Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Advanced Drug Discovery
Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Advanced Drug Discovery
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, sustainable, and cost-effective synthetic routes. A significant breakthrough in this domain is detailed in Chinese Patent CN113307790B, which discloses a novel preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. These heterocyclic structures are of paramount importance in medicinal chemistry, serving as critical scaffolds in numerous bioactive molecular frameworks and functioning as versatile ligands in coordination chemistry for applications ranging from drug discovery to organic light-emitting diodes (OLEDs). The innovation lies in its ability to construct these complex architectures through a direct oxidative cyclization strategy that bypasses the limitations of traditional multi-step syntheses. By leveraging a metal-free catalytic system, this technology offers a robust pathway for producing high-purity intermediates that are essential for the development of next-generation therapeutics and functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been fraught with significant operational challenges that hinder large-scale commercial adoption. Traditional methodologies often rely on quinoline-2-carboxylic acid as the primary starting material, necessitating a cumbersome five-step reaction sequence to achieve the final target molecule. This lengthy synthetic route not only results in a dismal overall yield of approximately 17% but also imposes severe reaction conditions that demand rigorous control over temperature and atmosphere. Furthermore, the reliance on multiple purification steps between each transformation increases the consumption of solvents and silica gel, driving up both the environmental footprint and the production costs. For procurement managers and supply chain directors, these inefficiencies translate into higher raw material costs, extended lead times, and greater complexity in waste management, making the conventional approach economically unviable for mass production.
The Novel Approach
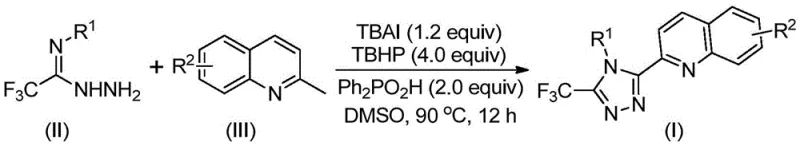
In stark contrast, the methodology outlined in patent CN113307790B introduces a streamlined, one-pot oxidative cyclization protocol that fundamentally reshapes the production economics of these valuable intermediates. This innovative route utilizes readily available and inexpensive starting materials, specifically 2-methylquinoline and trifluoroacetimidoyl hydrazide, which are coupled directly in the presence of tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP). The reaction proceeds efficiently in dimethyl sulfoxide (DMSO) at moderate temperatures between 80°C and 100°C, eliminating the need for stringent anhydrous or oxygen-free environments. This simplification of reaction conditions not only enhances operational safety but also drastically reduces the energy consumption associated with cryogenic cooling or inert gas purging. The result is a highly convergent synthesis that delivers the target 3-quinolyl-5-trifluoromethyl-1,2,4-triazole compounds in substantially improved yields, often exceeding 90% for optimized substrates.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The success of this synthetic strategy hinges on a sophisticated yet elegant mechanistic pathway driven by the synergistic interaction between the iodide catalyst and the peroxide oxidant. The process initiates with the TBAI-promoted oxidation of the methyl group on the 2-methylquinoline substrate, effectively converting it into a reactive 2-quinolinecarbaldehyde equivalent in situ. This transient aldehyde species then undergoes a condensation reaction with the trifluoroacetimidoyl hydrazide to form a dehydrated hydrazone intermediate. Subsequent oxidative iodination activates the hydrazone nitrogen, facilitating an intramolecular electrophilic substitution that closes the triazole ring. The final aromatization step releases the iodine species back into the catalytic cycle, ensuring high atom economy. This radical-mediated or cationic pathway avoids the formation of stable byproducts that typically plague transition-metal catalyzed couplings, thereby simplifying the impurity profile of the crude reaction mixture.
