Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Pharmaceutical Manufacturing
Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Pharmaceutical Manufacturing
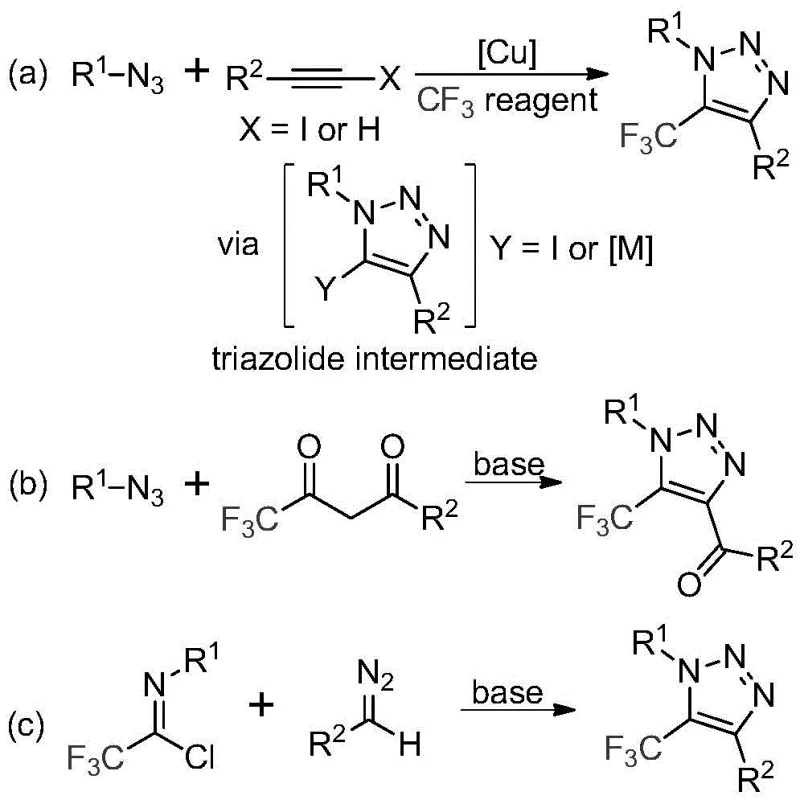
The rapid evolution of medicinal chemistry demands increasingly sophisticated heterocyclic scaffolds that offer enhanced metabolic stability and bioavailability, with the 1,2,3-triazole ring standing out as a privileged structure in modern drug design. Patent CN113121462B introduces a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds, addressing critical limitations in current synthetic methodologies by utilizing a metal-free, base-promoted cyclization strategy. This innovation is particularly significant for the development of high-purity pharmaceutical intermediates, as it circumvents the stringent regulatory hurdles associated with residual heavy metals in active pharmaceutical ingredients (APIs). By leveraging readily available diazo compounds and trifluoroethylimidoyl chlorides, this technology offers a robust pathway for constructing complex nitrogen-containing heterocycles that are essential for next-generation therapeutics and agrochemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on copper-catalyzed azide-alkyne cycloaddition (CuAAC) reactions or organocatalytic 1,3-dipolar cycloadditions involving toxic azides. These traditional pathways present substantial safety and purification challenges; specifically, the handling of organic azides poses significant explosion risks, while the use of copper catalysts necessitates expensive and time-consuming purification steps to meet strict ppm-level limits for residual metals in final drug products. Furthermore, the requirement for specialized trifluoromethylating reagents in some conventional routes often drives up raw material costs and complicates supply chain logistics for large-scale manufacturing operations. The reliance on transition metals also introduces potential variability in reaction outcomes due to catalyst deactivation or ligand sensitivity, which can hinder consistent commercial production.
