Revolutionizing 5-Trifluoromethyl-1,2,3-Triazole Production via Metal-Free Base-Promoted Cyclization
Revolutionizing 5-Trifluoromethyl-1,2,3-Triazole Production via Metal-Free Base-Promoted Cyclization
The landscape of heterocyclic chemistry is constantly evolving, driven by the need for safer, more efficient, and scalable synthetic routes for high-value intermediates. A significant breakthrough in this domain is detailed in Chinese Patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. This technology represents a paradigm shift away from hazardous traditional methodologies, offering a metal-free, base-promoted pathway that utilizes readily available starting materials. For R&D directors and procurement managers in the pharmaceutical and agrochemical sectors, this innovation addresses critical pain points regarding safety, cost, and supply chain reliability. The core of this invention lies in the reaction between trifluoroethylimidoyl chloride and diazo compounds, facilitated by cesium carbonate, to construct the triazole scaffold with high efficiency and exceptional functional group tolerance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,3-triazoles has been fraught with significant safety and operational challenges. As illustrated in the prior art, traditional routes predominantly rely on copper-catalyzed [3+2] cycloadditions between alkynes and organic azides, or organocatalytic reactions involving azides and trifluoromethyl ketones. 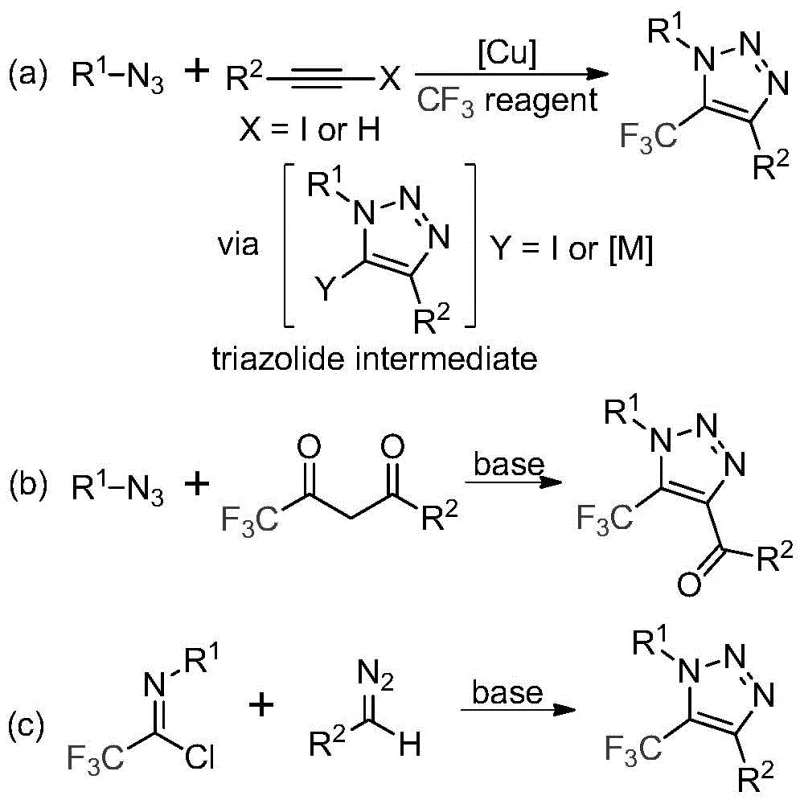 The reliance on organic azides is a major bottleneck; these reagents are notoriously toxic, unstable, and possess a high risk of explosion, necessitating specialized equipment and rigorous safety protocols that drive up operational expenditures. Furthermore, copper-catalyzed methods often require the removal of trace heavy metals to meet stringent pharmaceutical purity standards, adding complex purification steps and increasing waste generation. These factors collectively hinder the commercial scale-up of complex pharmaceutical intermediates, creating supply chain vulnerabilities and limiting the speed at which new drug candidates can be advanced.
The reliance on organic azides is a major bottleneck; these reagents are notoriously toxic, unstable, and possess a high risk of explosion, necessitating specialized equipment and rigorous safety protocols that drive up operational expenditures. Furthermore, copper-catalyzed methods often require the removal of trace heavy metals to meet stringent pharmaceutical purity standards, adding complex purification steps and increasing waste generation. These factors collectively hinder the commercial scale-up of complex pharmaceutical intermediates, creating supply chain vulnerabilities and limiting the speed at which new drug candidates can be advanced.
The Novel Approach
In stark contrast, the methodology described in patent CN113121462B circumvents these hazards entirely by eliminating the need for organic azides and transition metal catalysts. The novel approach employs a base-promoted reaction between trifluoroethylimidoyl chloride and diazo compounds. This strategy not only enhances the safety profile of the manufacturing process but also simplifies the reaction workflow. By avoiding toxic azides and expensive metal catalysts, the process inherently reduces the cost of goods sold (COGS) and minimizes the environmental footprint associated with heavy metal waste disposal. The use of commercially available and stable starting materials ensures a reliable supply chain, while the mild reaction conditions allow for broader substrate scope, enabling the synthesis of diverse derivatives crucial for medicinal chemistry optimization campaigns.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic pathway of this transformation is a fascinating example of efficient bond construction under mild conditions. The reaction is hypothesized to proceed through a base-promoted intermolecular nucleophilic addition-elimination sequence. Initially, the diazo compound acts as a nucleophile, attacking the electrophilic carbon of the trifluoroethylimidoyl chloride. This step facilitates the formation of a critical carbon-carbon bond while eliminating a chloride ion. Following this initial addition, the intermediate undergoes an intramolecular 5-endo-dig cyclization. This cyclization step is pivotal, as it closes the five-membered triazole ring, establishing the final heterocyclic architecture. The electron-withdrawing nature of the trifluoromethyl group plays a crucial role in stabilizing the intermediates and driving the reaction forward, ensuring high yields even with diverse substituents.
