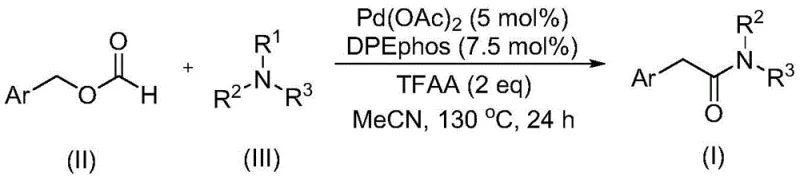
Advanced Palladium-Catalyzed Synthesis of High-Purity Aryl Acetamide Compounds for Commercial Scale-Up
This patent CN111978194B introduces a groundbreaking methodology for the synthesis of aryl acetamide compounds, a critical class of molecules with extensive applications in pharmaceutical development due to their physiological and pharmacological activities. The innovation centers on a palladium-catalyzed carbonylation process that uniquely employs benzyl formate as both a carbon monoxide source and reactant while utilizing tertiary amines as the amine source—a significant advancement over conventional approaches that typically require primary or secondary amines. This approach eliminates the need for additional oxidants typically required in C-N bond cleavage reactions, addressing a major limitation in existing methodologies where explosive CO/O₂ mixtures or excessive oxidants create substantial safety hazards and operational complexities. The process demonstrates exceptional substrate flexibility with broad functional group tolerance across diverse aryl substituents including alkyl, alkoxy, trifluoromethyl, cyano, and halogen groups at ortho, meta, or para positions. With optimized reaction parameters at 130°C for 24 hours in acetonitrile solvent using palladium acetate (5 mol%) and DPEphos ligand (7.5 mol%), this method achieves high yields while maintaining operational simplicity and cost-effectiveness through commercially available starting materials that are cheap and easy to obtain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing aryl acetamide compounds primarily rely on amidation reactions of phenylacetic acid derivatives which suffer from multiple limitations including harsh reaction conditions that often require elevated temperatures or strong acids/bases that degrade sensitive functional groups. More advanced transition metal-catalyzed carbonylation methods have emerged as promising alternatives but typically require primary or secondary amines as amine sources, significantly restricting substrate diversity and limiting access to complex tertiary amine-containing structures essential for many pharmaceutical applications. Critically, existing methodologies for tertiary amine C-N bond activation frequently necessitate the use of excessive oxidants or explosive CO/O₂ mixtures that create substantial safety hazards requiring specialized equipment and stringent operational protocols that complicate process scale-up and increase capital investment costs. These constraints not only increase production costs through additional purification steps needed to remove oxidation byproducts but also limit the structural diversity of accessible aryl acetamide compounds due to narrow functional group tolerance under harsh oxidative conditions.
The Novel Approach
The patented methodology overcomes these limitations through an innovative palladium-catalyzed system that operates without additional oxidants while utilizing tertiary amines directly as amine sources—a breakthrough that addresses a significant gap in current synthetic methodologies. By employing benzyl formate as a dual-function reagent serving as both carbon monoxide source and reactant, the process eliminates the need for handling hazardous gas mixtures while maintaining high reaction efficiency through an integrated carbonylation mechanism that avoids external CO infrastructure requirements. The optimized catalyst system comprising palladium acetate (5 mol%) and DPEphos ligand (7.5 mol%) in trifluoroacetic anhydride medium enables C-N bond cleavage under mild thermal conditions (130°C) with exceptional functional group compatibility across diverse substrates including those containing sensitive groups like cyano or halogen substituents that would typically degrade under conventional oxidative conditions. This approach accommodates a wide range of aryl substituents at various positions while tolerating diverse tertiary amine structures with C₂-C₅ alkyl substituents without requiring structural modifications to either component.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The catalytic cycle begins with oxidative addition of palladium(0) into the C-O bond of benzyl formate (II), generating an acyl-palladium intermediate that subsequently undergoes decarbonylation to form a palladium-carbene complex—a key innovation that enables carbon monoxide transfer without external gas sources. This carbene intermediate then coordinates with the tertiary amine substrate (III), facilitating C-N bond cleavage through a β-hydride elimination pathway that does not require external oxidants—a significant advancement over conventional methods where oxidation steps typically generate problematic byproducts requiring extensive purification. The resulting iminium ion intermediate is captured by the acyl-palladium species to form the final aryl acetamide product (I) after reductive elimination regenerates the active palladium(0) catalyst completing the catalytic cycle. This mechanism explains the exceptional functional group tolerance observed across diverse substrates including those with electron-donating or electron-withdrawing substituents at various positions on the aromatic ring.