The Novel Approach
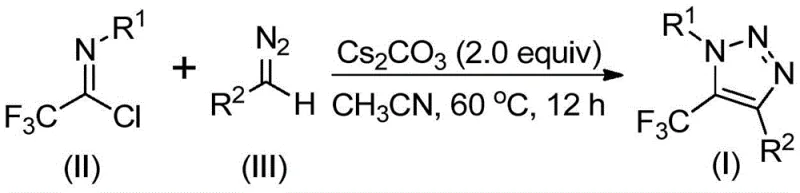
In stark contrast to these legacy methods, the technology disclosed in CN113121462B employs a simple yet highly efficient base-promoted reaction between trifluoroethylimidoyl chloride and diazo compounds, completely eliminating the need for transition metal catalysts and hazardous azide reagents. This novel approach utilizes inexpensive and commercially accessible starting materials, such as cesium carbonate as a promoter, to drive the cyclization under mild thermal conditions typically around 60°C. The absence of metal catalysts not only simplifies the downstream purification process—often requiring only filtration and standard column chromatography—but also inherently ensures a cleaner impurity profile suitable for sensitive biological applications. This method represents a paradigm shift towards greener, safer, and more cost-effective synthesis of valuable fluorinated heterocycles.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic pathway of this transformation involves a sophisticated sequence of base-promoted intermolecular nucleophilic addition and elimination processes that facilitate carbon-carbon bond formation without external metal activation. Initially, the base activates the diazo compound or the imidoyl chloride to generate a reactive nucleophilic species, which attacks the electrophilic center of the partner molecule to form a linear intermediate. This is followed by a crucial intramolecular 5-endo-dig cyclization step that closes the triazole ring, driven by the thermodynamic stability of the aromatic heterocyclic system and the release of nitrogen gas or chloride ions depending on the specific mechanistic branch. The use of aprotic solvents like acetonitrile is critical in this mechanism, as they effectively solvate the ionic intermediates and promote the reaction kinetics without interfering with the sensitive diazo functionality.
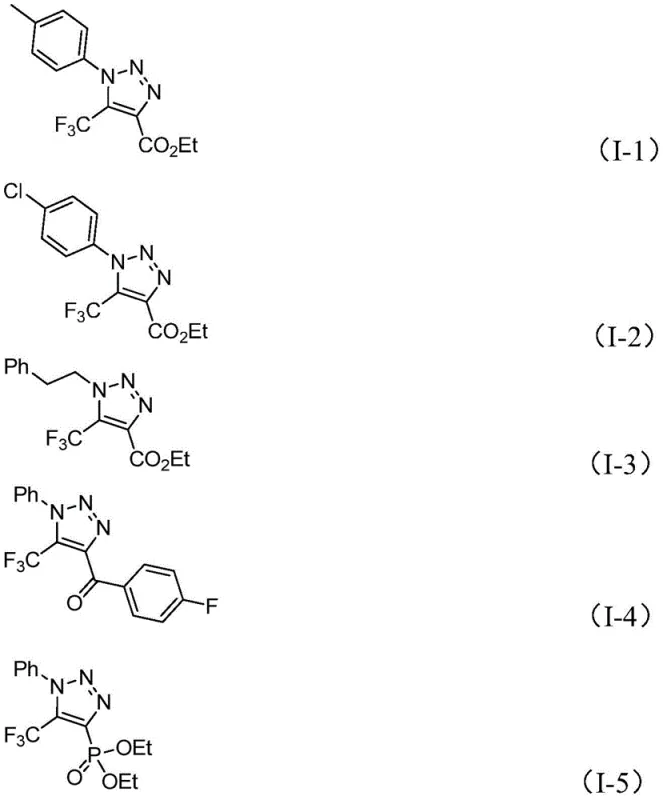
From an impurity control perspective, this metal-free mechanism offers distinct advantages by avoiding the formation of metal-complexed side products that are notoriously difficult to separate from the final API. The reaction exhibits excellent functional group tolerance, accommodating a wide range of substituents on both the N1 and C4 positions of the triazole ring, including esters, ketones, and phosphonates, without significant degradation or side reactions. The structural integrity of the trifluoromethyl group is maintained throughout the process, ensuring the desired lipophilicity and metabolic stability characteristics are preserved in the final molecule. This robustness allows for the synthesis of diverse libraries of triazole derivatives, enabling rapid structure-activity relationship (SAR) studies during the drug discovery phase.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
The operational simplicity of this synthesis protocol makes it highly attractive for both laboratory-scale optimization and pilot-plant production, requiring standard glassware and common laboratory equipment. The process typically involves charging a reaction vessel with cesium carbonate, the specific trifluoroethylimidoyl chloride derivative, and the corresponding diazo compound in an anhydrous organic solvent such as acetonitrile. Detailed standardized synthesis steps, including precise molar ratios, temperature ramping profiles, and specific workup procedures for maximizing yield and purity, are outlined in the technical guide below for qualified partners.
