Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Patent CN112239456B introduces a significant advancement in the preparation of substituted 2,3-dihydroquinolone compounds, a privileged scaffold found in numerous therapeutic agents. As illustrated in the biological context, these structures serve as the core framework for potent antitumor agents and analgesics, highlighting their critical value in drug discovery pipelines. The disclosed method leverages a transition metal palladium-catalyzed carbonylation strategy, utilizing N-pyridine sulfonyl-o-iodoaniline and olefins as key starting materials. This approach addresses long-standing challenges in heterocyclic synthesis by offering a pathway that is not only operationally simple but also compatible with a broad spectrum of functional groups, thereby expanding the chemical space accessible to medicinal chemists.
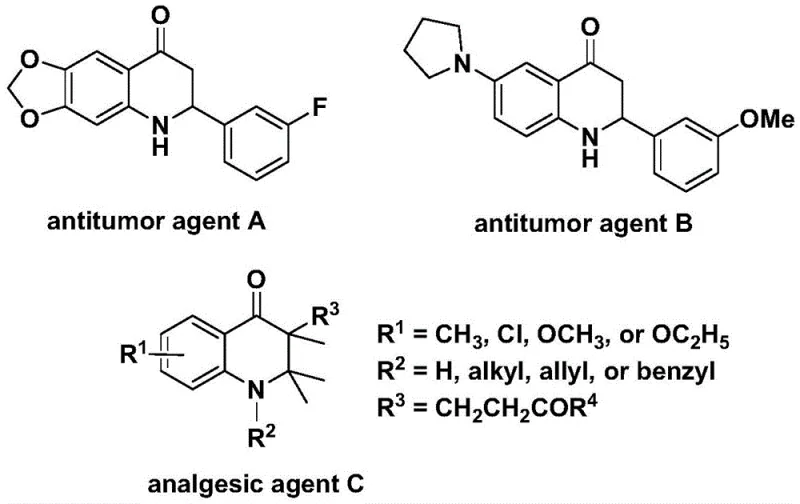
The significance of this technology extends beyond mere academic interest; it provides a practical solution for the reliable pharmaceutical intermediate supplier seeking to optimize production workflows. By enabling the efficient construction of the 2,3-dihydroquinolone skeleton through direct carbonylation, the method circumvents the need for multi-step sequences that often plague traditional syntheses. The versatility of the reaction allows for the generation of both 2-aryl and 3-alkyl substituted variants simply by modifying the olefin substrate, offering unparalleled flexibility for structure-activity relationship (SAR) studies. Furthermore, the use of a solid carbon monoxide surrogate enhances the safety profile of the process, making it highly attractive for commercial scale-up where handling hazardous gases is a major logistical and regulatory hurdle.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3-dihydroquinolone derivatives has relied on methodologies that often suffer from significant drawbacks regarding safety, efficiency, and substrate scope. Traditional carbonylation reactions frequently necessitate the use of high-pressure carbon monoxide gas, which poses severe safety risks due to its toxicity and flammability, requiring specialized high-pressure equipment and rigorous safety protocols that increase capital expenditure. Additionally, many conventional routes involve harsh reaction conditions or utilize stoichiometric amounts of toxic reagents, leading to complex waste streams and challenging purification processes that drive up manufacturing costs. The limited tolerance for sensitive functional groups in older methods often restricts the diversity of analogs that can be synthesized, forcing researchers to employ tedious protecting group strategies that add steps and reduce overall yield. These limitations collectively hinder the rapid development and cost reduction in API manufacturing, creating a bottleneck for bringing new therapies to market efficiently.
