Revolutionizing Pharmaceutical Intermediate Production Through Advanced Dihydroquinolone Synthesis Technology
Patent CN112239456B discloses an innovative method for synthesizing substituted 2,3-dihydroquinolone compounds that serve as critical scaffolds in numerous bioactive molecules including antitumor agents A and B with demonstrated anti-cancer activity against human cells and analgesic agent C with potent pain-relieving properties as documented in peer-reviewed literature spanning decades of pharmaceutical research.
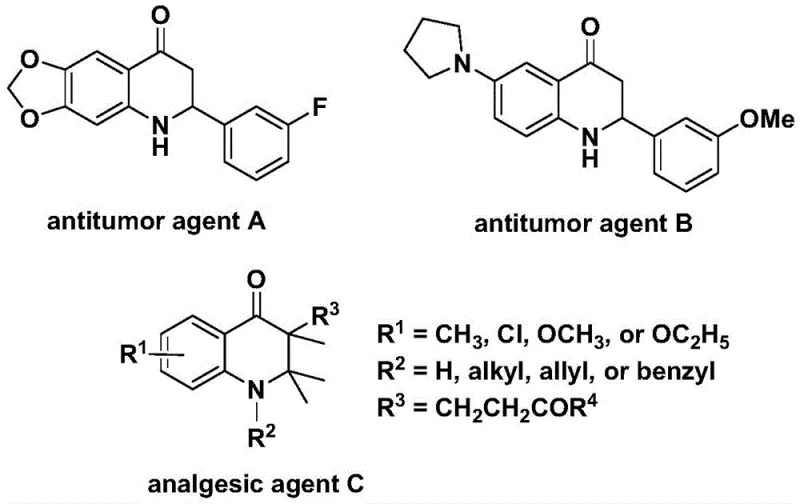
This breakthrough leverages palladium-catalyzed carbonylation to overcome traditional synthetic limitations through a single-step transformation operating under mild conditions of 100–120°C for 24–48 hours using dioxane solvent with Pd(acac)₂/dppp catalysts. The process achieves high yields without requiring complex purification steps while demonstrating exceptional substrate versatility that enables tailored synthesis of both 2-aryl and 3-alkyl derivatives essential for pharmaceutical development programs targeting oncology and pain management therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional syntheses of dihydroquinolone frameworks frequently rely on harsh reaction conditions such as strong acids or temperatures exceeding 150°C that cause decomposition of sensitive functional groups and reduce overall yields significantly across diverse substrate classes. These methods typically exhibit narrow substrate scope particularly when handling sterically hindered or electron-deficient aromatics common in modern drug candidates which limits their applicability in medicinal chemistry campaigns requiring structural diversity. Multi-step sequences involving pre-functionalized precursors increase both material costs and processing time while introducing additional impurity profiles that complicate regulatory approval pathways through increased analytical burden during quality control testing phases.
The Novel Approach
The patented methodology introduces a single-step palladium-catalyzed carbonylation process that operates under significantly milder conditions using commercially available Pd(acac)₂ with dppp ligand in dioxane solvent while employing N-pyridine sulfonyl-o-iodoaniline and olefin substrates with triethylamine additive as key components. This approach eliminates the need for pre-formed carbonyl components or protecting groups that plague conventional routes thereby reducing both material costs and processing time while maintaining exceptional functional group tolerance across diverse substrates including methyl-, chloro-, fluoro-substituted aromatics and various alkyl chains up to silyl groups. Crucially the simplified workup procedure involving only filtration and column chromatography minimizes waste generation while consistently delivering product purity meeting stringent pharmaceutical intermediate requirements without requiring specialized equipment or hazardous reagent handling protocols.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The catalytic cycle begins with oxidative addition of palladium(0) into the carbon-nitrogen bond of N-pyridine sulfonyl-o-iodoaniline forming an aryl palladium intermediate that subsequently undergoes carbon monoxide insertion from mesitylene phenol ester to generate an acyl palladium species which then coordinates with olefin substrates facilitating migratory insertion to form new carbon-carbon bonds before reductive elimination releases the final dihydroquinolone product while regenerating active catalyst species.