- Combine cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an aprotic solvent like acetonitrile.
- Heat the reaction mixture to 60°C and stir for 12 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route translates into tangible strategic benefits regarding cost stability and supply continuity. By removing the dependency on volatile copper prices and specialized ligand suppliers, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing through the use of commodity-grade bases and widely available organic building blocks. The simplified purification workflow reduces solvent consumption and waste generation, aligning with increasingly stringent environmental regulations and lowering the overall cost of goods sold (COGS) for high-volume production campaigns.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated scavenging resins required for their removal drastically lowers the direct material costs per kilogram of product. Furthermore, the use of cheap promoters like cesium carbonate instead of precious metal complexes ensures that raw material price fluctuations have a minimal impact on the final budget, providing greater financial predictability for long-term contracts. The high atom economy of the reaction also means less waste disposal cost, contributing to a leaner and more efficient production model.
- Enhanced Supply Chain Reliability: Since the key starting materials, such as diazo compounds and imidoyl chlorides, are derived from common industrial feedstocks, the risk of supply disruption due to niche reagent shortages is significantly mitigated. This robustness allows for the establishment of multi-source supply chains, ensuring that production schedules remain uninterrupted even during global logistical challenges. The stability of the reagents also simplifies storage and transportation requirements, reducing the need for specialized cold-chain logistics or hazardous material handling protocols.
- Scalability and Environmental Compliance: The mild reaction conditions (50-70°C) and the absence of pyrophoric or explosive azides make this process inherently safer and easier to scale from gram to multi-ton quantities without extensive re-engineering. The reduced toxicity profile of the reagents and byproducts facilitates easier compliance with environmental health and safety (EHS) standards, accelerating regulatory approvals for new manufacturing sites. This scalability ensures that the technology can seamlessly transition from early-stage clinical supply to commercial launch volumes without bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, based on the detailed experimental data and scope provided in the patent literature. Understanding these nuances is critical for R&D teams evaluating the feasibility of integrating this route into their existing process development pipelines.
Q: Does this synthesis method involve toxic azides or heavy metals?
A: No, unlike traditional CuAAC methods, this patented process utilizes stable diazo compounds and imidoyl chlorides, completely eliminating the need for explosive organic azides and toxic copper catalysts.
Q: What is the typical yield and scalability of this reaction?
A: The patent reports yields ranging from 40% to 91% across various substrates. The mild conditions (60°C) and simple workup make it highly suitable for scale-up from gram to kilogram levels.
Q: What types of substituents are tolerated on the triazole ring?
A: The method demonstrates broad functional group tolerance, accommodating esters, ketones, phosphonates, and various aryl groups with electron-donating or withdrawing substituents at the N1 and C4 positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
As a leading CDMO partner, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to market-ready supply is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole intermediate delivered meets the highest industry standards for potency and impurity control. We understand the critical nature of timeline adherence in drug development and are committed to providing a reliable supply chain that supports your clinical and commercial milestones.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project requirements and volume needs. By engaging with us early in your development cycle, you can leverage our expertise to optimize your synthetic route, secure specific COA data for your regulatory filings, and obtain comprehensive route feasibility assessments that de-risk your manufacturing strategy. Let us be your strategic partner in delivering high-quality fluorinated heterocycles for your next breakthrough therapy.
