Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial Pharmaceutical Applications
Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial Pharmaceutical Applications
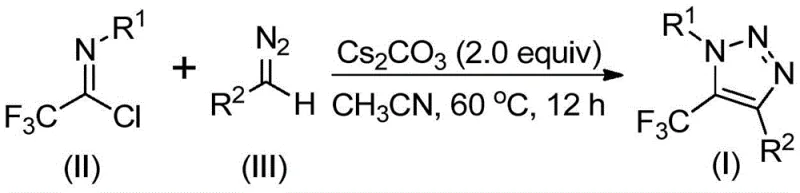
The rapid evolution of medicinal chemistry demands increasingly sophisticated heterocyclic scaffolds that offer enhanced metabolic stability and bioavailability. Among these, the 1,2,3-triazole ring system stands out as a privileged structure in drug discovery, frequently utilized as a bioisostere for amide bonds and other linkers due to its robustness and ability to engage in hydrogen bonding interactions. Specifically, the introduction of a trifluoromethyl group at the 5-position of the triazole ring significantly amplifies these pharmacological properties by increasing lipophilicity and electronegativity, which are critical parameters for optimizing drug-receptor binding affinity. Patent CN113121462B discloses a groundbreaking preparation method for these valuable 5-trifluoromethyl substituted 1,2,3-triazole compounds, addressing long-standing synthetic challenges associated with traditional routes. This technological breakthrough offers a streamlined, metal-free pathway that utilizes readily available diazo compounds and trifluoroethylimidoyl chlorides, marking a significant shift towards safer and more efficient industrial manufacturing processes for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted triazoles has relied heavily on copper-catalyzed azide-alkyne cycloaddition (CuAAC) reactions or organocatalytic 1,3-dipolar cycloadditions involving trifluoromethyl ketones. While effective in academic settings, these conventional methodologies present substantial hurdles for large-scale commercial production. The primary concern lies in the utilization of organic azides, which are notoriously toxic and possess high explosive potential, posing severe safety risks during scale-up operations. Furthermore, copper-catalyzed variants inevitably introduce transition metal contaminants into the reaction mixture. Removing these heavy metal residues to meet the stringent purity specifications required for active pharmaceutical ingredients (APIs) necessitates complex and costly downstream purification steps, such as scavenging resins or repeated recrystallizations. Additionally, the requirement for specialized trifluoromethylating reagents in some protocols can drive up raw material costs and complicate supply chain logistics, making these traditional routes less attractive for cost-sensitive manufacturing environments.
The Novel Approach
In stark contrast to the hazardous and metal-dependent legacy methods, the novel approach detailed in the patent utilizes a base-promoted cyclization strategy that completely circumvents the need for transition metal catalysts and explosive azide precursors. By employing trifluoroethylimidoyl chlorides and diazo compounds as the foundational building blocks, this method achieves the formation of the triazole core through a mechanistically distinct pathway that is inherently safer and more atom-economical. The reaction proceeds under mild thermal conditions, typically between 50°C and 70°C, utilizing cesium carbonate as a benign promoter in common organic solvents like acetonitrile. This shift not only mitigates safety hazards associated with azide handling but also eliminates the regulatory burden of heavy metal testing and removal. The versatility of this approach is further evidenced by its compatibility with a wide array of functional groups, allowing for the facile synthesis of diverse derivatives suitable for various therapeutic applications.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic pathway proposed for this transformation involves a sophisticated sequence of bond-forming events initiated by the deprotonation or activation of the diazo species by the carbonate base. It is hypothesized that the reaction commences with a base-promoted intermolecular nucleophilic addition of the diazo compound to the electrophilic carbon of the trifluoroethylimidoyl chloride. This initial step facilitates the formation of a critical carbon-carbon bond while eliminating a chloride leaving group, generating a reactive intermediate that is primed for cyclization. Subsequently, the system undergoes an intramolecular 5-endo-dig cyclization, a process that efficiently closes the five-membered triazole ring. This specific cyclization mode is particularly advantageous as it allows for the precise placement of the trifluoromethyl group at the 5-position, ensuring the correct regiochemistry required for biological activity. The absence of metal coordination complexes simplifies the electronic landscape of the reaction, reducing the likelihood of side reactions such as homocoupling or polymerization that often plague metal-catalyzed systems.