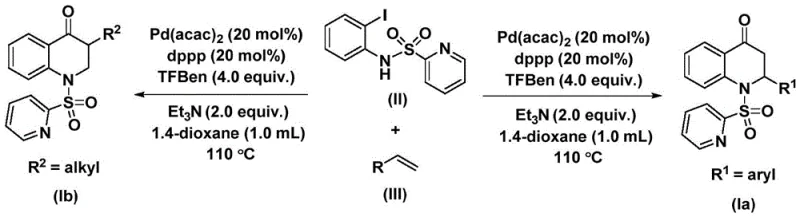
This well-defined pathway inherently minimizes impurity formation through controlled reaction kinetics while avoiding radical side reactions common in alternative methods; substrate design flexibility allows precise control over molecular architecture at both positions of the dihydroquinolone core enabling pharmaceutical researchers to rapidly generate compound libraries without modifying core synthetic protocols while maintaining consistent purity profiles essential for regulatory compliance in drug substance manufacturing.
How to Synthesize Substituted Dihydroquinolones Efficiently
This patented synthesis route represents a significant advancement over conventional methods by providing a direct pathway to complex dihydroquinolone structures through a single catalytic transformation that eliminates multiple protection/deprotection steps while maintaining high selectivity across diverse substrates using commercially available starting materials and standard laboratory equipment offering immediate practical utility for medicinal chemistry teams seeking rapid access to novel compound libraries.
- Prepare reaction mixture by combining N-pyridine sulfonyl-o-iodoaniline (0.5 mmol), olefin (2.0 mmol), Pd(acac)₂ (0.05 mmol), dppp ligand (0.1 mmol), triethylamine (2.0 equiv.), mesitylene phenol ester (4.0 mmol), and dioxane solvent (1 mL) in a Schlenk tube under inert atmosphere.
- Heat reaction mixture to 110°C with continuous stirring under nitrogen atmosphere for precisely 48 hours to ensure complete conversion while maintaining optimal reaction kinetics.
- Perform post-treatment by filtering reaction mixture through silica gel followed by standard column chromatography purification using established elution protocols to isolate pure substituted dihydroquinolone product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process directly addresses critical pain points in pharmaceutical supply chains by transforming production economics through simplified reaction sequences that reduce raw material consumption while enhancing yield reliability through consistent process control parameters applicable across different manufacturing scales without requiring extensive revalidation procedures.
- Cost Reduction in Manufacturing: The use of inexpensive starting materials such as o-iodoaniline derivatives combined with commercially available olefins substantially reduces raw material costs compared to conventional approaches requiring pre-functionalized building blocks; elimination of transition metal removal steps through efficient catalyst recovery lowers purification costs while avoiding expensive waste treatment procedures associated with heavy metal contamination.
- Enhanced Supply Chain Reliability: Reliance on globally sourced commodity chemicals rather than specialized intermediates minimizes supply chain vulnerabilities while enabling rapid scale-up from laboratory development to commercial production volumes; simplified process control parameters allow consistent manufacturing across different geographic locations without requiring extensive revalidation procedures.
- Scalability and Environmental Compliance: Reaction compatibility with standard stainless steel equipment and absence of hazardous byproducts facilitate seamless transition from gram-scale development to multi-ton production without infrastructure modifications; reduced solvent usage and elimination of toxic reagents significantly lower environmental impact while meeting increasingly stringent regulatory requirements for green chemistry practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented synthesis method in pharmaceutical manufacturing environments each response derived directly from experimental data documented in the original patent filing supporting informed decision-making by procurement supply chain and R&D leadership teams.
Q: What limitations does this method overcome compared to conventional dihydroquinolone syntheses?
A: Traditional methods require harsh conditions exceeding 150°C with narrow substrate scope and multi-step sequences generating complex impurity profiles; this patented approach operates at milder temperatures (100–120°C) with exceptional functional group tolerance across diverse substrates including sterically hindered compounds while eliminating protection/deprotection steps.
Q: How does substrate design flexibility benefit pharmaceutical development timelines?
A: The modular system enables precise control over substitution patterns at both positions of the dihydroquinolone core—aryl olefins produce 2-substituted derivatives while alkyl olefins yield 3-substituted products—allowing rapid generation of compound libraries for structure-activity relationship studies without modifying core synthetic conditions.
Q: What evidence supports scalability for commercial pharmaceutical manufacturing?
A: The patent demonstrates successful gram-scale synthesis with consistent yields across multiple substrates under standardized conditions using common laboratory equipment; this straightforward process utilizes globally available materials and simple workup procedures enabling seamless transition to multi-ton production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Dihydroquinolone Supplier
Our patented methodology represents a transformative approach to manufacturing complex heterocyclic intermediates essential for next-generation pharmaceuticals NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through state-of-the-art QC labs equipped with advanced analytical instrumentation ensuring consistent delivery meeting global regulatory standards for pharmaceutical applications.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative process can optimize your specific supply chain requirements please contact us to obtain detailed COA data and route feasibility assessments tailored to your production needs.
