Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
The landscape of modern pharmaceutical synthesis is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign methodologies to access complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN111423381B, which discloses a novel preparation method for 2-trifluoromethyl substituted imidazole compounds. These nitrogen-containing five-membered heterocycles are pivotal structural motifs found in numerous bioactive molecules, including drugs and functional materials, where the introduction of a trifluoromethyl group significantly enhances electronegativity, metabolic stability, and lipophilicity. The patent outlines a transition metal palladium-catalyzed carbonylation series reaction that efficiently constructs these valuable cores from cheap and easily obtained starting materials. This technological advancement represents a paradigm shift for a reliable pharmaceutical intermediate supplier, offering a robust pathway to high-value chemical building blocks that were previously difficult or expensive to manufacture.
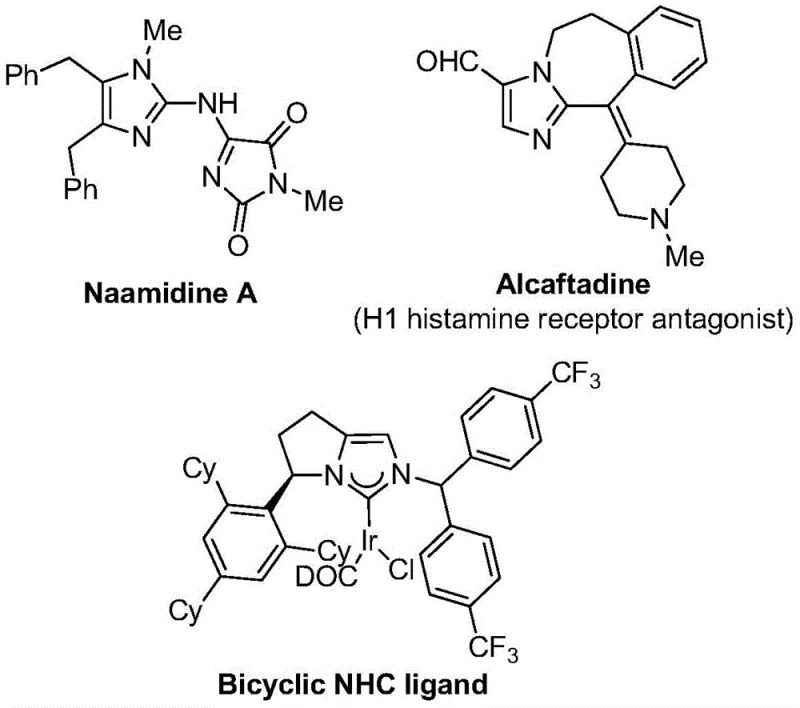
The strategic importance of this synthesis cannot be overstated when considering the broader context of drug discovery and development. Imidazole derivatives serve as critical intermediates in the production of active pharmaceutical ingredients (APIs) targeting various therapeutic areas, from antihistamines to antifungal agents. The ability to introduce a trifluoromethyl group regioselectively at the 2-position of the imidazole ring opens up new avenues for medicinal chemists to optimize lead compounds. As illustrated in the provided structural examples, molecules like Naamidine A and Alcaftadine showcase the diverse biological potential inherent in imidazole-based architectures. By leveraging the methodology described in CN111423381B, manufacturers can now access these sophisticated structures with greater ease, thereby accelerating the timeline from benchtop research to clinical trials and ultimately to market availability for life-saving medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has been fraught with significant challenges that hinder large-scale adoption. Traditional methods often rely on the direct reaction of synthons containing trifluoromethyl groups with suitable substrates, utilizing reagents such as trifluorodiazoethane or trifluoroethylimidoyl halides. While these reagents can effectively transfer the CF3 group, they are frequently associated with severe safety hazards, high costs, and limited commercial availability. Furthermore, many conventional protocols require harsh reaction conditions, including elevated temperatures, strong bases, or toxic solvents, which complicate the purification process and generate substantial chemical waste. The narrow substrate scope of older methods also restricts the structural diversity of the final products, limiting the ability of chemists to explore structure-activity relationships (SAR) effectively. These limitations create bottlenecks in the supply chain for high-purity pharmaceutical intermediates, driving up costs and extending lead times for downstream drug manufacturers.
