Advanced Synthesis of N-N Axis Chiral Pyrrole Derivatives for Oncology Applications
The pharmaceutical industry is constantly seeking novel scaffolds that offer unique stereochemical properties and potent biological activities, particularly in the realm of oncology. Patent CN114524701B introduces a groundbreaking methodology for the synthesis of N-N axis chiral pyrrole derivatives, a class of compounds that has historically been difficult to access with high stereoselectivity. This patent details a robust catalytic system utilizing chiral phosphoric acids to facilitate the condensation of indoleamines or pyrrole amines with 1,4-diketone derivatives. The resulting structures, represented generally by Formula 3 and Formula 5, exhibit a distinct N-N axial chirality that is crucial for interacting with specific biological targets. Unlike traditional methods that rely on dynamic kinetic resolution or desymmetrization, this invention employs an innovative in-situ ring formation strategy. This approach not only expands the structural diversity of available chiral pyrroles but also ensures exceptional enantiomeric excess values, reaching up to 96% ee in optimized conditions. For research directors and procurement specialists, this represents a significant leap forward in accessing high-value intermediates for anticancer drug discovery.
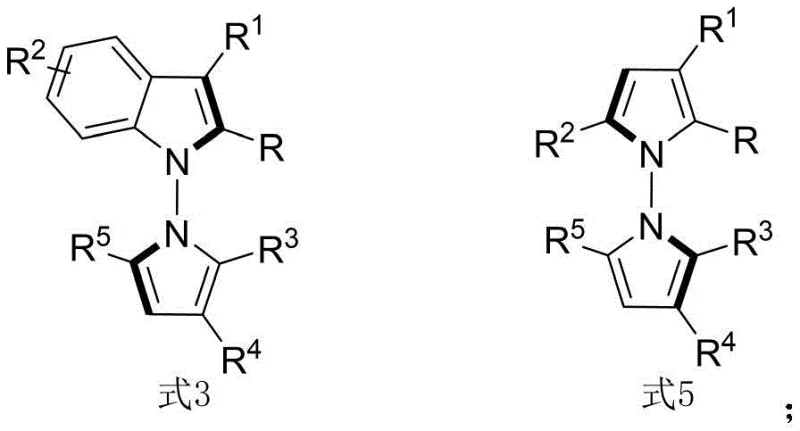
The limitations of conventional methods for constructing N-N axially chiral systems have long hindered the rapid development of this chemical space. Historically, chemists have relied on complex multi-step sequences involving dynamic kinetic resolution, which often suffer from a theoretical maximum yield of 50% unless specific recycling strategies are employed. Furthermore, existing desymmetrization reactions frequently require expensive transition metal catalysts, such as palladium or rhodium complexes, which introduce significant cost burdens and potential heavy metal contamination issues in the final API. These traditional pathways are often sensitive to moisture and air, requiring stringent inert atmosphere conditions that complicate scale-up operations. In contrast, the novel approach described in CN114524701B utilizes a metal-free organocatalytic system. By leveraging the dual hydrogen-bonding activation capability of chiral phosphoric acids, the reaction proceeds efficiently at room temperature (25°C) in carbon tetrachloride. This shift from metal catalysis to organocatalysis fundamentally alters the economic and operational landscape of producing these intermediates, offering a cleaner, safer, and more atom-economical route that is inherently more suitable for GMP manufacturing environments.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
The core of this technological breakthrough lies in the precise activation of the 1,4-diketone substrate by the chiral phosphoric acid catalyst. The catalyst, particularly the spiro-skeleton derivative designated as compound 6j, acts as a bifunctional activator. The acidic proton of the phosphate group forms a strong hydrogen bond with the carbonyl oxygen of the diketone, increasing its electrophilicity. Simultaneously, the basic phosphoryl oxygen can interact with the nucleophilic amine group of the indole or pyrrole substrate. This dual activation brings the reactants into a rigid, well-defined chiral environment within the catalyst's pocket. This spatial confinement is critical for inducing the axial chirality during the ring-closing step. The steric bulk of the substituents on the catalyst backbone (such as the 2,4,6-trimethylphenyl groups in 6j) effectively blocks one face of the approaching nucleophile, ensuring that the cyclization occurs with high facial selectivity. This mechanistic elegance allows for the formation of the sterically hindered N-N bond with remarkable precision, avoiding the formation of racemic byproducts that would otherwise require difficult and yield-lossing separation processes.
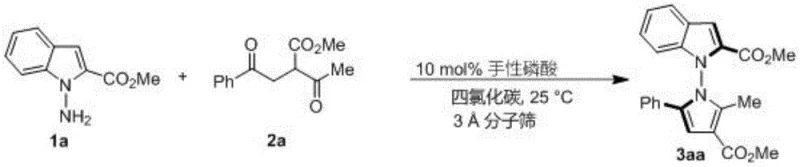
Impurity control is another critical aspect where this mechanism offers distinct advantages. In traditional metal-catalyzed couplings, side reactions such as homocoupling or beta-hydride elimination can lead to complex impurity profiles that are challenging to purge. However, the organocatalytic nature of this reaction minimizes such pathways. The reaction is highly chemoselective for the condensation between the amine and the specific carbonyl centers of the 1,4-diketone. The use of 3A molecular sieves as an additive further drives the equilibrium towards the product by sequestering the water generated during the condensation, thereby preventing hydrolysis of the product or the starting materials. This results in a cleaner crude reaction mixture, simplifying the downstream purification process. For quality control teams, this means a more predictable impurity profile and a higher likelihood of meeting stringent residual solvent and heavy metal specifications required for pharmaceutical intermediates without extensive reprocessing.
