Advanced Palladium-Catalyzed Synthesis of Aryl Acetamides for Commercial Scale-Up
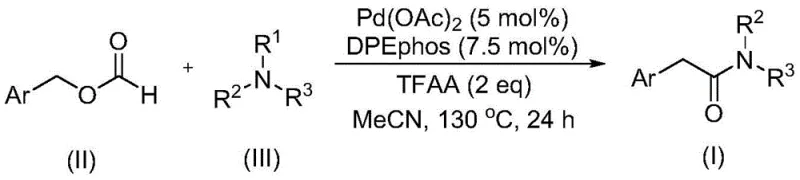
The pharmaceutical and agrochemical industries continuously demand efficient routes to complex amide scaffolds, particularly aryl acetamides, which serve as critical building blocks for bioactive molecules. A significant technological breakthrough in this domain is documented in patent CN111978194B, which discloses a novel preparation method for aryl acetamide compounds. This innovation addresses long-standing challenges in amide synthesis by utilizing a transition metal-catalyzed carbonylation strategy that is both operationally simple and highly effective. The core of this methodology lies in the dual function of benzyl formate, which acts simultaneously as a carbon monoxide source and a reactant, paired with a tertiary amine as the nitrogen source. This approach eliminates the necessity for hazardous gaseous carbon monoxide and avoids the use of excessive oxidants typically required for C-N bond activation, thereby offering a safer and more sustainable pathway for the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of aryl acetamide compounds has relied heavily on the direct amidation of phenylacetic acid and its derivatives. While conceptually straightforward, these classical methods often suffer from significant drawbacks, including harsh reaction conditions, poor atom economy, and limited functional group tolerance. Furthermore, alternative strategies involving transition metal-catalyzed carbonylation have historically been constrained by the use of primary or secondary amines, leaving the activation of tertiary amines as a formidable challenge. Existing protocols for tertiary amine carbonylation frequently require explosive mixtures of carbon monoxide and oxygen or rely on stoichiometric amounts of strong external oxidants, which introduce severe safety hazards and complicate the purification process. These limitations hinder the scalability and economic viability of producing diverse aryl acetamide libraries needed for modern drug discovery and development pipelines.
The Novel Approach
In stark contrast to these legacy techniques, the method described in patent CN111978194B introduces a paradigm shift by employing benzyl formate as a safe, liquid carbon monoxide surrogate. This innovative route enables the efficient coupling of benzyl formate with various tertiary amines under palladium catalysis to yield aryl acetamides with exceptional efficiency. The reaction proceeds smoothly at elevated temperatures without the need for additional oxidants to facilitate the cleavage of the tertiary amine C-N bond, a feat that has eluded many previous attempts. By integrating the carbon source and the reactant into a single, stable molecule, this process drastically simplifies the reaction setup and enhances safety profiles. The versatility of this system allows for the synthesis of a wide array of aryl acetamide derivatives, accommodating various substituents on the aromatic ring and different alkyl groups on the amine nitrogen, thus providing a robust platform for the reliable aryl acetamide supplier seeking to expand their portfolio.
Mechanistic Insights into Pd-Catalyzed Carbonylation
The success of this transformation hinges on a sophisticated catalytic cycle driven by a palladium catalyst, specifically palladium acetate, in conjunction with the bidentate phosphine ligand DPEphos. The mechanism likely involves the oxidative addition of the palladium species into the C-O bond of the benzyl formate or a related activated intermediate generated in situ by trifluoroacetic anhydride (TFAA). TFAA plays a crucial role in activating the formate ester, facilitating the release of carbon monoxide equivalents directly within the coordination sphere of the metal. This in situ generation of CO prevents the accumulation of free gas and ensures high local concentration at the active site. Subsequently, the tertiary amine coordinates to the metal center, where the unique electronic environment created by the DPEphos ligand promotes the difficult C-N bond cleavage. This step is critical, as it allows the nitrogen atom to insert into the acyl-palladium species, forming the new amide bond while releasing the alkyl fragment from the tertiary amine. The reductive elimination step then releases the final aryl acetamide product and regenerates the active palladium catalyst, completing the cycle with high turnover numbers.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over traditional oxidation methods. Because the reaction does not rely on harsh external oxidants that can lead to over-oxidation of sensitive functional groups, the resulting crude product exhibits a cleaner impurity profile. The tolerance of the catalytic system towards various substituents, such as electron-donating methoxy groups, electron-withdrawing trifluoromethyl groups, and halogens, demonstrates the robustness of the ligand-metal complex. This high selectivity minimizes the formation of side products like N-oxides or degraded amine species, which are common pitfalls in tertiary amine functionalization. Consequently, the downstream purification burden is significantly reduced, allowing for the isolation of high-purity OLED material precursors or pharmaceutical intermediates with fewer processing steps. The ability to maintain structural integrity across diverse substrates ensures that the process is suitable for the commercial scale-up of complex polymer additives and fine chemicals.
