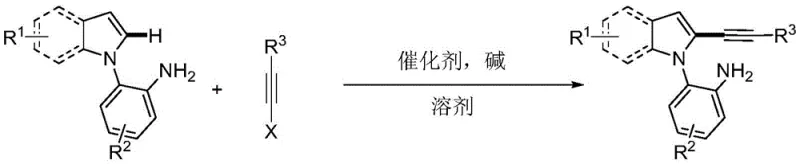
Advanced Synthesis of 2-Alkynyl Indoles via Primary Amine Direction for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds with high precision and efficiency. A significant breakthrough in this domain is documented in patent CN108864164B, which discloses a novel synthesis method for primary amine-guided 2-alkynyl indole compounds. This technology addresses long-standing challenges in organic synthesis by leveraging a primary amine as a directing group to achieve exceptional regioselectivity at the C2 position of the indole ring. The process utilizes a palladium salt catalyst and operates under relatively mild conditions, distinguishing itself from harsher traditional protocols. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and cost-effective manufacturing of key pharmaceutical intermediates. The ability to construct these highly functionalized structures using water as a solvent not only adheres to green chemistry principles but also simplifies the downstream processing requirements, offering tangible benefits for commercial scale-up operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of functionalized indole derivatives has relied heavily on pre-functionalization strategies, such as C-H halogenation followed by classical cross-coupling reactions. These conventional pathways are often plagued by inherent limitations, particularly regarding regioselectivity. Due to the electronic nature of the indole nucleus, the C3 position is significantly more reactive towards electrophilic substitution and metallization than the C2 position. Consequently, achieving exclusive functionalization at the C2 site typically requires multi-step protection and deprotection sequences, which drastically reduce overall atom economy and increase production costs. Furthermore, many existing transition metal-catalyzed direct alkynylation methods suffer from narrow substrate scope or require expensive, specialized ligands and anhydrous organic solvents. These factors create substantial bottlenecks in supply chain reliability and elevate the environmental burden associated with solvent disposal, making them less attractive for large-scale industrial applications where cost and safety are paramount concerns.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN108864164B introduces a streamlined cross-coupling reaction between 2-(1H-indol-1-yl)aniline and alkyne halides. By employing a primary amine group as an internal directing group, the system effectively overrides the natural electronic bias of the indole ring, guiding the palladium catalyst specifically to the C2 position. This innovation eliminates the need for cumbersome protection groups and allows for direct C-H activation with high single-selectivity. A defining feature of this novel approach is its compatibility with water as a solvent, either alone or in mixtures, which is a rare and valuable trait for palladium-catalyzed couplings. This shift not only reduces the reliance on hazardous organic solvents but also facilitates easier product isolation. The method demonstrates broad applicability across various substrates, tolerating electron-donating and electron-withdrawing groups alike, thereby providing a versatile platform for synthesizing diverse libraries of 2-alkynyl indoles essential for drug discovery and material science.
Mechanistic Insights into Pd-Catalyzed C-H Alkynylation
The mechanistic pathway of this synthesis is a sophisticated example of directed C-H activation, driven by the coordination capabilities of the primary amine. Under the promotion of a base, the 2-(1H-indol-1-yl)aniline substrate coordinates with the palladium salt catalyst. This coordination is the critical step that forms a stable six-membered palladacycle intermediate. The formation of this cyclic intermediate effectively locks the metal center in proximity to the C2-H bond, lowering the activation energy required for cleavage and ensuring that the reaction occurs exclusively at this position rather than the more electronically favored C3 site. Once the palladacycle is established, the alkyne halide undergoes oxidative addition to the palladium center. This is followed by a reductive elimination step that forges the new carbon-carbon bond, releasing the desired 2-alkynyl indole product and regenerating the active catalyst species. This catalytic cycle is highly efficient and minimizes the formation of regioisomeric impurities, which is a common pain point in indole chemistry.
From an impurity control perspective, this mechanism offers distinct advantages for manufacturing high-purity intermediates. Because the regioselectivity is governed by the thermodynamic stability of the six-membered palladacycle rather than purely electronic effects, the process is robust against variations in substrate electronics. Whether the indole ring bears electron-withdrawing groups like cyano or halogens, or electron-donating groups like methyl or methoxy, the directing effect of the amine remains dominant. This consistency ensures a clean reaction profile with minimal byproduct formation, simplifying the purification process. For quality control teams, this translates to a more predictable impurity profile and higher yields of the target compound. The use of water as a solvent further aids in suppressing side reactions that might occur in organic media, contributing to the overall purity of the final product and reducing the burden on downstream chromatographic separation steps.
