Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Pharmaceutical Intermediates
The landscape of heterocyclic chemistry is constantly evolving to meet the rigorous demands of modern drug discovery, particularly regarding safety profiles and synthetic efficiency. A groundbreaking development detailed in patent CN113121462B introduces a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. This technology represents a paradigm shift away from hazardous traditional protocols, offering a robust, metal-free pathway that leverages readily available diazo compounds and trifluoroethylimidoyl chlorides. For R&D directors and procurement specialists alike, this innovation addresses critical pain points in the supply chain of high-value pharmaceutical intermediates. By eliminating the reliance on explosive azides and expensive transition metal catalysts, this method not only enhances operational safety but also streamlines the purification process, ensuring high-purity outputs suitable for sensitive biological applications.
The strategic importance of the 1,2,3-triazole scaffold cannot be overstated, as it serves as a privileged structure in numerous bioactive molecules, including potent β3 adrenergic receptor agonists and various agrochemical agents. The introduction of a trifluoromethyl group further amplifies the pharmacological potential by improving metabolic stability, lipophilicity, and electronegativity. Consequently, securing a reliable pharmaceutical intermediate supplier capable of delivering these complex fluorinated heterocycles through a sustainable and safe manufacturing route is paramount for maintaining competitive advantage in the global market. The following analysis dissects the technical merits and commercial implications of this patented methodology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has been fraught with significant safety and operational challenges. The literature predominantly describes two main approaches, both of which suffer from inherent drawbacks that hinder large-scale adoption. The first method involves copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides to form triazole metal complexes, which subsequently react with trifluoromethyl reagents. The second approach utilizes organocatalytic 1,3-dipolar cycloaddition between azides and trifluoromethyl ketones. The common denominator in these legacy processes is the reliance on organic azides, which are notoriously toxic and possess high explosion hazards, posing severe risks during storage, transport, and reaction handling. Furthermore, the involvement of transition metals like copper necessitates rigorous downstream processing to remove trace metal residues, a requirement that is increasingly stringent in the pharmaceutical industry to prevent catalyst contamination in the final API.
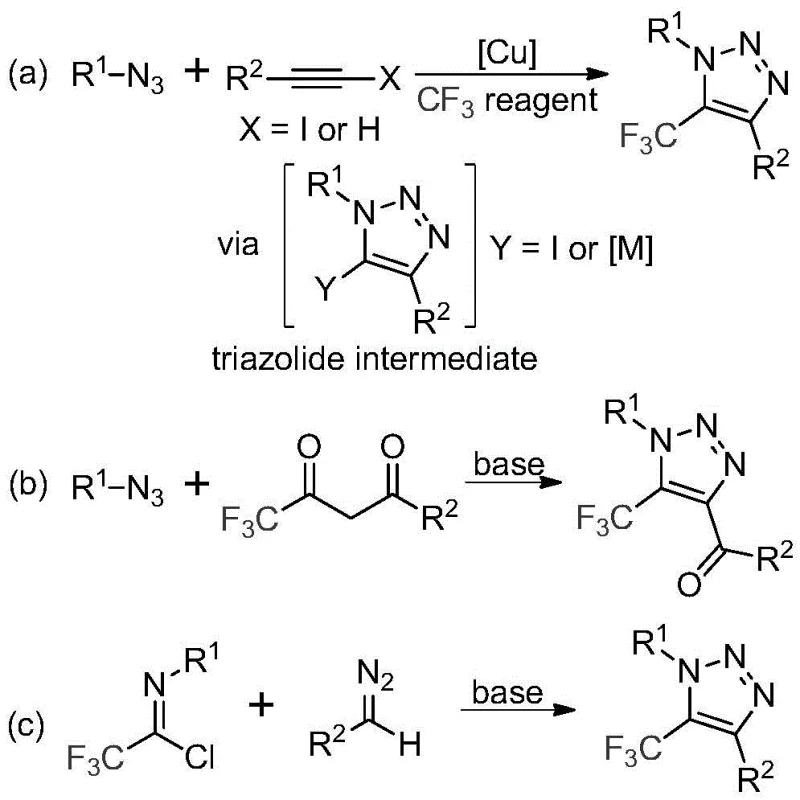
The Novel Approach
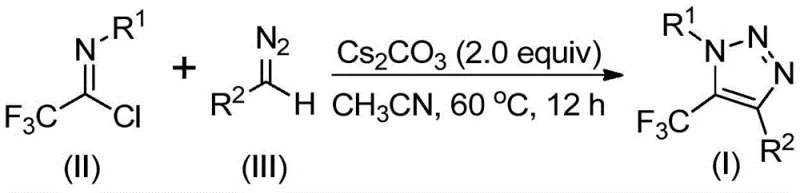
In stark contrast to these hazardous precedents, the methodology disclosed in patent CN113121462B offers a transformative solution by completely bypassing the use of azides and transition metals. This innovative route employs a base-promoted reaction between cheap and readily available diazo compounds and trifluoroethylimidoyl chlorides. As illustrated in the reaction scheme below, the process operates under mild thermal conditions, typically between 50°C and 70°C, utilizing cesium carbonate as a benign promoter in common organic solvents like acetonitrile. This shift in reagent strategy fundamentally alters the risk profile of the synthesis, replacing high-energy explosive precursors with stable, commercially accessible starting materials. The elimination of heavy metal catalysts not only simplifies the workup procedure—often requiring only filtration and column chromatography—but also aligns perfectly with green chemistry principles, reducing the environmental footprint associated with heavy metal waste disposal.