How to Synthesize N-N Axis Chiral Pyrrole Derivatives Efficiently
The synthesis protocol outlined in the patent provides a straightforward and reproducible method for generating these high-value chiral scaffolds. The process begins with the preparation of the reaction mixture in a standard laboratory setup, requiring no specialized high-pressure equipment. The key to success lies in the precise stoichiometric ratio of the reactants and the choice of the optimal catalyst loading. Typically, a slight excess of the 1,4-diketone derivative is used to ensure complete consumption of the more valuable amine substrate. The reaction is monitored via thin-layer chromatography (TLC), allowing for real-time assessment of conversion. Once the reaction is deemed complete, the workup involves simple filtration to remove the molecular sieves and catalyst residues, followed by concentration under reduced pressure. The final purification is achieved through standard silica gel column chromatography using a petroleum ether and ethyl acetate system. This simplicity makes the technology highly transferable from bench scale to pilot plant operations.
- Prepare the reaction mixture by combining indoleamine or pyrrole amine with 1,4-diketone derivatives in carbon tetrachloride solvent.
- Add 3A molecular sieves as an additive and introduce 10 mol% of chiral phosphoric acid catalyst (preferably compound 6j).
- Stir the reaction at 25°C until TLC indicates completion, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers compelling strategic benefits that extend beyond mere technical feasibility. The shift towards organocatalysis significantly reduces the dependency on precious metals, which are subject to volatile market pricing and supply chain disruptions. By eliminating the need for palladium or rhodium catalysts, manufacturers can achieve substantial cost savings in raw material procurement. Furthermore, the removal of heavy metals from the process simplifies the regulatory compliance burden, as there is no need for expensive metal scavenging resins or rigorous ICP-MS testing for residual metals in the final product. This streamlined workflow translates directly into reduced manufacturing lead times and lower overall production costs.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the use of inexpensive, commercially available organocatalysts and solvents. Unlike transition metal catalysts that often require ligand synthesis and strict handling protocols, chiral phosphoric acids are robust and can be recovered or used in low loadings (10 mol%). The mild reaction conditions (25°C) eliminate the need for energy-intensive heating or cryogenic cooling, resulting in significantly lower utility costs. Additionally, the high yields (up to 98%) and excellent stereoselectivity minimize the loss of valuable starting materials and reduce the volume of waste generated, contributing to a more sustainable and cost-efficient manufacturing model.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including various substituted indoleamines and 1,4-diketones, are readily accessible from global chemical suppliers. The broad substrate scope demonstrated in the patent indicates that the process is tolerant to a wide range of functional groups, allowing for flexibility in sourcing. This versatility mitigates the risk of supply bottlenecks associated with niche or custom-synthesized precursors. Moreover, the robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, which is critical for maintaining a stable supply of intermediates for downstream drug development programs.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method aligns well with green chemistry principles. The absence of toxic heavy metals reduces the hazard profile of the waste stream, simplifying disposal and treatment procedures. The use of carbon tetrachloride as a solvent is noted, but the process efficiency allows for effective solvent recovery and recycling systems to be implemented at scale. The simple workup procedure involving filtration and chromatography is easily adaptable to large-scale continuous processing or batch reactors, facilitating the commercial scale-up of complex pharmaceutical intermediates without the need for specialized infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: What is the primary advantage of using chiral phosphoric acid in this synthesis?
A: The use of chiral phosphoric acid catalysts, specifically spiro-skeleton derivatives like 6j, enables extremely high enantioselectivity (up to 96% ee) under mild room temperature conditions, eliminating the need for harsh reagents or expensive transition metals.
Q: What biological activity do these N-N axis chiral pyrrole derivatives exhibit?
A: Biological testing demonstrates that these derivatives possess strong cytotoxic activity and high sensitivity against QGP-1 pancreatic tumor cells, indicating significant potential for oncology drug development.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process utilizes conventional reaction conditions, readily available substrates, and simple post-treatment procedures (filtration and chromatography), making it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Pyrrole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technology described in CN114524701B for the development of next-generation oncology therapeutics. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative academic research into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee the high enantiomeric excess and chemical purity required for your API synthesis.
We invite you to collaborate with us to leverage this cutting-edge synthetic route for your drug discovery pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Contact us today to request specific COA data for our catalog of chiral intermediates or to discuss route feasibility assessments for your custom synthesis projects. Let us help you secure a reliable supply of high-quality N-N axis chiral pyrrole derivatives to accelerate your path to clinical success.
