Advanced NTFTS Reagent Technology for Scalable Trifluoroacetophenone Synthesis
Advanced NTFTS Reagent Technology for Scalable Trifluoroacetophenone Synthesis
The landscape of fluorinated organic synthesis is undergoing a significant transformation driven by the urgent need for safer, more efficient, and environmentally benign methodologies. A pivotal advancement in this domain is detailed in Chinese Patent CN110041235B, which introduces N-phenyl-N-p-toluenesulfonyl trifluoroacetamide, commonly abbreviated as NTFTS, as a superior trifluoroacetylating agent. This novel reagent addresses critical bottlenecks in the production of trifluoroacetophenone compounds, which are indispensable scaffolds in the development of high-performance pharmaceuticals and agrochemicals. By leveraging a robust palladium-catalyzed cross-coupling strategy, this technology enables the direct introduction of the trifluoroacetyl group onto aryl rings with exceptional selectivity and yield. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic elegance and operational simplicity of this patent is crucial for optimizing supply chains and reducing overall manufacturing costs.
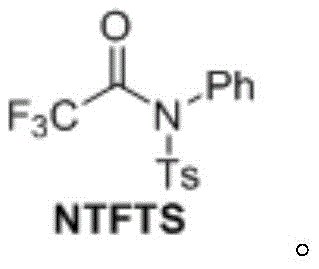
The structural integrity and stability of the NTFTS reagent are fundamental to its commercial viability. As depicted in the molecular structure, the presence of the tosyl (Ts) group and the phenyl ring creates a sterically and electronically balanced environment that stabilizes the trifluoroacetyl moiety until activation. This stability allows the reagent to be stored and transported without the stringent safety protocols required for gaseous or highly reactive liquid acylating agents. Furthermore, the design of this molecule facilitates a clean reaction profile where the leaving group is well-defined, minimizing the formation of difficult-to-remove impurities. This level of control is paramount for manufacturers aiming to produce high-purity trifluoroacetophenone derivatives that meet the rigorous quality standards of the global pharmaceutical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoroacetophenones has relied on methodologies that are increasingly untenable in modern green chemistry frameworks. Traditional Friedel-Crafts acylation, for instance, necessitates the use of corrosive strong acids or potent Lewis acids such as thionyl chloride or phosphorus trichloride, often requiring the handling of hazardous trifluoroacetyl chloride gas. These conditions not only pose severe safety risks to personnel and infrastructure but also generate substantial quantities of acidic chemical waste that require costly disposal procedures. Alternatively, Grignard reactions involve air-sensitive organometallic reagents that demand strictly anhydrous and inert conditions, limiting their practicality for large-scale operations. Oxidation methods, while avoiding some of these issues, often suffer from poor atom economy and require stoichiometric amounts of oxidants, leading to complex post-reaction workups and potential environmental contamination.
The Novel Approach
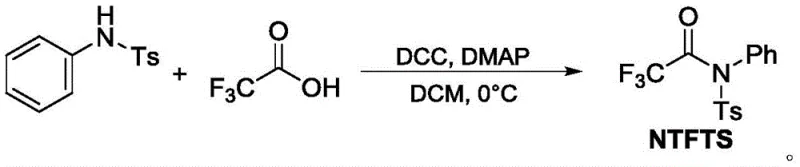
In stark contrast, the novel approach utilizing NTFTS represents a paradigm shift towards sustainable and operationally simple synthesis. The preparation of the NTFTS reagent itself is straightforward, involving the condensation of N-phenyl-p-toluenesulfonamide with trifluoroacetic acid using DCC and DMAP in dichloromethane at 0°C. This precursor synthesis is high-yielding and produces a stable solid that serves as a convenient source of the trifluoroacetyl group. When employed in the subsequent coupling reaction, NTFTS eliminates the need for harsh acidic conditions or sensitive organometallics. Instead, it participates in a mild transition-metal catalyzed cycle that tolerates a wide array of functional groups. This shift from hazardous, waste-intensive processes to a catalytic, modular system significantly lowers the barrier to entry for producing complex fluorinated intermediates, offering substantial cost savings and enhanced safety profiles for chemical manufacturers.
Mechanistic Insights into Palladium-Catalyzed Trifluoroacetylation
The core of this technological breakthrough lies in the efficient palladium-catalyzed cross-coupling mechanism between the arylboronic acid and the NTFTS reagent. The reaction proceeds through a catalytic cycle where the palladium species, typically generated from precursors like palladium acetate or tris(dibenzylideneacetone)dipalladium, undergoes oxidative addition or transmetallation steps facilitated by specialized ligands such as tri-tert-butylphosphine or tricyclohexylphosphine. The choice of ligand is critical, as it modulates the electronic density and steric bulk around the metal center, thereby accelerating the rate-determining steps and preventing catalyst deactivation. The base, such as potassium carbonate or cesium carbonate, plays a dual role in activating the boronic acid for transmetallation and neutralizing acidic by-products, ensuring the catalytic cycle continues uninterrupted. This sophisticated interplay of catalyst, ligand, and base allows the reaction to proceed smoothly at mild temperatures ranging from 25°C to 50°C.
From an impurity control perspective, the mechanism offers distinct advantages over classical electrophilic substitutions. Because the reaction is catalytic and highly selective, side reactions such as poly-acylation or rearrangement of the carbon skeleton are virtually eliminated. The by-product derived from the NTFTS leaving group is typically a sulfonamide derivative, which is chemically distinct from the target ketone and can be easily separated via standard purification techniques like column chromatography or recrystallization. This clean reaction profile ensures that the final trifluoroacetophenone products possess high chemical purity, reducing the burden on downstream purification processes. For process chemists, this means fewer unit operations, reduced solvent consumption, and a more robust process capable of delivering consistent quality batch after batch, which is essential for regulatory compliance in pharmaceutical manufacturing.

