Advanced Palladium-Catalyzed Carbonylation for Scalable N-Acyl Indole Production
Advanced Palladium-Catalyzed Carbonylation for Scalable N-Acyl Indole Production
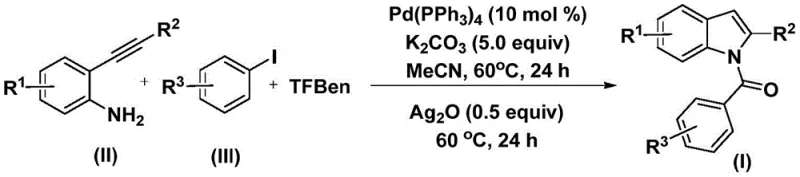
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing complex heterocyclic scaffolds, particularly indole derivatives which serve as critical backbones in numerous bioactive molecules. Patent CN112898192B introduces a significant advancement in this domain by disclosing a highly efficient preparation method for N-acyl indole compounds. This technology leverages a palladium-catalyzed carbonylation cyclization strategy that operates under remarkably mild conditions, utilizing 2-alkynyl aniline and aryl iodides as primary building blocks. The significance of this innovation lies in its ability to bypass the traditional limitations associated with gaseous carbon monoxide handling while maintaining high reaction efficiency and broad substrate tolerance. As a leading entity in chemical manufacturing, we recognize that such methodological improvements are pivotal for enhancing the reliability of pharmaceutical intermediate supplier networks globally.
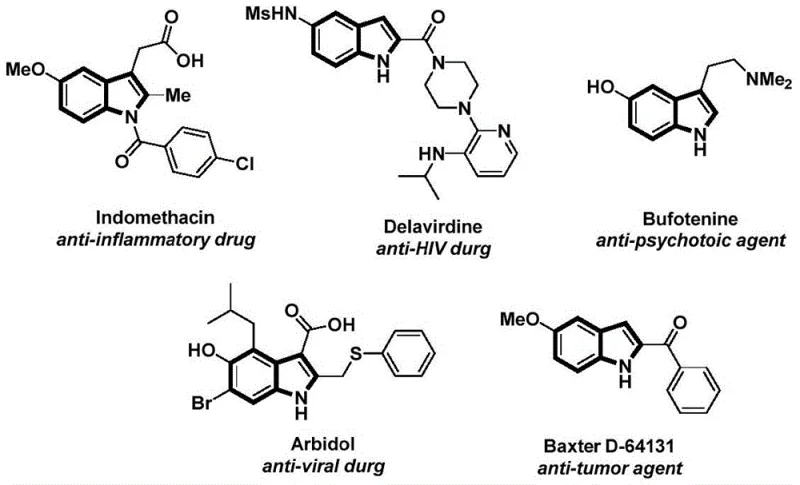
Indole structures are ubiquitous in medicinal chemistry, found in potent agents ranging from anti-inflammatory drugs like Indomethacin to anti-HIV medications such as Delavirdine. The capability to synthesize N-acylated variants of these structures efficiently opens new avenues for drug discovery and process optimization. The disclosed method not only simplifies the synthetic route but also aligns with modern green chemistry principles by employing solid CO surrogates, thereby addressing safety concerns inherent in large-scale carbonylation processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl indoles via carbonylation has been fraught with significant operational challenges that hinder industrial adoption. Traditional protocols often rely on the direct use of carbon monoxide gas, which poses severe safety risks due to its high toxicity and flammability, necessitating specialized high-pressure equipment and rigorous safety protocols that escalate capital expenditure. Furthermore, conventional palladium-catalyzed methods frequently suffer from poor atom economy and require harsh reaction conditions, such as elevated temperatures exceeding 100°C or the use of strong bases that can degrade sensitive functional groups on the substrate. These factors collectively contribute to inconsistent yields, complex impurity profiles, and difficulties in scaling up processes for commercial production, creating bottlenecks for procurement managers seeking cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in patent CN112898192B presents a transformative solution by utilizing phenol 1,3,5-tricarboxylate (TFBen) as a safe and effective solid carbon monoxide substitute. This innovation allows the reaction to proceed at a moderate temperature of 60°C in acetonitrile, significantly reducing energy consumption and thermal stress on the reactants. The process is designed as a streamlined one-pot sequence where the initial carbonylation is followed by an oxidative cyclization step mediated by silver oxide, eliminating the need for isolating unstable intermediates. This approach not only enhances operational safety by removing gaseous CO from the workflow but also improves the overall process mass intensity, making it an attractive option for high-purity pharmaceutical intermediate production.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The mechanistic pathway of this transformation is a sophisticated orchestration of organometallic steps that ensure high selectivity and yield. The cycle initiates with the oxidative addition of the palladium(0) catalyst into the carbon-iodine bond of the aryl iodide substrate, generating a reactive aryl-palladium(II) species. Subsequently, carbon monoxide, which is slowly released in situ from the decomposition of TFBen, inserts into the palladium-carbon bond to form an acyl-palladium intermediate. This acyl species then undergoes nucleophilic attack by the amino group of the 2-alkynyl aniline, followed by reductive elimination to yield an amide intermediate. The elegance of this system lies in the second phase, where the addition of silver oxide promotes the intramolecular cyclization of the alkyne moiety,最终 forming the rigid N-acyl indole core with high regioselectivity.
