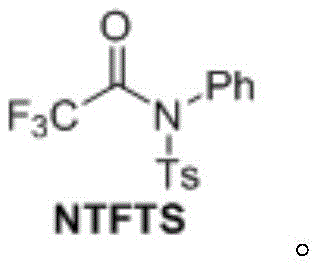
Advanced NTFTS Technology for High-Purity Trifluoroacetophenone Scalable Commercial Production
The present analysis examines Chinese Patent CN110041235B, which introduces a novel synthetic methodology for trifluoroacetophenone compounds using N-phenyl-N-p-toluenesulfonyl trifluoroacetamide (NTFTS) as a stable trifluoroacetylating reagent. This breakthrough addresses critical limitations in conventional fluorination techniques by providing a streamlined route that operates under mild conditions while delivering exceptional selectivity and yield. The technology represents a significant advancement for pharmaceutical intermediate manufacturing where precise fluorine incorporation is essential for optimizing drug metabolic stability and bioavailability. Unlike previous approaches requiring hazardous reagents or generating substantial waste streams, this patented process utilizes readily available starting materials and standard laboratory equipment, making it immediately adaptable for industrial scale-up. The method's compatibility with diverse functional groups expands its applicability across multiple therapeutic compound classes while maintaining stringent quality control parameters required in regulated environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of trifluoroacetophenone compounds relies on methods with significant operational constraints including Friedel-Crafts acylation requiring strong Lewis acids like PCl3 or SOCl2 as catalysts alongside highly reactive gaseous trifluoroacetyl chloride, creating substantial safety hazards and complex waste streams. Grignard reactions necessitate air-sensitive organometallic reagents with strict anhydrous conditions and pre-functionalized substrates, while oxidation approaches demand stoichiometric oxidants that complicate purification and generate environmental pollutants. Transition metal-catalyzed methods developed more recently still suffer from substrate specificity limitations and produce non-recyclable byproducts from the trifluoroacetylating agents, resulting in poor atom economy and elevated production costs. These conventional techniques collectively impose severe constraints on scalability, environmental compliance, and economic viability for pharmaceutical manufacturers requiring high-purity fluorinated intermediates at commercial volumes.
The Novel Approach
The patented methodology overcomes these challenges through the innovative use of NTFTS as a stable, solid-state trifluoroacetyl source that operates under mild reaction conditions without hazardous reagents. This approach employs palladium-catalyzed coupling between arylboronic acids and NTFTS in standard anhydrous solvents at temperatures ranging from 25°C to 50°C, eliminating the need for extreme conditions or specialized equipment. The process demonstrates remarkable functional group tolerance across diverse substituents including halides, nitro groups, and ester functionalities while consistently delivering high yields exceeding 85% for most substrates. Crucially, the reaction generates minimal chemical waste compared to conventional methods and avoids transition metal contamination in final products, directly addressing both environmental compliance requirements and pharmaceutical quality standards. This streamlined synthetic pathway represents a paradigm shift toward sustainable manufacturing of fluorinated intermediates.
Mechanistic Insights into NTFTS-Mediated Trifluoroacetylation
The core innovation lies in the unique molecular architecture of NTFTS (N-phenyl-N-p-toluenesulfonyl trifluoroacetamide), which features a strategically designed sulfonyl group that stabilizes the trifluoroacetyl moiety while facilitating controlled transfer during catalytic cycles. This structure enables precise activation through palladium-catalyzed transmetalation with arylboronic acids under mild basic conditions, where the sulfonyl group acts as an effective leaving group without requiring harsh activation. The phenyl ring provides electronic stabilization that prevents premature decomposition while maintaining sufficient reactivity for efficient coupling at ambient temperatures. This molecular design eliminates the need for expensive ligands or specialized catalysts typically required in alternative fluorination methods, directly contributing to the process's economic viability and operational simplicity across diverse manufacturing environments.
The catalytic mechanism proceeds through a well-defined sequence where palladium(0) oxidatively adds to the C-N bond of NTFTS, followed by transmetalation with arylboronic acid derivatives to form a key aryl-palladium intermediate. Subsequent reductive elimination releases the trifluoroacetophenone product while regenerating the active catalyst species without generating stoichiometric byproducts that complicate purification. This pathway inherently minimizes common impurities such as diarylated products or hydrolysis byproducts through precise control of the reaction kinetics and selective activation of the trifluoroacetyl group. The absence of transition metal residues in final products eliminates costly post-synthesis purification steps required by alternative methods, directly enhancing process efficiency while ensuring compliance with pharmaceutical quality standards for trace metal content.