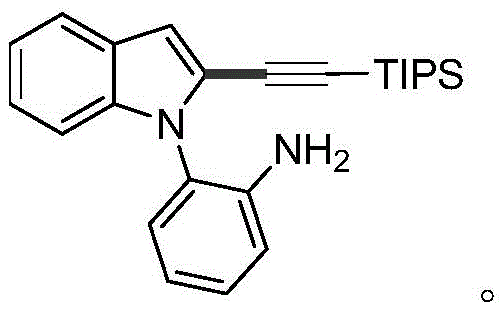
How to Synthesize 2-Alkynyl Indole Efficiently
The operational simplicity of this synthesis route makes it highly attractive for process chemists aiming to translate laboratory success to pilot plant production. The procedure involves charging a reactor with the 2-(1H-indol-1-yl)aniline substrate, the alkyne halide (typically a silyl-protected bromoalkyne), a palladium catalyst such as palladium tetranitrate tetrafluoroborate, and a cesium or potassium base. The reaction is conducted in water or a water-toluene mixture at temperatures ranging from 80°C to 110°C. After stirring for 12 to 24 hours, the mixture is cooled, extracted with ethyl acetate, and purified via standard column chromatography. This straightforward workflow minimizes the need for specialized equipment or inert atmosphere techniques, lowering the barrier for implementation. For detailed standardized synthesis steps and specific molar ratios optimized for different substrates, please refer to the guide below.
- Charge the reactor with 2-(1H-indol-1-yl)aniline substrate, alkyne halide, palladium salt catalyst, base, and water solvent.
- Stir the reaction mixture at 80-110°C for 12-24 hours to facilitate the cross-coupling reaction.
- Cool to room temperature, extract with ethyl acetate, dry over magnesium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis technology offers compelling strategic advantages centered around cost efficiency and operational reliability. The primary driver for cost reduction lies in the solvent system; replacing expensive, flammable, and toxic organic solvents with water significantly lowers raw material costs and reduces the expenses associated with solvent recovery and hazardous waste disposal. Furthermore, the starting materials, such as o-iodoaniline and indole derivatives, are commodity chemicals that are readily available from multiple global suppliers, mitigating the risk of supply chain disruptions. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or reagent quality, leading to more consistent batch-to-batch performance and reduced rejection rates.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from several key factors inherent to its design. Firstly, the use of water as a solvent eliminates the need for costly anhydrous conditions and expensive organic solvents, directly impacting the variable cost of goods sold. Secondly, the high atom economy and single selectivity of the reaction mean that less raw material is wasted on forming unwanted isomers or byproducts, maximizing the yield of the valuable 2-alkynyl indole. Thirdly, the simplified workup procedure, which avoids complex quenching steps often required for sensitive organometallic reactions, reduces labor and utility costs. Collectively, these factors contribute to a leaner manufacturing process that enhances profit margins without compromising on product quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the accessibility and stability of the reagents involved. The palladium catalysts used are common industrial grades, and the alkyne halides can be synthesized from widely available precursors. This reduces dependency on niche suppliers who might face production bottlenecks. Additionally, the tolerance of the reaction to various functional groups allows for flexibility in sourcing; if a specific substituted aniline is unavailable, alternative derivatives can often be substituted without requiring a complete re-optimization of the process. This flexibility ensures continuity of supply even when market dynamics shift for specific raw materials, providing a buffer against volatility in the chemical marketplace.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by its benign operating conditions. The absence of pyrophoric reagents or extreme pressures makes the technology safer to operate in large reactors, reducing insurance and safety compliance costs. From an environmental standpoint, the alignment with green chemistry principles—specifically the use of water and the generation of less hazardous waste—simplifies regulatory compliance. Facilities can operate with lower environmental permits burdens, and the reduced ecological footprint enhances the corporate sustainability profile, which is increasingly important for partnerships with major pharmaceutical companies committed to ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this primary amine-guided synthesis technology. These insights are derived directly from the patent specifications and experimental data, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is regioselectivity critical in indole alkynylation?
A: Indole compounds naturally possess higher electron cloud density at the C3 position compared to the C2 position, making C3 metallization and subsequent reaction much easier. Achieving selective functionalization at the C2 position is a significant synthetic challenge that this primary amine-directed method successfully overcomes.
Q: What are the environmental advantages of this synthesis method?
A: Unlike traditional methods that rely heavily on volatile organic compounds (VOCs), this novel approach utilizes water as the primary solvent. This aligns with green chemistry principles, significantly reducing the environmental footprint and simplifying waste treatment protocols for large-scale manufacturing.
Q: Does this method tolerate diverse functional groups?
A: Yes, the methodology demonstrates excellent substrate universality. It effectively accommodates various substituents on both the indole ring (such as fluoro, chloro, methyl, methoxy, and cyano groups) and the aniline moiety, allowing for the construction of a wide library of highly functionalized derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkynyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in modern drug development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN108864164B can be seamlessly translated into industrial reality. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We understand that the transition from benchtop to plant floor requires not just chemical expertise but also deep engineering knowledge to manage heat transfer, mixing, and safety protocols effectively, all of which are core competencies of our technical team.
We invite you to collaborate with us to leverage this efficient synthesis route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. By partnering with us, you gain access to specific COA data and comprehensive route feasibility assessments that will help you make informed decisions about your supply chain strategy. Contact us today to discuss how we can support your need for high-purity 2-alkynyl indoles and drive value through chemical innovation.
