Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which enhance metabolic stability and lipophilicity. Patent CN111423381A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing critical synthetic challenges in modern drug discovery. This technology leverages a transition metal palladium-catalyzed carbonylation cascade reaction, utilizing readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts. Unlike conventional approaches that often require hazardous reagents or extreme conditions, this novel pathway operates under remarkably mild parameters, specifically at 30°C, ensuring high reaction efficiency and exceptional substrate compatibility. For R&D directors and procurement specialists, this represents a significant opportunity to streamline the synthesis of valuable intermediates used in bioactive molecules, potentially reducing lead times for high-purity pharmaceutical intermediates while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles functionalized with trifluoromethyl groups has relied heavily on synthons like trifluorodiazoethane. While effective in certain contexts, these traditional reagents pose significant safety hazards due to their explosive nature and instability, complicating storage and handling in large-scale manufacturing environments. Furthermore, many existing protocols necessitate the use of high-pressure carbon monoxide gas or specialized equipment to introduce the carbonyl functionality, which drastically increases capital expenditure and operational complexity. The reliance on such苛刻 conditions often limits the scope of compatible functional groups, leading to lower yields when complex substrates are employed. Consequently, the industry has faced persistent challenges in achieving cost reduction in API manufacturing for trifluoromethylated scaffolds, as the safety measures and specialized infrastructure required drive up production costs and extend supply chain lead times.
The Novel Approach
The methodology disclosed in CN111423381A offers a transformative solution by employing a palladium-catalyzed carbonylation series reaction that circumvents the need for gaseous carbon monoxide. Instead, it utilizes a mixture of formic acid and acetic anhydride as a safe and efficient carbon monoxide surrogate. This approach allows the reaction to proceed smoothly at a mild temperature of 30°C over a period of 16 to 24 hours. The strategic use of trifluoroethylimidoyl chloride and diaryliodonium salts as key building blocks not only enhances safety but also broadens the chemical space accessible to chemists. As illustrated in the general reaction scheme below, this multi-component coupling efficiently constructs the imidazole core with high atom economy.
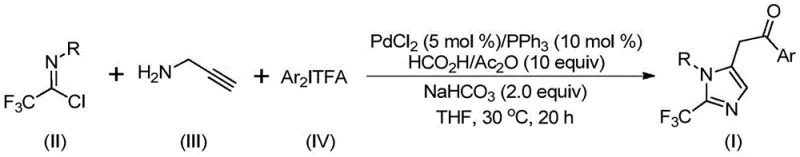
By replacing hazardous diazo compounds with stable imidoyl chlorides, this novel approach significantly simplifies the operational workflow, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming to diversify their portfolio with safer, more sustainable chemistry.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
A deep understanding of the catalytic cycle is essential for R&D teams looking to optimize this process for specific analogues. The reaction mechanism likely initiates with a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine, generating a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization to facilitate the next critical step. The palladium catalyst, generated in situ from palladium chloride and triphenylphosphine, then engages in an aminopalladation of the alkyne moiety. This forms an alkenyl palladium intermediate, which further isomerizes into a more stable alkyl palladium species. The presence of the formic acid and acetic anhydride mixture is crucial here, as it releases carbon monoxide in situ, which inserts into the palladium-carbon bond to form an acyl palladium intermediate.
Following carbonylation, the diaryliodonium salt plays a pivotal role by undergoing oxidative addition with the acyl palladium species, generating a high-valent tetravalent palladium intermediate. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted imidazole compound and regenerates the active palladium catalyst. This intricate cascade ensures high selectivity and minimizes the formation of undesired by-products. The robustness of this mechanism is evidenced by its tolerance to a wide range of electronic environments on the aromatic rings. Whether the substrate contains electron-donating groups like methoxy or tert-butyl, or electron-withdrawing groups such as nitro or halogens, the catalytic system maintains high efficiency, as demonstrated by the diverse array of successful examples shown below.