How to Synthesize Aryl Acetamide Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it highly attractive for process chemistry teams aiming to reduce lead time for high-purity aryl acetamides. The procedure involves charging a sealed vessel with the palladium catalyst, ligand, trifluoroacetic anhydride, benzyl formate, and the chosen tertiary amine in an aprotic organic solvent such as acetonitrile. The mixture is then heated to approximately 130°C and stirred for 24 hours to drive the reaction to completion. Following the reaction, the workup is remarkably straightforward, involving simple filtration to remove catalyst residues, followed by silica gel treatment and standard column chromatography. This streamlined workflow eliminates the need for specialized high-pressure equipment often associated with gas-phase carbonylation, thereby lowering the barrier to entry for laboratories and manufacturing facilities alike. For detailed standardized synthetic steps and specific molar ratios, please refer to the guide below.
- Combine palladium acetate catalyst, DPEphos ligand, trifluoroacetic anhydride, benzyl formate, and tertiary amine in an organic solvent such as acetonitrile.
- Heat the reaction mixture to 130°C and maintain stirring for 24 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity aryl acetamide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into tangible strategic benefits regarding cost stability and operational reliability. The shift from hazardous gaseous reagents to stable liquid surrogates like benzyl formate fundamentally alters the risk profile of the manufacturing process, reducing insurance costs and regulatory compliance burdens associated with toxic gas handling. Furthermore, the use of commercially available and inexpensive starting materials, such as formic acid derivatives and common tertiary amines, ensures a resilient supply chain that is less susceptible to market volatility. The simplicity of the post-treatment process also implies lower labor costs and reduced solvent consumption during purification, contributing to overall process efficiency. These factors collectively enhance the attractiveness of this route for large-scale production, offering a competitive edge in the global market for fine chemical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous external oxidants, combined with the use of cheap benzyl formate as a dual-purpose reagent, leads to substantial cost savings in raw material procurement. Additionally, the high reaction efficiency and selectivity minimize waste generation, reducing the costs associated with waste disposal and environmental compliance. The ability to use standard stainless steel reactors without the need for specialized high-pressure gas infrastructure further lowers capital expenditure requirements for scaling up production capacity.
- Enhanced Supply Chain Reliability: Since all key reagents, including the palladium catalyst and ligands, are readily available from commercial suppliers, the risk of supply disruption is significantly mitigated. The robustness of the reaction conditions allows for consistent batch-to-batch reproducibility, ensuring that delivery schedules can be met with high confidence. This reliability is crucial for maintaining continuous production lines in the pharmaceutical and agrochemical sectors, where delays can have cascading effects on downstream drug development timelines.
- Scalability and Environmental Compliance: The process operates under relatively mild pressure conditions compared to traditional carbonylation, making it inherently safer and easier to scale from kilogram to tonne quantities. The absence of toxic CO gas emissions and the reduced need for stoichiometric oxidants align with green chemistry principles, facilitating easier permitting and environmental approval. This sustainability profile is increasingly important for multinational corporations aiming to meet strict carbon footprint and environmental, social, and governance (ESG) targets in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on reaction parameters and substrate compatibility. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this technology into their existing manufacturing workflows.
Q: What is the primary advantage of using benzyl formate in this synthesis?
A: Benzyl formate serves as a safe and effective carbon monoxide surrogate, eliminating the need for handling toxic CO gas or explosive CO/O2 mixtures while simultaneously acting as a reactant.
Q: Can this method activate the C-N bond of tertiary amines?
A: Yes, unlike conventional methods that struggle with tertiary amines, this palladium-catalyzed protocol effectively cleaves the C-N bond of tertiary amines without requiring additional external oxidants.
Q: What catalyst system is employed for this transformation?
A: The reaction utilizes a robust catalytic system comprising palladium acetate (Pd(OAc)2) and the bidentate ligand DPEphos, which ensures high turnover and substrate tolerance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Acetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN111978194B for the production of high-value intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of aryl acetamide delivered meets the exacting standards required for pharmaceutical and agrochemical applications. We are committed to leveraging cutting-edge chemistry to drive innovation and efficiency for our global partners.
We invite you to collaborate with us to explore how this efficient synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with reliable, high-quality chemical solutions.