Mechanistic Insights into Base-Promoted Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction is hypothesized to proceed through a sophisticated cascade initiated by the base-promoted intermolecular nucleophilic addition of the diazo compound to the trifluoroethylimidoyl chloride. This initial step facilitates the formation of a critical carbon-carbon bond, setting the stage for the subsequent ring closure. Following this addition-elimination sequence, the intermediate undergoes an intramolecular 5-endo-dig cyclization. This specific cyclization mode is electronically favored due to the presence of the electron-withdrawing trifluoromethyl group, which activates the alkyne-like character of the intermediate towards nucleophilic attack by the nitrogen center. The result is the efficient construction of the 1,2,3-triazole core with the trifluoromethyl group precisely positioned at the 5-position. This mechanistic pathway ensures high regioselectivity, minimizing the formation of unwanted isomers that often plague traditional cycloaddition reactions.
From an impurity control perspective, this mechanism offers distinct advantages. The absence of radical pathways, which are common in metal-catalyzed processes, significantly reduces the generation of complex polymeric byproducts and homocoupling impurities. The use of cesium carbonate, a mild inorganic base, ensures that sensitive functional groups on the aromatic rings of the starting materials—such as esters, halogens, or phosphonates—remain intact throughout the reaction. This high functional group tolerance is evidenced by the successful synthesis of diverse derivatives, including those with electron-donating methoxy groups and electron-withdrawing fluoro or chloro substituents. For quality control laboratories, this translates to a cleaner crude reaction profile, allowing for simpler purification strategies and higher overall recovery rates of the target high-purity pharmaceutical intermediate.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it highly attractive for process chemistry teams looking to transfer technology from the lab to the pilot plant. The protocol involves a straightforward one-pot procedure where the solid base, liquid solvent, and liquid/solid reagents are combined in a standard reactor. The reaction progress is monitored to ensure complete consumption of the limiting reagent, typically achieved within 8 to 16 hours depending on the specific electronic nature of the substrates. Upon completion, the heterogeneous mixture is filtered to remove inorganic salts, and the filtrate is concentrated. The resulting crude material is then subjected to standard silica gel chromatography to isolate the pure triazole product. For a detailed, step-by-step breakdown of the standardized operating procedures, please refer to the technical guide below.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in anhydrous acetonitrile under inert atmosphere.
- Heat the reaction mixture to 60°C and stir for 12 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers compelling economic and logistical benefits that extend far beyond simple yield metrics. The primary value proposition lies in the drastic simplification of the raw material portfolio and the enhancement of workplace safety, which directly correlates to reduced operational overheads. By shifting away from specialized, hazardous azides to commodity chemicals like diazo compounds and imidoyl chlorides, manufacturers can leverage existing supply chains and negotiate better pricing due to the high availability of these feedstocks. Furthermore, the removal of transition metal catalysts eliminates the need for expensive scavenging resins or complex extraction protocols required to meet strict residual metal limits, thereby reducing both material costs and processing time.
- Cost Reduction in Manufacturing: The economic impact of this metal-free process is substantial. Traditional copper-catalyzed routes incur hidden costs associated with catalyst procurement, ligand optimization, and extensive purification to remove trace metals. In this novel approach, the use of inexpensive cesium carbonate as a promoter significantly lowers the direct material cost per kilogram of product. Additionally, the simplified workup procedure reduces solvent consumption and labor hours required for purification. The ability to operate at moderate temperatures (60°C) rather than extreme cryogenic or high-heat conditions also results in lower energy consumption for heating and cooling utilities, contributing to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the stability and availability of starting materials. Organic azides, often required for conventional triazole synthesis, are frequently subject to strict transportation regulations and limited supplier bases due to their instability. In contrast, the diazo compounds and trifluoroethylimidoyl chlorides utilized in this method are stable, shelf-stable, and widely produced by multiple chemical vendors globally. This diversification of the supply base mitigates the risk of single-source bottlenecks and ensures continuous production capability. Moreover, the robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or reagent quality, leading to more consistent batch-to-batch performance and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this methodology is inherently designed for scalability. The absence of explosive azides removes a major barrier to scaling up reaction volumes, allowing for safe production in large-scale reactors without the need for specialized blast-proof infrastructure. From an environmental standpoint, the process generates less hazardous waste. The elimination of heavy metals means that wastewater streams are easier to treat and dispose of in compliance with increasingly stringent environmental regulations. This 'green' profile not only reduces waste disposal costs but also enhances the corporate sustainability credentials of the manufacturing facility, a factor that is becoming increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial aspects of this synthesis method, we have compiled answers to common inquiries based on the patent data. These insights address the specific concerns of technical decision-makers regarding safety, scalability, and product quality. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into your existing production workflows.
Q: Why is this new synthesis method safer than traditional triazole production?
A: Traditional methods often rely on organic azides, which are toxic and potentially explosive. This patented route utilizes stable diazo compounds and imidoyl chlorides, eliminating the safety risks associated with handling high-energy azide intermediates.
Q: What represents the primary cost driver in this manufacturing process?
A: The process utilizes cesium carbonate as a promoter, which is relatively inexpensive compared to transition metal catalysts like copper or palladium. Additionally, the absence of heavy metals removes the need for costly purification steps to meet residual metal specifications.
Q: Can this reaction be scaled for industrial production?
A: Yes, the patent explicitly states that the method can be easily extended to the gram level and beyond. The use of common solvents like acetonitrile and mild temperatures (50-70°C) facilitates straightforward scale-up in standard reactor vessels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis method described in patent CN113121462B for the production of advanced heterocyclic intermediates. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale optimization to multi-ton manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 5-trifluoromethyl-1,2,3-triazole meets the highest standards of quality and consistency required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact us directly to request specific COA data for our reference standards and to discuss route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you secure not just a supplier, but a strategic ally committed to driving efficiency and innovation in your supply chain.