Impurity profile control is achieved through precise stoichiometric balance between benzyl formate (1 equiv), tertiary amine (5 equiv), and palladium catalyst (0.05 mol%), which minimizes side reactions such as over-carbonylation or amine decomposition while maintaining high selectivity toward the desired product. The reaction's selectivity is further enhanced by acetonitrile solvent choice which provides optimal polarity to stabilize charged intermediates while facilitating product precipitation during workup without requiring specialized separation techniques. The absence of external oxidants eliminates potential oxidation byproducts that commonly complicate purification in traditional methods, resulting in cleaner reaction profiles with fewer impurities requiring removal during post-treatment processing steps.
How to Synthesize Aryl Acetamide Compounds Efficiently
This patented methodology represents a significant advancement in aryl acetamide synthesis, offering pharmaceutical manufacturers a robust and scalable route to these critical intermediates through an operationally simple process that eliminates traditional limitations associated with tertiary amine C-N bond activation while maintaining exceptional substrate flexibility across diverse molecular architectures. The process leverages commercially available starting materials under mild thermal conditions that facilitate seamless technology transfer from laboratory development to manufacturing scale without requiring specialized equipment modifications or hazardous material handling infrastructure typically needed for conventional carbonylation approaches.
- Combine palladium acetate catalyst (5 mol%), DPEphos ligand (7.5 mol%), trifluoroacetic anhydride (2 equiv), benzyl formate, tertiary amine, and acetonitrile solvent in a sealed reactor
- Heat the mixture to 130°C under nitrogen atmosphere and maintain reaction conditions for 24 hours with continuous stirring
- Perform post-treatment through filtration, silica gel mixing, and column chromatography purification to obtain high-purity aryl acetamide product
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic route addresses critical pain points in pharmaceutical intermediate supply chains by offering a more reliable and cost-effective pathway to high-value aryl acetamide compounds through strategic elimination of hazardous reagents and simplification of manufacturing requirements that directly impact procurement decisions and supply chain performance metrics across global operations.
- Cost Reduction in Manufacturing: Eliminating expensive oxidants and specialized gas handling infrastructure delivers substantial cost savings throughout production by avoiding capital investments in hazardous material containment systems while reducing operational complexity during manufacturing runs. The integrated use of benzyl formate as both carbon monoxide source and reactant eliminates costly CO gas infrastructure requirements while maintaining high reaction efficiency through commercially available feedstocks that are cheap and easy to obtain from multiple global suppliers.
- Enhanced Supply Chain Reliability: Relying on readily available starting materials—formic acid, benzyl alcohol, and common tertiary amines—ensures consistent supply chain performance even during market fluctuations due to multiple sourcing options across different geographic regions that eliminate single-point failure risks associated with specialized reagents required by conventional methods.
- Scalability and Environmental Compliance: Mild reaction conditions (130°C) enable straightforward scale-up from laboratory to commercial production without requiring specialized equipment modifications while eliminating hazardous gas mixtures significantly reduces environmental impact through simplified waste streams that lower waste treatment costs compared to traditional methods requiring extensive purification to remove oxidation byproducts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented aryl acetamide synthesis methodology based on direct analysis of patent specifications and experimental data supporting evidence-based decision-making across procurement, R&D, and supply chain management functions within pharmaceutical manufacturing organizations.
Q: Why doesn't this method require additional oxidants for tertiary amine C-N bond cleavage?
A: The patented mechanism utilizes benzyl formate as both carbon monoxide source and reactant, enabling C-N bond cleavage through a unique β-hydride elimination pathway that doesn't require external oxidants. This eliminates safety hazards associated with explosive CO/O2 mixtures while simplifying the reaction setup.
Q: How does this method achieve broad functional group tolerance?
A: The palladium-catalyzed system operates through neutral intermediates under mild thermal conditions (130°C), avoiding harsh oxidative environments that typically degrade sensitive substituents. This allows compatibility with alkyl, alkoxy, trifluoromethyl, cyano, and halogen groups at various positions on the aryl ring.
Q: What are the key advantages of using benzyl formate as the carbon monoxide source?
A: Benzyl formate serves as both CO source and reactant, eliminating the need for hazardous gas handling infrastructure while maintaining high reaction efficiency. Its commercial availability and low cost significantly improve supply chain reliability compared to traditional methods requiring specialized CO delivery systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Acetamide Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs that ensure consistent quality across all production volumes. This patented methodology represents just one example of our commitment to developing innovative synthetic routes that address the most challenging production requirements for complex pharmaceutical intermediates through advanced catalytic technologies that deliver both operational efficiency and regulatory compliance.
We invite you to request a Customized Cost-Saving Analysis tailored to your specific manufacturing needs from our technical procurement team who can provide detailed information including specific COA data and route feasibility assessments to help evaluate how this innovative synthesis can enhance your supply chain performance while reducing overall production costs.
