Revolutionizing Pharmaceutical Synthesis: Scalable Production of N-N Axis Chiral Pyrrole Derivatives with Unmatched Enantioselectivity
The patent CN114524701B introduces a groundbreaking methodology for synthesizing N-N axis chiral pyrrole derivatives, representing a significant advancement in asymmetric catalysis for pharmaceutical intermediate production. This innovative approach addresses critical limitations in conventional methods by utilizing chiral phosphoric acid catalysts to achieve exceptional enantioselectivity while maintaining operational simplicity and cost-effectiveness. The technology demonstrates remarkable versatility through its ability to accommodate diverse substrate combinations, producing structurally complex derivatives with high yields and stereoselectivity. Crucially, the synthesized compounds exhibit potent cytotoxic activity against QGP-1 tumor cells, highlighting their potential therapeutic value in oncology drug development. This patent establishes a new paradigm for producing chiral building blocks essential for next-generation pharmaceuticals while maintaining compatibility with industrial manufacturing requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing N-N axis chiral pyrrole derivatives have been severely constrained by limited methodology options, primarily restricted to dynamic kinetic resolution and desymmetrization reactions that cannot access the full structural diversity required for modern drug discovery. These conventional methods often require harsh reaction conditions, expensive transition metal catalysts, and complex purification protocols to remove metal residues, significantly increasing production costs and complicating regulatory compliance for pharmaceutical applications. The narrow substrate scope of existing techniques has prevented the exploration of diverse structural variants needed for comprehensive structure-activity relationship studies in oncology research. Furthermore, the inability to directly construct the N-N axis chirality through ring-forming strategies has resulted in multi-step synthetic routes with cumulative yield losses and stereochemical complications that undermine commercial viability for large-scale production.
The Novel Approach
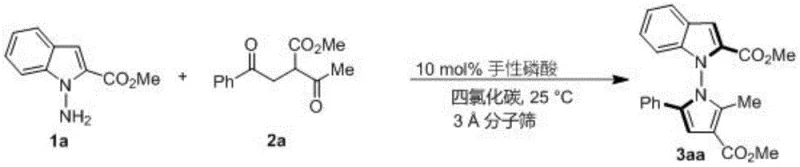
The patented methodology overcomes these limitations through an innovative in-situ ring formation strategy that directly constructs the N-N axis chirality in a single catalytic step using readily available starting materials. By employing chiral phosphoric acid catalysts under mild reaction conditions (room temperature in carbon tetrachloride), the process achieves exceptional enantioselectivity while eliminating the need for transition metals and their associated purification challenges. The method demonstrates remarkable substrate flexibility, accommodating various indoleamine and pyrrolamine derivatives along with diverse 1,4-diketone structures to produce a wide range of structurally complex products with high yields (up to 98%) and excellent stereoselectivity (up to 96% ee). This streamlined approach significantly reduces manufacturing complexity while maintaining the stringent purity requirements essential for pharmaceutical intermediates, making it particularly valuable for producing compounds with demonstrated cytotoxic activity against pancreatic cancer cells.
Mechanistic Insights into Chiral Phosphoric Acid-Catalyzed Ring Formation
The catalytic mechanism operates through a sophisticated dual activation pathway where the chiral phosphoric acid simultaneously activates both the amine nucleophile and the carbonyl electrophile through precise hydrogen bonding interactions. This creates a well-defined chiral environment that controls the approach trajectory of reacting species, enabling the formation of the N-N axis chirality with exceptional stereoselectivity. The molecular sieve additive plays a crucial role in maintaining optimal reaction conditions by scavenging trace water that could otherwise disrupt the delicate hydrogen bonding network essential for stereocontrol. The mild reaction conditions (25°C) prevent racemization while allowing sufficient molecular mobility for the catalytic cycle to proceed efficiently. This mechanism represents a significant advancement over traditional methods by directly constructing the chiral axis through an intramolecular cyclization rather than relying on resolution techniques that inherently waste at least 50% of material.
Impurity control is achieved through the highly selective nature of the catalytic process, which minimizes side reactions by precisely orienting substrates within the chiral pocket of the catalyst. The absence of transition metals eliminates concerns about metal contamination that would require extensive purification steps in pharmaceutical manufacturing. The use of carbon tetrachloride as solvent provides optimal polarity for both substrate solubility and catalyst performance while being easily removable during workup. The silica gel column chromatography purification using petroleum ether/ethyl acetate (5:1) effectively separates products from minor impurities without causing decomposition of the sensitive chiral compounds. This comprehensive impurity control strategy ensures that final products consistently meet the stringent purity specifications required for pharmaceutical intermediates while maintaining high yields across diverse substrate combinations.