The Novel Approach
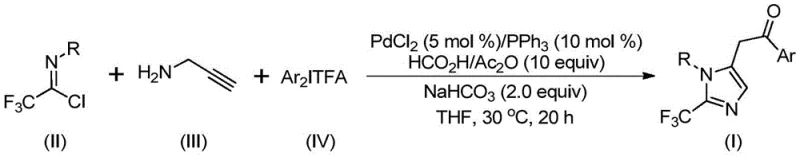
In stark contrast to these legacy techniques, the novel approach disclosed in the patent utilizes a sophisticated yet operationally simple palladium-catalyzed cascade reaction. This method employs trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts as the primary building blocks, reacting them in the presence of a palladium catalyst and a carbon monoxide surrogate. The reaction proceeds under remarkably mild conditions, typically at 30°C for 16 to 24 hours, which drastically reduces energy consumption and minimizes the risk of thermal degradation of sensitive functional groups. The use of formic acid and acetic anhydride as an in situ source of carbon monoxide eliminates the need for handling hazardous CO gas cylinders, enhancing workplace safety. This innovative strategy not only simplifies the operational workflow but also exhibits exceptional substrate compatibility, allowing for the incorporation of various electron-donating and electron-withdrawing groups. As shown in the general reaction scheme below, this one-pot transformation efficiently constructs the 2-trifluoromethyl imidazole core with high atom economy.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize the process for specific applications. The reaction initiates with the formation of a trifluoroacetamidine compound through an intermolecular carbon-nitrogen bond promoted by the base. This intermediate subsequently undergoes isomerization to align the reactive centers for cyclization. The palladium catalyst then engages in a complex cycle involving the activation of the alkyne moiety in the propargylamine derivative. Specifically, the palladium species coordinates to the alkyne, facilitating a palladation event that generates an alkenyl palladium intermediate. This species is pivotal as it sets the stage for the subsequent migratory insertion steps that build the heterocyclic ring system. The precise control over these elementary steps ensures that the reaction proceeds with high regioselectivity, favoring the formation of the desired 2-trifluoromethyl substituted product over potential isomers.
Following the initial palladation, the mechanism involves a series of isomerizations and insertions that are critical for the construction of the imidazole ring. The alkenyl palladium intermediate isomerizes to an alkyl palladium species, which then undergoes a carbonylation reaction. This step is facilitated by the carbon monoxide released from the decomposition of the formic acid/acetic anhydride mixture, leading to the formation of an acyl palladium intermediate. The oxidative addition of the diaryl iodonium salt to this acyl palladium species generates a high-valent tetravalent palladium intermediate, a key feature of this catalytic cycle. Finally, reductive elimination occurs to release the final 2-trifluoromethyl-substituted imidazole compound and regenerate the active palladium catalyst. This intricate dance of organometallic steps highlights the sophistication of the method, allowing for the assembly of complex molecular architectures from simple precursors with minimal byproduct formation.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction parameters to ensure optimal yields and purity. The protocol is designed to be user-friendly, utilizing standard Schlenk techniques and commercially available reagents. The molar ratios of the components are critical; typically, a slight excess of the trifluoroethylimidoyl chloride and diaryl iodonium salt relative to the propargylamine is employed to drive the reaction to completion. The choice of solvent is also paramount, with tetrahydrofuran (THF) being identified as the preferred medium due to its ability to dissolve all reactants effectively while promoting the catalytic cycle. Post-reaction processing is straightforward, involving filtration to remove inorganic salts followed by standard chromatographic purification. For those seeking a detailed procedural guide, the standardized synthesis steps are outlined below.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, formic acid, trifluoroethylimidoyl chloride, propargylamine, and diaryl iodide in an organic solvent such as THF.