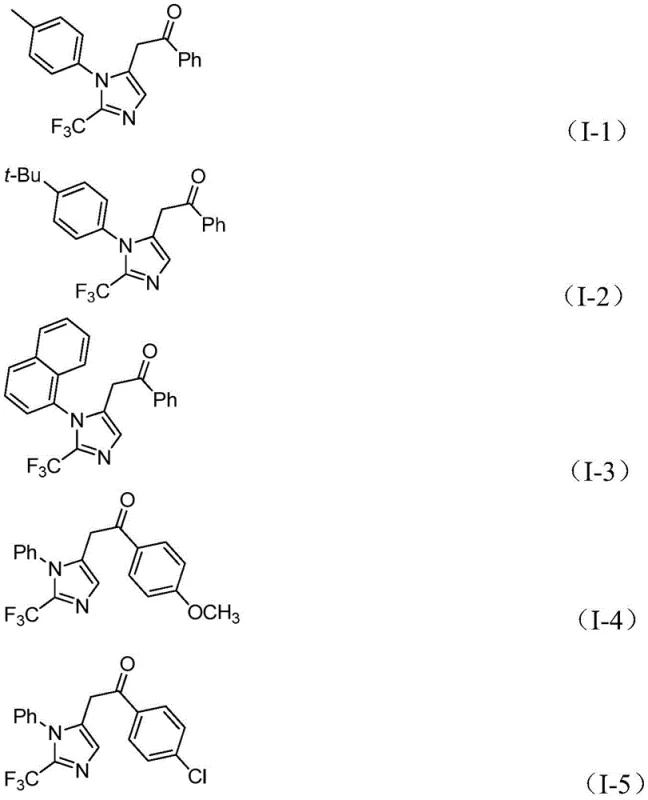
This mechanistic resilience is vital for impurity control, as it prevents the accumulation of partially reacted intermediates that could complicate downstream purification, thereby ensuring the delivery of high-purity OLED material or pharmaceutical precursors with consistent quality profiles.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and purity. The protocol involves combining the palladium catalyst, ligand, base, and CO surrogate in an aprotic organic solvent such as tetrahydrofuran (THF), which has been identified as the optimal medium for dissolving all reactants and promoting conversion. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the guide below to assist technical teams in replicating these results effectively.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, and a formic acid/acetic anhydride mixture in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt to the reaction vessel under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, then filter and purify via column chromatography to isolate the target imidazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this patented technology are profound. By shifting away from hazardous and expensive reagents towards commodity chemicals, manufacturers can achieve substantial cost savings without compromising on quality. The mild reaction conditions eliminate the need for specialized high-pressure reactors, reducing both capital investment and energy consumption during production. Furthermore, the simplicity of the post-processing workflow, which involves basic filtration and standard chromatography, streamlines the manufacturing timeline, allowing for faster turnaround times and improved responsiveness to market demands.
- Cost Reduction in Manufacturing: The utilization of inexpensive starting materials such as trifluoroethylimidoyl chloride and propargylamine, coupled with a low-loading palladium catalyst system, drastically lowers the raw material cost per kilogram of product. Since the reaction does not require external carbon monoxide cylinders or cryogenic conditions, operational expenditures related to safety compliance and utility usage are significantly minimized. This economic efficiency translates directly into more competitive pricing for downstream clients seeking cost reduction in electronic chemical manufacturing or pharmaceutical production.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are commercially available and widely produced, mitigating the risk of supply disruptions often associated with specialized or custom-synthesized reagents. The robustness of the reaction across various substrates means that a single manufacturing platform can produce a diverse library of intermediates, enhancing flexibility. This reliability ensures consistent availability of critical building blocks, reducing lead time for high-purity pharmaceutical intermediates and supporting continuous manufacturing schedules.
- Scalability and Environmental Compliance: The process has been validated at the gram level and is designed for easy scale-up to industrial quantities, facilitating the commercial scale-up of complex polymer additives or drug candidates. Additionally, the avoidance of toxic diazo compounds and high-pressure gases aligns with increasingly stringent environmental regulations. The simplified waste stream, primarily consisting of standard organic solvents and salts, eases the burden on waste treatment facilities, promoting a greener and more sustainable production lifecycle.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the patent specifications and experimental data. These insights are intended to clarify the feasibility and advantages of adopting this technology for your specific production needs.
Q: What are the advantages of this palladium-catalyzed method over traditional trifluoromethylation?
A: Unlike traditional methods that often rely on unstable or hazardous trifluorodiazoethane, this patented process utilizes stable and commercially available trifluoroethylimidoyl chlorides. It operates under mild conditions (30°C) without the need for high-pressure carbon monoxide gas, significantly enhancing operational safety and scalability for industrial production.
Q: What is the substrate compatibility of this synthesis route?
A: The method demonstrates excellent functional group tolerance. It successfully accommodates various substituents on both the aryl ring of the imidoyl chloride and the diaryliodonium salt, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens, nitro, and trifluoromethyl groups, yielding products with high efficiency.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly states that the method is scalable to the gram level and potentially beyond. The use of inexpensive catalysts like palladium chloride, combined with simple post-processing steps involving filtration and standard column chromatography, makes it highly viable for cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed carbonylation technology in advancing the synthesis of complex heterocyclic scaffolds. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl imidazole delivered meets the highest international standards for pharmaceutical and fine chemical applications.
We invite you to collaborate with us to leverage this innovative chemistry for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us help you optimize your supply chain with reliable, high-quality intermediates.
