Advanced Palladium-Catalyzed Synthesis of N-Acyl Indoles for Pharmaceutical Applications
Advanced Palladium-Catalyzed Synthesis of N-Acyl Indoles for Pharmaceutical Applications
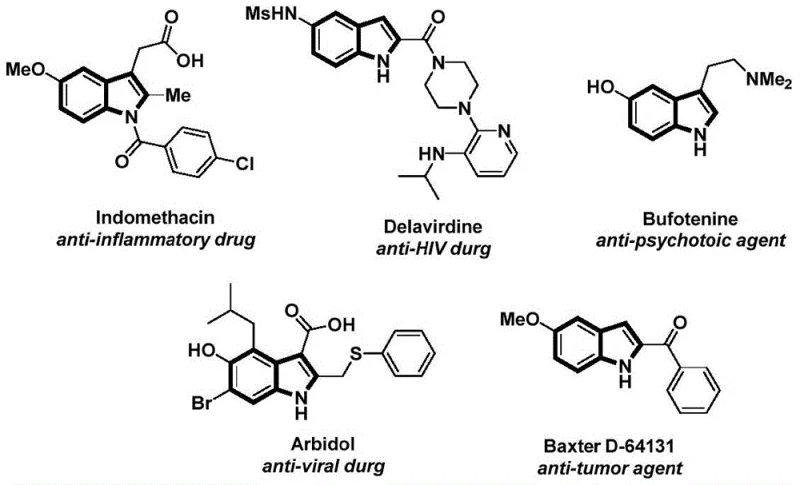
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, safer, and cost-effective synthetic routes. A significant breakthrough in this domain is documented in Chinese Patent CN112898192B, which discloses a robust preparation method for N-acyl indole compounds. Indole scaffolds are ubiquitous in medicinal chemistry, serving as the core structure for a vast array of bioactive molecules including antitumor, anti-inflammatory, and antiviral agents. The structural diversity and biological relevance of these compounds make them a critical focus for R&D teams globally. As illustrated in the provided data, well-known drugs such as Indomethacin, Delavirdine, and Bufotenine all share this privileged indole backbone, highlighting the immense commercial value of mastering their synthesis.
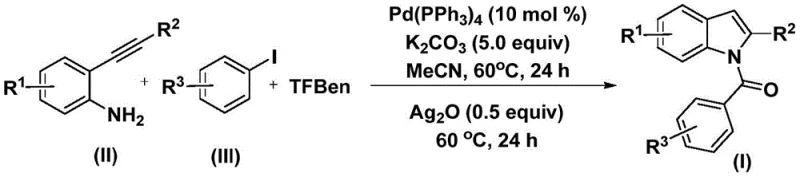
This patent introduces a novel approach that leverages palladium-catalyzed carbonylation cyclization to construct the N-acyl indole core in a single pot. Unlike traditional methods that may require harsh conditions or multiple steps, this technique utilizes 2-alkynyl aniline and aryl iodides as readily available starting materials. The process is characterized by its operational simplicity, high reaction efficiency, and exceptional substrate compatibility. For procurement managers and supply chain directors, the ability to access such versatile intermediates through a streamlined process translates directly into reduced lead times and enhanced supply continuity for downstream API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl indole compounds via carbonylation reactions has been underrepresented in the literature and industrial practice, despite the clear potential of the carbonylation strategy for constructing carbonyl-containing frameworks. Conventional carbonylation often relies on the use of carbon monoxide gas, which poses significant safety hazards due to its toxicity and the requirement for high-pressure equipment. Furthermore, existing methods for N-acylation of indoles frequently suffer from limited substrate scope, requiring specific functional group protection or deprotection strategies that add steps and reduce overall atom economy. The lack of widely applied, general methods for this transformation has created a bottleneck in the rapid development of new indole-based therapeutics, forcing R&D teams to rely on less efficient multi-step sequences that drive up costs and extend project timelines.
The Novel Approach
The methodology described in Patent CN112898192B offers a transformative solution to these challenges by employing a palladium-catalyzed system that operates under mild conditions. The core innovation lies in the use of phenyl 1,3,5-tricarboxylate (TFBen) as a solid carbon monoxide substitute, which safely releases CO in situ, thereby circumventing the need for handling hazardous CO gas. The reaction proceeds efficiently at a moderate temperature of 60°C in acetonitrile, utilizing potassium carbonate as a base and tetrakis(triphenylphosphine)palladium as the catalyst. This one-pot protocol effectively merges the carbonylation and cyclization steps, allowing for the direct conversion of 2-alkynyl anilines and aryl iodides into the desired N-acyl indoles. The general reaction scheme highlights the elegance of this transformation, showcasing how complex heterocyclic systems can be assembled from simple precursors with high precision.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
Understanding the mechanistic pathway is crucial for R&D directors aiming to optimize this process for specific targets. The reaction initiates with the oxidative addition of the palladium catalyst into the aryl iodide bond, generating a reactive aryl-palladium intermediate. Subsequently, carbon monoxide, which is slowly released from the decomposition of the phenyl 1,3,5-tricarboxylate additive, inserts into this palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the amino group of the 2-alkynyl aniline, followed by reductive elimination to yield an amide precursor. The final and critical step involves the addition of silver oxide, which promotes the intramolecular cyclization of the amide to form the stable N-acyl indole ring system. This mechanistic sequence ensures high regioselectivity and minimizes the formation of unwanted byproducts.