Understanding the impurity profile is essential for R&D teams aiming to replicate this process at scale. The use of cesium carbonate as a promoter is particularly advantageous due to its solubility properties and basicity, which effectively drive the reaction to completion without promoting excessive side reactions. The patent data indicates that the reaction tolerates a wide range of functional groups on both the N1 and C4 positions of the triazole ring, including esters, ketones, and phosphonates. This robustness suggests that the mechanism is highly selective, minimizing the formation of regioisomers or polymeric byproducts that often plague triazole syntheses. Consequently, the downstream purification is streamlined, typically requiring only standard filtration and column chromatography to achieve high-purity products suitable for further biological evaluation.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The practical implementation of this synthesis is straightforward, making it accessible for both laboratory discovery and pilot plant operations. The general procedure involves combining the trifluoroethylimidoyl chloride, the specific diazo compound, and cesium carbonate in an aprotic organic solvent. Acetonitrile is identified as the preferred solvent due to its ability to dissolve the reactants effectively and promote high conversion rates. The reaction mixture is then heated to a moderate temperature, typically around 60°C, and stirred for a period of 8 to 16 hours. Upon completion, the workup is simple: the mixture is filtered to remove inorganic salts, concentrated, and the residue is purified via silica gel chromatography. 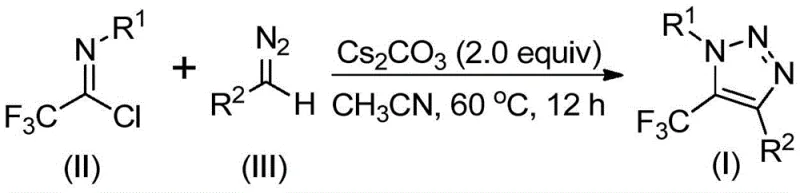 The detailed standardized synthesis steps, including specific molar ratios and purification parameters for various derivatives, are outlined below to guide technical teams in adopting this methodology.
The detailed standardized synthesis steps, including specific molar ratios and purification parameters for various derivatives, are outlined below to guide technical teams in adopting this methodology.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an aprotic organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature range of 50-70°C and maintain stirring for 8 to 16 hours to ensure complete conversion.
- Filter the reaction mixture, concentrate, and purify the crude product via silica gel column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers tangible strategic advantages beyond mere chemical novelty. The primary value proposition lies in the drastic simplification of the supply chain and the enhancement of operational safety. By removing the requirement for hazardous organic azides, facilities can significantly reduce their insurance premiums and safety compliance costs. Furthermore, the elimination of transition metal catalysts removes the need for expensive scavenging resins and complex metal testing, directly contributing to cost reduction in pharmaceutical intermediate manufacturing. The reliance on stable, commercially available starting materials ensures consistent supply continuity, mitigating the risks associated with sourcing exotic or unstable reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from multiple vectors. Firstly, the avoidance of copper catalysts eliminates the cost of the metal itself and the downstream processing required to remove trace metal residues to ppm levels, which is a regulatory requirement for APIs. Secondly, the use of cesium carbonate, while a premium base, is offset by the high reaction efficiency and yield, reducing the overall material throughput required per kilogram of product. Thirdly, the simplified workup procedure reduces labor hours and solvent consumption during purification. These factors combine to create a leaner, more cost-effective production model that improves margin potential for high-volume commercial runs.
- Enhanced Supply Chain Reliability: Supply chain resilience is critical in the post-pandemic era. This synthesis route relies on building blocks like aromatic amines and diazo precursors that are widely produced and stocked by global chemical suppliers. Unlike specialized azide reagents which may have limited vendors and long lead times, the inputs for this process are commodity chemicals. This abundance ensures that production schedules are not disrupted by raw material shortages. Additionally, the robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, further stabilizing the supply output and reducing the rate of batch failures.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method is superior. The absence of explosive azides allows for safer scale-up from gram to multi-kilogram quantities without requiring blast-proof infrastructure. The generation of waste is minimized as there are no heavy metal contaminants to treat, simplifying wastewater management. The process aligns well with green chemistry principles by utilizing atom-economical transformations and reducing the E-factor of the synthesis. This compliance with stricter environmental regulations future-proofs the manufacturing process against tightening global standards on chemical emissions and waste disposal.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common questions regarding the process specifics and its applicability to various projects. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear picture of what to expect during technology transfer.
Q: Why is this new synthesis method safer than traditional triazole production?
A: Traditional methods often rely on organic azides, which are toxic and potentially explosive. This patented process utilizes stable diazo compounds and imidoyl chlorides, significantly reducing safety hazards and handling costs in a manufacturing environment.
Q: What are the optimal reaction conditions for scaling this process?
A: The patent indicates that using cesium carbonate as a base in acetonitrile at 60°C provides high conversion rates. The reaction is robust and can be extended from gram-scale to industrial production with standard post-treatment procedures like filtration and chromatography.
Q: Can this method accommodate diverse functional groups on the triazole ring?
A: Yes, the method demonstrates excellent substrate tolerance. It supports various substituents on both the N1 and C4 positions, including aryl, alkoxycarbonyl, and phosphoryl groups, making it highly versatile for designing complex API intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free cyclization technology for the development of next-generation therapeutics and agrochemicals. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorine chemistry and heterocycle synthesis, adhering to stringent purity specifications and rigorous QC labs to guarantee the highest quality intermediates for your supply chain.
We invite you to leverage our technical expertise to optimize your production costs and accelerate your project timelines. Whether you require custom synthesis of specific triazole derivatives or full-scale commercial manufacturing, our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can become your trusted partner in delivering high-purity 5-trifluoromethyl-1,2,3-triazoles.