How to Synthesize Trifluoroacetophenone Efficiently
The implementation of this synthesis route is designed for practicality and scalability, making it accessible for both laboratory discovery and industrial production. The protocol involves charging a reactor with the palladium catalyst, phosphine ligand, inorganic base, and the NTFTS reagent under an inert nitrogen atmosphere to prevent oxidation of the sensitive catalytic species. Anhydrous solvents such as toluene, 1,4-dioxane, or tetrahydrofuran are selected to maintain catalyst activity and solubility of the reactants. Once the mixture is equilibrated, the arylboronic acid derivative is introduced, and the system is heated gently to promote the coupling. The reaction times typically span 16 to 24 hours, allowing for complete conversion even with sterically hindered substrates. Following the reaction, the workup is uncomplicated, involving filtration to remove inorganic salts and chromatographic purification to isolate the pure ketone. Detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction mixture by combining the metal catalyst (e.g., Pd(OAc)2), ligand (e.g., P(t-Bu)3), base (e.g., K2CO3), and NTFTS reagent in an anhydrous organic solvent like toluene or dioxane under nitrogen atmosphere.
- Add the arylboronic acid derivative to the stirred mixture and maintain the reaction temperature between 25°C and 50°C for 16 to 24 hours to ensure complete conversion.
- Upon completion, purify the crude trifluoroacetophenone product using standard separation techniques such as silica gel column chromatography or recrystallization to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the NTFTS-based methodology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Unlike traditional methods that rely on volatile, hazardous, or import-restricted reagents like trifluoroacetyl chloride, NTFTS is a stable solid that can be sourced reliably and stored safely for extended periods. This stability mitigates the risk of supply disruptions caused by transportation regulations or storage limitations, ensuring a continuous flow of materials for production schedules. Furthermore, the elimination of expensive and toxic Lewis acids reduces the dependency on specialized corrosion-resistant equipment, allowing for the utilization of standard stainless steel reactors which are more readily available and cost-effective to maintain.
- Cost Reduction in Manufacturing: The economic impact of switching to this novel reagent is profound, primarily driven by the reduction in waste treatment costs and the optimization of raw material usage. By avoiding the generation of large volumes of acidic waste associated with Friedel-Crafts reactions, facilities can significantly lower their environmental compliance expenditures. Additionally, the high yields reported in the patent examples, often exceeding 90% for various substrates, mean that less starting material is wasted, directly improving the cost per kilogram of the final product. The ability to operate at near-room temperature also results in substantial energy savings compared to processes requiring cryogenic conditions or high-temperature reflux, further enhancing the overall cost reduction in API manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the NTFTS reagent contributes to a more resilient supply chain capable of withstanding market fluctuations. Since the reagent is synthesized from cheap and commercially available starting materials like N-phenyl-p-toluenesulfonamide and trifluoroacetic acid, the risk of raw material scarcity is minimized. This accessibility ensures that manufacturers can scale production up or down based on demand without facing bottlenecks related to reagent availability. Moreover, the mild reaction conditions reduce the likelihood of batch failures due to thermal runaways or sensitivity to moisture, leading to more predictable lead times and reliable delivery schedules for customers relying on high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden challenges, but the NTFTS method is inherently designed for scalability. The use of heterogeneous bases and common organic solvents simplifies the workup and solvent recovery processes, which are critical factors in large-scale operations. From an environmental standpoint, the process aligns with green chemistry principles by reducing the E-factor (mass of waste per mass of product) and eliminating the use of heavy metal oxidants or corrosive gases. This alignment not only facilitates easier regulatory approval but also enhances the corporate sustainability profile of the manufacturer, appealing to end-clients who prioritize eco-friendly sourcing in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing portfolios.
Q: What are the primary advantages of NTFTS over traditional trifluoroacetylating reagents?
A: Unlike traditional reagents like trifluoroacetyl chloride which require harsh Lewis acids and generate significant waste, NTFTS is a stable, solid reagent that operates under mild transition-metal catalyzed conditions, significantly reducing environmental impact and operational hazards.
Q: What is the substrate scope for this trifluoroacetylation method?
A: The method demonstrates excellent functional group tolerance, successfully reacting with arylboronic acids containing alkyl, alkoxy, ester, phenoxy, vinyl, naphthyl, halide, nitro, and cyano groups, making it highly versatile for complex molecule synthesis.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes cheap and easily available raw materials, avoids air-sensitive reagents like Grignard compounds, and operates at mild temperatures (25-50°C), which facilitates safe and efficient commercial scale-up of complex fluorinated intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable NTFTS Supplier
The technological potential of N-phenyl-N-p-toluenesulfonyl trifluoroacetamide is immense, offering a pathway to superior fluorinated building blocks that drive innovation in drug discovery and material science. At NINGBO INNO PHARMCHEM, we pride ourselves on being at the forefront of adopting such advanced synthetic methodologies to serve our global clientele. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory curiosity to industrial reality is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of NTFTS or derived trifluoroacetophenone meets the highest international standards for pharmaceutical and agrochemical applications.
We invite forward-thinking organizations to collaborate with us to leverage this cutting-edge chemistry for their specific project needs. Whether you require custom synthesis of novel analogs or bulk supply of established intermediates, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for efficiency, cost, and reliability in the competitive global market.