From an impurity control perspective, the mild nature of the base-promoted mechanism contributes significantly to the cleanliness of the crude reaction profile. Traditional copper-catalyzed reactions often suffer from the formation of oxidative byproducts or Glaser coupling products derived from alkyne impurities. In this novel protocol, the reliance on stable imidoyl chlorides and controlled diazo reactivity minimizes the generation of such complex impurity profiles. The use of cesium carbonate, a weak base compared to alkoxides or hydrides, ensures that sensitive functional groups on the aromatic rings—such as esters, ketones, or halides—remain intact throughout the synthesis. This high level of chemoselectivity is paramount for R&D directors aiming to synthesize complex analogs without requiring extensive protecting group strategies. Furthermore, the reaction's tolerance to moisture and oxygen, relative to organometallic alternatives, enhances its robustness in standard manufacturing facilities, thereby reducing the risk of batch-to-batch variability caused by environmental factors.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The operational simplicity of this synthesis makes it highly accessible for both laboratory-scale optimization and pilot-plant production. The protocol dictates a straightforward mixing procedure where cesium carbonate, the imidoyl chloride, and the diazo compound are combined in an aprotic solvent. The reaction is then heated to a moderate temperature range, typically around 60°C, and maintained for a duration of 8 to 16 hours to ensure complete conversion. Post-reaction processing is equally uncomplicated, involving basic filtration to remove inorganic salts followed by standard silica gel chromatography. This ease of execution significantly lowers the barrier to entry for process chemists looking to adopt this technology. For a detailed breakdown of the standardized operating procedures, stoichiometry, and specific workup instructions, please refer to the technical guide below.
- Combine cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature between 50°C and 70°C and maintain stirring for 8 to 16 hours.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this metal-free synthesis methodology represents a strategic opportunity to optimize both cost structures and supply reliability. The elimination of expensive transition metal catalysts, such as copper salts and specialized ligands, directly reduces the bill of materials for every kilogram of product manufactured. Moreover, the avoidance of hazardous azide reagents removes the need for specialized storage infrastructure and explosive-proof reactor setups, leading to substantial capital expenditure savings and lower insurance premiums. The reliance on commercially available starting materials, such as trifluoroethylimidoyl chlorides and diazo compounds, ensures a stable supply chain that is less susceptible to the geopolitical fluctuations often associated with rare earth metals or exotic reagents. This stability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the downstream purification train. By removing the necessity for heavy metal scavenging steps, manufacturers can significantly reduce the consumption of expensive purification resins and the associated waste disposal costs. The high atom economy of the reaction means that a larger proportion of the input mass is converted into the desired product, minimizing raw material waste. Additionally, the use of cesium carbonate as a promoter is cost-effective compared to proprietary organocatalysts, and the ability to use common solvents like acetonitrile further aligns with standard solvent recovery protocols, enhancing overall process economics without compromising yield or quality.
- Enhanced Supply Chain Reliability: The robustness of the starting materials contributes to a more resilient supply chain. Trifluoroethylimidoyl chlorides and diazo compounds are established commodities in the fine chemical sector, meaning they can be sourced from multiple qualified vendors, reducing the risk of single-source bottlenecks. The mild reaction conditions (50-70°C) allow the process to be run in standard glass-lined or stainless steel reactors without requiring specialized high-pressure or cryogenic equipment. This flexibility enables contract manufacturing organizations to easily integrate the process into existing production lines, ensuring that lead times for high-purity pharmaceutical intermediates remain short and predictable even during periods of high market demand.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) standpoint, this methodology offers a greener alternative to traditional triazole synthesis. The absence of toxic heavy metals simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site. The reaction generates minimal hazardous byproducts, primarily inorganic salts that are easy to handle and dispose of in compliance with international regulations. The scalability of the process has been demonstrated to be effective from gram-scale laboratory experiments to multi-kilogram pilot runs, indicating a smooth path to commercial tonnage production. This scalability ensures that the technology can grow alongside the clinical development of the drug candidate, providing a seamless transition from early-stage R&D to full-scale commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on reaction parameters and substrate compatibility. Understanding these nuances is essential for process development teams evaluating the feasibility of this route for their specific pipeline candidates.
Q: Why is this metal-free synthesis preferred over traditional copper-catalyzed methods?
A: Traditional copper-catalyzed methods often require toxic and explosive azide compounds and leave behind heavy metal residues that are difficult to remove in pharmaceutical grades. This novel base-promoted method eliminates the need for transition metals and hazardous azides, simplifying purification and enhancing safety.
Q: What are the optimal reaction conditions for high yield?
A: The patent data indicates that using cesium carbonate (2.0 equivalents) in acetonitrile at 60°C for 12 hours provides optimal conversion rates. The molar ratio of trifluoroethylimidoyl chloride to diazo compound is preferably maintained at 1:1.5.
Q: Does this method support diverse functional groups on the substrate?
A: Yes, the method demonstrates broad substrate tolerance. It successfully accommodates various substituents on the aryl rings, including methyl, methoxy, halogens (chlorine, fluorine), and different acyl groups, making it versatile for generating diverse chemical libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity 5-trifluoromethyl-1,2,3-triazole derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in metal-free synthesis allows us to offer products with superior impurity profiles, reducing the burden on your downstream formulation teams and accelerating your time to market.
We invite you to collaborate with us to leverage this innovative synthetic technology for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis needs. Together, we can drive efficiency and innovation in the production of complex pharmaceutical intermediates.