The Novel Approach
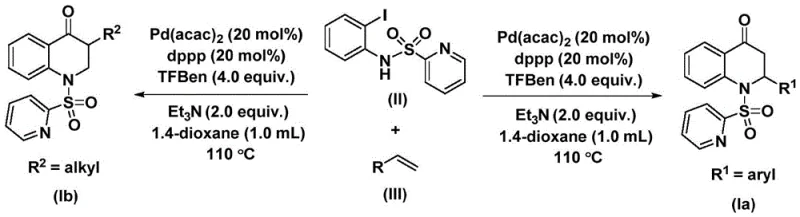
In stark contrast, the novel approach detailed in the patent utilizes a palladium-catalyzed system that operates under significantly milder and safer conditions by employing 1,3,5-trimesic acid phenol ester as a solid carbon monoxide surrogate. This innovation effectively eliminates the hazards associated with gaseous CO, allowing the reaction to proceed in standard laboratory glassware or reactors without the need for high-pressure infrastructure. The general reaction scheme demonstrates the seamless coupling of N-pyridine sulfonyl-o-iodoaniline with various olefins to form the target heterocycle in a single pot. This streamlined process not only simplifies the operational workflow but also enhances the atom economy of the transformation. The method's ability to accommodate diverse substituents on the olefin, ranging from electron-rich to electron-deficient aryl groups and various alkyl chains, underscores its robustness. By integrating these features, the novel approach offers a superior alternative for the commercial scale-up of complex pharmaceutical intermediates, ensuring high purity and consistent quality.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
The mechanistic pathway of this transformation is a sophisticated dance of organometallic steps orchestrated by the palladium catalyst. The cycle initiates with the oxidative addition of the palladium species into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate. This step is crucial as it activates the aromatic ring for subsequent functionalization. Following this activation, the carbon monoxide, released in situ from the thermal decomposition of the 1,3,5-trimesic acid phenol ester, inserts into the palladium-carbon bond to form an acyl-palladium species. This carbonyl insertion is the key step that introduces the ketone functionality essential for the quinolone core. The presence of the dppp ligand is vital here, as it stabilizes the palladium center and facilitates the necessary geometric arrangements for efficient insertion. Understanding these elementary steps allows process chemists to fine-tune reaction parameters such as temperature and ligand loading to maximize turnover numbers and minimize catalyst deactivation.
Subsequent to the formation of the acyl-palladium intermediate, the olefin substrate coordinates to the metal center and undergoes migratory insertion, creating a new carbon-carbon bond and extending the molecular framework. This alkene insertion step determines the regioselectivity of the final product, ensuring the correct placement of the substituent at the 2-position of the dihydroquinolone ring. The cycle concludes with an intramolecular nucleophilic attack or reductive elimination event that closes the six-membered ring and regenerates the active palladium catalyst. The specific structures of the reactants, such as the N-pyridine sulfonyl moiety shown in the formula, play a dual role: they act as the coupling partner and potentially assist in directing the cyclization through coordination or electronic effects. This detailed mechanistic understanding provides a strong foundation for troubleshooting potential impurities and optimizing the reaction for high-purity pharmaceutical intermediate production, ensuring that side reactions like beta-hydride elimination are minimized.

How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The execution of this synthesis is designed for practicality and reproducibility, making it ideal for both laboratory optimization and pilot plant operations. The protocol involves charging a reaction vessel with the palladium catalyst, the bidentate phosphine ligand, the base, the CO surrogate, the iodoaniline derivative, and the selected olefin in an aprotic organic solvent like dioxane. The mixture is then heated to temperatures between 100°C and 120°C, typically for a period of 24 to 48 hours, to drive the reaction to completion. The choice of solvent is critical, as it must effectively dissolve all reagents while remaining stable under the thermal conditions; dioxane has been identified as particularly effective for achieving high conversion rates. Post-reaction workup is straightforward, involving filtration to remove insoluble palladium residues followed by standard chromatographic purification. For a comprehensive guide on the specific molar ratios and detailed procedural nuances, please refer to the standardized synthesis steps outlined below.
- Combine palladium bis(acetylacetonate), dppp ligand, triethylamine, 1,3,5-trimesic acid phenol ester, N-pyridine sulfonyl-o-iodoaniline, and the chosen olefin in an organic solvent such as dioxane.