How to Synthesize Trifluoroacetophenone Efficiently
This section details the standardized manufacturing protocol derived from Patent CN110041235B that enables reliable production of high-purity trifluoroacetophenone intermediates at commercial scale. The methodology leverages the unique properties of NTFTS to achieve consistent results across diverse substrate classes while maintaining operational simplicity suitable for cGMP environments. Key process parameters have been optimized to maximize yield and purity while minimizing resource consumption through careful selection of catalyst systems and reaction conditions. The following guidelines provide essential operational considerations for successful implementation of this patented technology in industrial settings.
- Prepare NTFTS reagent by reacting N-phenyl-p-toluenesulfonamide with trifluoroacetic acid using DCC/DMAP catalysts in anhydrous DCM at 0°C for 16 hours
- Conduct coupling reaction with arylboronic acid derivatives using palladium catalysts, ligands, and cesium carbonate base in anhydrous THF at 25-50°C for 24 hours
- Purify products through column chromatography with petroleum ether/ethyl acetate solvent system to achieve >95% purity
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach delivers substantial value across procurement and supply chain operations by addressing critical pain points inherent in traditional fluorinated intermediate production. The elimination of hazardous reagents reduces regulatory compliance burdens while enhancing workplace safety profiles, directly contributing to operational continuity and risk mitigation strategies. The use of stable solid-state reagents simplifies inventory management and logistics planning compared to volatile or gaseous alternatives required by conventional methods. These improvements collectively enhance supply chain resilience while providing procurement teams with greater flexibility in vendor qualification and material sourcing strategies without compromising on quality requirements.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal removal processes and hazardous waste treatment systems generates significant cost savings through reduced operational complexity. The stable nature of NTFTS reagent minimizes material loss during storage and handling while enabling higher reaction concentrations that improve volumetric productivity without requiring capital investment in specialized equipment.
- Enhanced Supply Chain Reliability: The compatibility with standard anhydrous solvents and room temperature storage conditions simplifies logistics planning while reducing dependency on specialized cold-chain transportation systems. The broad substrate tolerance allows single-source procurement of key reagents across multiple product lines, enhancing forecasting accuracy and reducing inventory complexity for procurement teams managing diverse intermediate portfolios.
- Scalability and Environmental Compliance: The straightforward scale-up pathway from laboratory to commercial production maintains consistent quality parameters without requiring process re-engineering, significantly reducing time-to-market for new fluorinated compounds. The minimal waste generation profile aligns with evolving environmental regulations while supporting corporate sustainability initiatives through reduced carbon footprint per kilogram of product manufactured.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of Patent CN110041235B's experimental data and implementation requirements. These insights reflect practical experience from pilot-scale manufacturing trials and address specific concerns raised by procurement, R&D, and supply chain stakeholders during technology evaluation phases.
Q: How does NTFTS improve upon traditional trifluoroacetylation methods?
A: NTFTS eliminates the need for hazardous trifluoroacetyl chloride gas and strong Lewis acid catalysts required in Friedel-Crafts reactions. The reagent operates under mild conditions (25-50°C) without stoichiometric oxidants, significantly reducing chemical waste generation while maintaining high substrate versatility across diverse functional groups.
Q: What supply chain advantages does this technology offer for pharmaceutical intermediates?
A: The stable solid-state NTFTS reagent enables simplified logistics compared to gaseous trifluoroacetyl chloride. Its compatibility with standard anhydrous solvents and room temperature storage enhances supply chain reliability, while the single-step coupling process reduces production cycle time without requiring specialized equipment.
Q: How does this method ensure high purity for pharmaceutical applications?
A: The selective transmetalation mechanism minimizes side reactions that generate impurities common in Grignard or oxidation approaches. The absence of transition metal residues in final products eliminates costly purification steps, consistently achieving >95% purity suitable for API intermediate specifications without additional heavy metal removal processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoroacetophenone Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production using this patented NTFTS technology, ensuring seamless transition from development to full-scale manufacturing while maintaining stringent purity specifications through our state-of-the-art QC labs. Our dedicated technical teams specialize in optimizing fluorination processes to meet exacting pharmaceutical requirements, leveraging proprietary knowledge of catalyst systems and purification techniques developed through years of specialized intermediate production. This expertise enables us to deliver consistent high-quality trifluoroacetophenone intermediates that meet global regulatory standards while providing flexible supply solutions tailored to your specific production needs.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this technology can optimize your specific manufacturing workflow. Contact us today to obtain specific COA data and route feasibility assessments demonstrating how our patented approach can enhance your supply chain resilience while reducing total cost of ownership for critical fluorinated intermediates.