From an impurity control perspective, the choice of reagents plays a pivotal role. The use of TFBen ensures a steady, low concentration of CO, which helps prevent side reactions such as homocoupling of the alkyne or over-carbonylation. Furthermore, the compatibility of the system with various functional groups—such as halogens (F, Cl, Br), alkyl groups (Me, tBu), and alkoxy groups (OMe)—demonstrates the robustness of the catalytic cycle. The patent data confirms that substituents at the R1, R2, and R3 positions do not significantly inhibit the reaction, allowing for the synthesis of a diverse library of derivatives. This broad tolerance is essential for medicinal chemists who need to explore structure-activity relationships (SAR) without being constrained by synthetic limitations.
How to Synthesize N-Acyl Indole Compounds Efficiently
The practical execution of this synthesis is designed for scalability and ease of operation, making it an attractive option for contract development and manufacturing organizations (CDMOs). The standard procedure involves charging a reaction vessel with the palladium catalyst, base, CO source, and substrates in an organic solvent, followed by a two-stage heating protocol. The detailed operational parameters, including specific molar ratios and temperature profiles, are critical for achieving the high yields reported in the patent examples. For technical teams looking to implement this route, adherence to the specified stoichiometry—particularly the 0.1:5:5:0.5 ratio of catalyst to base to CO source to silver oxide—is key to maximizing efficiency.
- Combine palladium catalyst, potassium carbonate, phenyl 1,3,5-tricarboxylate (TFBen), 2-alkynyl aniline, and aryl iodide in an organic solvent such as acetonitrile.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and amide formation steps.
- Add silver oxide to the mixture and continue heating at 60°C for another 24 hours to promote cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers distinct strategic advantages over traditional synthetic routes. The primary benefit stems from the significant simplification of the supply chain for raw materials. The starting materials, specifically 2-alkynyl anilines and aryl iodides, are commodity chemicals that are widely available from multiple global suppliers, reducing the risk of single-source dependency. Moreover, the replacement of gaseous carbon monoxide with a solid surrogate (TFBen) eliminates the need for specialized high-pressure reactors and associated safety infrastructure, which drastically lowers the capital expenditure required for setting up production lines. This shift not only enhances workplace safety but also streamlines regulatory compliance regarding hazardous material handling.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive and abundant reagents. By avoiding the use of expensive transition metal catalysts in excessive amounts and utilizing a cheap base like potassium carbonate, the direct material costs are kept low. Furthermore, the one-pot nature of the reaction reduces solvent consumption and energy usage associated with intermediate isolation and purification steps. The elimination of high-pressure equipment maintenance and the reduced need for specialized safety protocols further contribute to substantial operational cost savings, making the final N-acyl indole intermediates more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is vital for maintaining uninterrupted supply to downstream API manufacturers. The mild reaction temperature of 60°C allows for the use of standard heating equipment, reducing the likelihood of thermal runaways or equipment failures that could disrupt production schedules. Additionally, the high substrate compatibility means that the same production line can be easily adapted to synthesize different analogues by simply swapping the aryl iodide or alkyne components, providing the flexibility needed to respond quickly to changing market demands or custom synthesis requests.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the homogeneous nature of the reaction and the simplicity of the workup procedure, which typically involves filtration and standard chromatography. The use of acetonitrile as a solvent, while requiring proper recovery systems, is well-established in the industry, and the solid waste generated (primarily silver salts and palladium residues) can be managed through established metal recovery protocols. This aligns with modern green chemistry principles by improving atom economy and reducing the overall environmental footprint of the manufacturing process, thereby facilitating smoother environmental impact assessments and regulatory approvals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing a clear picture of what partners can expect when integrating this method into their production workflows. Understanding these nuances helps in making informed decisions about process adoption and resource allocation.
Q: What is the primary advantage of using phenyl 1,3,5-tricarboxylate (TFBen) in this synthesis?
A: TFBen serves as a solid, safe, and easy-to-handle carbon monoxide substitute, eliminating the need for high-pressure CO gas equipment and enhancing operational safety in industrial settings.
Q: What represents the typical reaction yield for this palladium-catalyzed method?
A: According to the patent data, the method demonstrates high reaction efficiency with yields ranging significantly across different substrates, often exceeding 50% to 80% for optimized examples like I-8 and I-15.
Q: Is this method suitable for large-scale manufacturing of drug intermediates?
A: Yes, the process utilizes commercially available starting materials, mild reaction temperatures (60°C), and standard post-treatment procedures like filtration and column chromatography, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the drug development pipeline. Our team of expert chemists has extensively evaluated the palladium-catalyzed carbonylation route described in CN112898192B and confirmed its potential for delivering high-purity N-acyl indoles. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet stringent purity specifications. Our rigorous QC labs are equipped to perform comprehensive impurity profiling, guaranteeing that every batch of N-acyl indole intermediate adheres to the highest industry standards for pharmaceutical applications.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthetic technology for their projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and target molecules. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary candidates. Let us help you accelerate your timeline to market with reliable, cost-effective, and scalable chemical solutions.