- Heat the reaction mixture to a temperature between 100°C and 120°C and maintain stirring for a duration of 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target substituted 2,3-dihydroquinolone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers transformative benefits that directly impact the bottom line and operational resilience. The shift from gaseous carbon monoxide to a solid surrogate fundamentally alters the risk profile of the manufacturing process, removing the need for specialized gas handling infrastructure and reducing insurance and compliance costs associated with toxic gas storage. This simplification translates into significant cost reduction in pharmaceutical intermediate manufacturing by lowering the barrier to entry for production facilities and minimizing downtime related to safety inspections. Furthermore, the high substrate compatibility means that a single production line can be utilized to generate a diverse library of analogs simply by swapping the olefin feedstock, thereby maximizing asset utilization and reducing the need for dedicated equipment for different products. This flexibility is invaluable for supply chain heads who must manage volatile demand and ensure continuity of supply for multiple drug candidates simultaneously.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and toxic gas handling systems drastically reduces capital expenditure and operational overhead. By using commercially available and inexpensive starting materials like olefins and simple iodoanilines, the raw material costs are kept low, while the high yields reported in the patent examples minimize waste and maximize output per batch. The simplified post-treatment process, which avoids complex extraction or distillation steps, further lowers utility consumption and labor costs, contributing to substantial overall cost savings.
- Enhanced Supply Chain Reliability: The reliance on stable, solid reagents rather than pressurized gases mitigates the risk of supply disruptions caused by transportation regulations or storage limitations. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of failed batches that can delay project timelines. Additionally, the broad availability of the required catalysts and ligands from global chemical suppliers ensures that the supply chain remains resilient against regional shortages, providing a reliable pharmaceutical intermediate supplier with the stability needed to meet long-term contractual obligations.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from milligram to gram scales with the potential for ton-scale production. The use of a solid CO surrogate minimizes the release of volatile organic compounds and toxic gases into the environment, aligning with increasingly stringent global environmental regulations. This green chemistry aspect not only simplifies waste disposal but also enhances the corporate sustainability profile, making the manufactured intermediates more attractive to environmentally conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and disclosures within the patent documentation, providing clarity on reaction parameters and substrate limitations. Understanding these details is essential for process development teams evaluating the feasibility of adopting this route for their specific pipeline candidates. The information covers catalyst loading, reaction times, and the nature of the substituents that can be successfully incorporated into the final heterocyclic structure.
Q: What is the primary advantage of using 1,3,5-trimesic acid phenol ester in this synthesis?
A: The use of 1,3,5-trimesic acid phenol ester serves as a solid carbon monoxide surrogate, eliminating the need for handling toxic and hazardous gaseous CO, thereby significantly enhancing operational safety and simplifying reactor requirements for industrial scale-up.
Q: What types of substituents are tolerated on the olefin substrate?
A: The method demonstrates excellent substrate compatibility, tolerating a wide range of substituents including substituted aryl groups (with methyl, tert-butyl, methoxy, fluorine, or chlorine) as well as linear or branched alkyl groups and silyl groups, allowing for diverse structural modifications.
Q: What catalyst system is employed for this carbonylation reaction?
A: The reaction utilizes a palladium catalyst system comprising palladium bis(acetylacetonate) [Pd(acac)2] as the metal source and 1,3-bis(diphenylphosphino)propane [dppp] as the ligand, which facilitates efficient oxidative addition and reductive elimination cycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into full-scale manufacturing. We are committed to delivering high-purity 2,3-dihydroquinolone intermediates that meet the most stringent purity specifications required by global regulatory bodies. Our state-of-the-art rigorous QC labs employ advanced analytical techniques to verify the identity and purity of every batch, guaranteeing that our clients receive materials that are ready for immediate use in downstream synthesis without the need for additional purification.
We invite you to collaborate with us to leverage this advanced palladium-catalyzed technology for your next project. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates how this specific route can optimize your budget and timeline. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us help you navigate the complexities of chemical manufacturing with confidence and precision.