- Stir the reaction mixture at a mild temperature of 30°C for a duration of 16 to 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and perform column chromatography purification to isolate the high-purity 2-trifluoromethyl substituted imidazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers compelling advantages that directly impact the bottom line and operational resilience. The shift towards using cheap and easily obtainable starting materials, such as aromatic amines and propargylamine, significantly reduces the raw material costs associated with producing these high-value intermediates. Unlike specialized fluorinating agents that may be subject to supply volatility or strict regulatory controls, the precursors used in this method are commodity chemicals with robust global supply chains. This accessibility ensures a stable supply of inputs, mitigating the risk of production delays caused by raw material shortages. Furthermore, the mild reaction conditions translate to lower energy costs and reduced wear and tear on reactor equipment, contributing to overall cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the elimination of expensive and hazardous reagents. By replacing costly trifluoromethyl sources with affordable trifluoroethylimidoyl chlorides, manufacturers can achieve substantial cost savings on a per-kilogram basis. Additionally, the use of palladium chloride, a relatively inexpensive palladium source compared to other complexes, combined with simple ligands like triphenylphosphine, keeps catalyst costs low. The high reaction efficiency and selectivity minimize the formation of difficult-to-remove impurities, thereby reducing the burden on downstream purification processes which are often the most expensive part of chemical manufacturing. These factors collectively enhance the profit margins for producers of high-purity OLED materials or pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is a top priority for multinational corporations, and this synthesis route bolsters reliability through its reliance on widely available feedstocks. The ability to source key ingredients from multiple vendors reduces dependency on single suppliers, a critical risk mitigation strategy in the current geopolitical climate. Moreover, the scalability of the reaction, which has been demonstrated to work effectively from gram to larger scales, ensures that production can be ramped up quickly to meet surging demand without requiring extensive process re-engineering. This flexibility allows supply chain managers to respond agilely to market fluctuations, ensuring that critical drug substances remain available to patients without interruption.
- Scalability and Environmental Compliance: Environmental sustainability is increasingly becoming a license to operate in the chemical industry, and this method aligns well with green chemistry principles. The avoidance of toxic gases like carbon monoxide and the use of milder temperatures reduce the environmental footprint of the manufacturing process. The simplified workup procedure, which avoids complex extraction sequences, leads to less solvent waste and lower disposal costs. As regulatory bodies tighten restrictions on chemical emissions and waste generation, adopting cleaner technologies like this palladium-catalyzed carbonylation provides a competitive edge. It positions manufacturers as responsible stewards of the environment while simultaneously improving operational efficiency and compliance with international standards.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled a set of frequently asked questions based on the patent specifications. These inquiries address common concerns regarding reaction scope, scalability, and product quality, providing decision-makers with the information needed to evaluate the feasibility of integrating this method into their existing portfolios. The answers are derived directly from the experimental data and theoretical framework presented in the patent documentation, ensuring accuracy and relevance for technical audiences.
Q: What are the primary advantages of this palladium-catalyzed method over traditional synthesis routes?
A: This method utilizes cheap and readily available starting materials like trifluoroethylimidoyl chloride and operates under extremely mild conditions (30°C), avoiding the harsh conditions and expensive reagents often required in conventional trifluoromethylation strategies.
Q: Does this synthetic route support a wide range of substrate functional groups?
A: Yes, the process demonstrates excellent substrate compatibility, tolerating various substituents such as methyl, tert-butyl, halogens, nitro, and methoxy groups on both the imidoyl chloride and the diaryl iodonium salt components.
Q: Is this methodology suitable for industrial-scale production of pharmaceutical intermediates?
A: Absolutely. The protocol is designed for scalability, utilizing common organic solvents like THF and standard post-processing techniques like column chromatography, making it highly viable for commercial manufacturing of complex heterocycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN111423381B for the production of next-generation pharmaceutical intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Whether you require custom synthesis of complex heterocycles or large-scale supply of key building blocks, our infrastructure is designed to support your most demanding projects with precision and reliability.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific applications. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your unique production needs, helping you identify opportunities for efficiency gains and expense reduction. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you navigate the complexities of modern chemical manufacturing and secure a competitive advantage in the global marketplace through innovation and operational excellence.