From a quality control perspective, the absence of heavy metal residues is a critical advantage for pharmaceutical applications where strict limits on elemental impurities are enforced by regulatory bodies such as the FDA and EMA. The use of diphenylphosphinic acid as an additive further stabilizes the reaction intermediates and promotes the cyclization efficiency, ensuring consistent batch-to-b reproducibility. The tolerance of this mechanism towards various electronic and steric environments on both the quinoline and the hydrazide aromatic rings allows for the synthesis of a diverse library of derivatives. Whether the substrate bears electron-donating groups like methoxy or electron-withdrawing groups like nitro or trifluoromethyl, the catalytic system maintains high reactivity, providing R&D teams with the flexibility to explore structure-activity relationships without being constrained by synthetic feasibility.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise adherence to the optimized stoichiometry and reaction parameters established in the patent data. The standard protocol involves charging a reactor with the specified molar ratios of 2-methylquinoline, trifluoroacetimidoyl hydrazide, TBAI, TBHP, and diphenylphosphinic acid in DMSO. The mixture is then heated to the target temperature and monitored until conversion is complete, typically within 8 to 14 hours. Following the reaction, the workup procedure is remarkably straightforward, involving filtration and silica gel treatment followed by standard column chromatography. For detailed operational parameters, stoichiometry tables, and specific purification protocols tailored to different substituents, please refer to the standardized synthesis guide below.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl hydroperoxide (TBHP), diphenylphosphinic acid, trifluoroacetimidoyl hydrazide, and 2-methylquinoline in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature range of 80°C to 100°C and maintain stirring for 8 to 14 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply chain resilience, this metal-free methodology presents a compelling value proposition that addresses several critical pain points in fine chemical manufacturing. The shift from a five-step linear synthesis to a direct oxidative coupling significantly compresses the production timeline, reducing the manpower and equipment hours required per kilogram of output. Moreover, the elimination of precious metal catalysts such as palladium or copper removes the necessity for expensive scavenging resins and complex metal removal validation studies, which are often bottlenecks in API manufacturing. This simplification translates directly into substantial cost savings and a more agile response to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the use of commodity chemicals rather than specialized reagents. Tetrabutylammonium iodide and tert-butyl hydroperoxide are bulk industrial chemicals with stable pricing and abundant global supply, insulating the production cost from the volatility often seen with rare earth or precious metal catalysts. Additionally, the high atom economy and reduced step count minimize solvent usage and waste generation, lowering the costs associated with solvent recovery and hazardous waste disposal. By avoiding the low-yield intermediate steps of the traditional route, the overall material throughput is maximized, ensuring that a higher percentage of input raw materials are converted into saleable product.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on widely available starting materials like 2-methylquinoline and aromatic amines, which are produced by multiple vendors worldwide. This diversification of the supply base mitigates the risk of single-source dependency that can plague specialized catalyst markets. Furthermore, the robustness of the reaction conditions means that the process is less susceptible to minor variations in utility quality or environmental factors, ensuring consistent production schedules. The ability to operate without strict inert atmospheres simplifies the engineering requirements for the reactor setup, allowing for faster turnaround times between batches and increased facility utilization rates.
- Scalability and Environmental Compliance: The transition from bench scale to commercial production is facilitated by the thermal stability and safety profile of the reagents involved. Unlike some energetic oxidants that require careful dosing at low temperatures, the TBHP/TBAI system operates safely at elevated temperatures in polar aprotic solvents, making it amenable to standard stainless steel reactor infrastructure. The absence of heavy metals simplifies the environmental compliance landscape, reducing the burden of wastewater treatment and meeting the increasingly stringent green chemistry standards demanded by global regulators. This eco-friendly profile not only reduces regulatory risk but also aligns with the sustainability goals of modern pharmaceutical and chemical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative cyclization technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: Does this synthesis method require toxic heavy metal catalysts?
A: No, the method described in patent CN113307790B utilizes an organocatalytic system involving tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), completely eliminating the need for expensive and toxic transition metal catalysts.
Q: What is the substrate scope for the quinoline component?
A: The process demonstrates excellent tolerance for various substituents on the quinoline ring, including hydrogen, methyl, methoxy, halogens (Cl, Br), and nitro groups at different positions, allowing for diverse structural modifications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction operates under relatively mild conditions (80-100°C) without strict anhydrous or oxygen-free requirements, uses commercially available raw materials, and has been demonstrated to be easily expandable from gram-scale to larger batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
As the demand for complex heterocyclic intermediates continues to grow in the pharmaceutical and agrochemical sectors, having a manufacturing partner with deep technical expertise is essential. NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of oxidative cyclization chemistries, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand that the transition from patent literature to commercial reality requires more than just mixing chemicals; it demands a comprehensive understanding of process safety, impurity control, and regulatory compliance.
We invite R&D directors and procurement specialists to collaborate with us to evaluate the feasibility of this metal-free route for your specific project needs. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this novel methodology. Please contact our technical procurement team today to request specific COA data for related triazole derivatives and to discuss route feasibility assessments tailored to your supply chain objectives. Together, we can accelerate the delivery of high-quality intermediates to the market while optimizing your overall production costs.