Controlling the impurity profile in such complex cascades is critical for regulatory compliance. The use of TFBen ensures a steady, low-concentration release of CO, which minimizes side reactions such as homocoupling of the aryl iodide or over-carbonylation that often plague batch processes with rapid gas introduction. Additionally, the choice of tetrakis(triphenylphosphine)palladium as the catalyst provides a stable ligand environment that prevents palladium black precipitation, ensuring consistent catalytic activity throughout the 48-hour reaction window. This mechanistic stability translates directly to a cleaner crude reaction mixture, reducing the burden on downstream purification units and facilitating the isolation of products with stringent purity specifications required for clinical applications.
How to Synthesize N-Acyl Indole Efficiently
Implementing this synthesis route requires precise control over stoichiometry and reaction timing to maximize the yield of the target N-acyl indole. The protocol dictates a specific molar ratio of reagents, typically employing 10 mol% of the palladium catalyst and 5.0 equivalents of potassium carbonate to drive the equilibrium forward. The reaction is conducted in two distinct thermal stages within the same vessel, first to establish the amide linkage and second to close the indole ring. For detailed operational parameters and specific reagent quantities tailored to your production scale, please refer to the standardized synthesis guide below.
- Combine palladium catalyst (Pd(PPh3)4), potassium carbonate, carbon monoxide substitute (TFBen), 2-alkynyl aniline, and aryl iodide in acetonitrile solvent.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and amide formation.
- Add silver oxide (Ag2O) to the mixture and continue heating at 60°C for another 24 hours to induce cyclization, followed by filtration and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers substantial benefits that extend beyond mere chemical yield. By shifting away from hazardous gaseous reagents to stable solid surrogates, the process drastically simplifies the infrastructure requirements for manufacturing facilities, allowing for production in standard glass-lined reactors without the need for specialized high-pressure autoclaves. This reduction in equipment complexity directly correlates to lower capital depreciation costs and reduced maintenance overheads, providing a clear pathway for cost reduction in fine chemical manufacturing. Furthermore, the reliance on commercially available starting materials such as aryl iodides and substituted anilines ensures a robust and resilient supply chain, mitigating the risks associated with sourcing exotic or proprietary precursors.
- Cost Reduction in Manufacturing: The elimination of high-pressure CO handling equipment and the use of mild reaction conditions (60°C) significantly lower the energy footprint and safety compliance costs associated with production. The high atom economy of the carbonylation step ensures that raw material costs are optimized, as fewer equivalents of expensive palladium catalysts are wasted on side reactions. Additionally, the simplified workup procedure, which involves basic filtration and chromatography, reduces solvent consumption and waste disposal fees, contributing to a more sustainable and economically viable production model.
- Enhanced Supply Chain Reliability: The substrates employed in this synthesis, specifically 2-alkynyl anilines and various aryl iodides, are commodity chemicals available from multiple global vendors, preventing single-source bottlenecks. The robustness of the reaction against moisture and air, facilitated by the stable nature of the solid CO source, allows for more flexible scheduling and reduced downtime due to environmental controls. This reliability is crucial for maintaining continuous supply lines for critical pharmaceutical intermediates, ensuring that downstream API synthesis is never interrupted by raw material shortages or quality deviations.
- Scalability and Environmental Compliance: The protocol's compatibility with acetonitrile, a widely accepted industrial solvent, facilitates easy scale-up from gram to multi-kilogram batches without significant re-optimization. The absence of toxic gaseous emissions simplifies the permitting process for new manufacturing lines and aligns with increasingly strict environmental regulations regarding volatile organic compounds (VOCs) and hazardous air pollutants. The high selectivity of the reaction minimizes the generation of difficult-to-separate byproducts, thereby reducing the load on wastewater treatment facilities and supporting the company's commitment to green chemistry initiatives.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for successful technology transfer and quality assurance. The following questions address common inquiries regarding the operational safety, substrate scope, and purification strategies associated with this palladium-catalyzed protocol. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation to assist technical teams in evaluating feasibility.
Q: What is the source of carbon monoxide in this synthesis?
A: The process utilizes phenol 1,3,5-tricarboxylate (TFBen) as a solid carbon monoxide substitute, eliminating the need for handling hazardous gaseous CO directly.
Q: What is the role of Silver Oxide (Ag2O) in the reaction?
A: Silver oxide acts as an oxidant and promoter in the second stage, facilitating the intramolecular cyclization of the intermediate amide to form the final N-acyl indole structure.
Q: Does this method tolerate diverse functional groups?
A: Yes, the protocol demonstrates excellent substrate compatibility, tolerating substituents such as halogens (F, Cl, Br), alkyl groups (Me, tBu), and alkoxy groups (OMe) on both the aniline and aryl iodide components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we leverage cutting-edge synthetic methodologies like the one described in CN112898192B to deliver superior chemical solutions to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of N-acyl indole intermediate meets the exacting standards required for pharmaceutical development and manufacturing.
We invite you to collaborate with us to explore how this advanced carbonylation technology can optimize your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your supply chain efficiency and product quality.