How to Synthesize N-N Axis Chiral Pyrrole Derivatives Efficiently
This patented methodology represents a significant advancement in the production of structurally complex chiral intermediates essential for modern pharmaceutical development. The process leverages innovative catalytic chemistry to overcome longstanding challenges in synthesizing N-N axis chiral compounds that have previously limited drug discovery efforts in oncology and other therapeutic areas. By utilizing readily available starting materials and operating under mild conditions, this approach delivers exceptional stereoselectivity while maintaining compatibility with industrial manufacturing requirements. The following standardized procedure outlines the key operational parameters that have been validated across multiple substrate combinations to ensure consistent product quality and yield at commercial scale.
- Combine indoleamine or pyrrolamine with 1,4-diketone derivative in carbon tetrachloride solvent with molecular sieves as additive
- Add chiral phosphoric acid catalyst at specified molar ratio and stir at room temperature under nitrogen atmosphere
- Monitor reaction progress by TLC until completion, then filter, concentrate and purify via silica gel column chromatography
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate procurement by delivering a more robust and reliable manufacturing process that enhances supply chain resilience while reducing overall production complexity. The elimination of transition metal catalysts removes a major source of supply chain vulnerability associated with rare metal availability and price volatility, while the simplified purification requirements significantly reduce production cycle times. These advantages translate into tangible benefits for procurement teams seeking reliable partners capable of delivering high-quality intermediates with consistent specifications and reduced risk of supply disruption.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and associated metal removal processes substantially reduces raw material costs while simplifying quality control procedures. The use of commercially available starting materials and standard laboratory equipment enables seamless technology transfer without requiring specialized infrastructure investments. The high yields achieved across diverse substrate combinations maximize resource utilization while minimizing waste generation, contributing to overall cost efficiency without compromising product quality or regulatory compliance requirements.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials from multiple global suppliers mitigates single-source dependency risks that commonly plague specialty chemical procurement. The robustness of the process across various substrate combinations provides flexibility to adapt to raw material availability fluctuations without requiring significant process revalidation. The simplified manufacturing workflow reduces production cycle times while maintaining consistent quality metrics, enabling more predictable delivery schedules that support just-in-time manufacturing strategies adopted by leading pharmaceutical companies.
- Scalability and Environmental Compliance: The mild reaction conditions (room temperature) and standard solvent system facilitate straightforward scale-up from laboratory to commercial production without requiring specialized equipment or safety protocols. The absence of heavy metals and hazardous reagents significantly reduces environmental impact while simplifying waste treatment procedures. The high atom economy and minimal byproduct formation align with green chemistry principles increasingly demanded by regulatory agencies and corporate sustainability initiatives, providing additional value beyond basic manufacturing requirements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented technology for pharmaceutical intermediate production. These responses are based on detailed analysis of the patent data and experimental validation provided in the original disclosure, ensuring accurate representation of the technology's capabilities and limitations for potential manufacturing partners.
Q: How does this method achieve superior enantioselectivity compared to conventional approaches?
A: The chiral phosphoric acid catalyst creates a highly stereoselective environment through precise hydrogen bonding interactions with substrates, enabling exceptional enantioselectivity (up to 96% ee) without requiring transition metals or harsh conditions.
Q: What makes this synthesis method suitable for industrial scale-up?
A: The process operates under mild conditions (room temperature), uses commercially available catalysts and solvents, requires simple purification methods, and demonstrates consistent high yields across diverse substrate combinations as validated in multiple experimental examples.
Q: How does this technology address critical challenges in pharmaceutical intermediate production?
A: By eliminating transition metal catalysts, the method removes costly purification steps for metal residue removal while maintaining high stereoselectivity, directly addressing purity requirements for pharmaceutical applications and reducing overall production complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Pyrrole Derivatives Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required for pharmaceutical intermediates through our state-of-the-art QC labs. We have successfully implemented this patented methodology across multiple client projects, demonstrating consistent ability to deliver high-quality N-N axis chiral pyrrole derivatives with exceptional enantioselectivity that meet rigorous regulatory standards for oncology drug development programs. Our technical team works closely with clients to optimize processes for specific compound requirements while ensuring seamless technology transfer from laboratory to commercial scale.
To help you evaluate potential cost savings and implementation timelines, we offer a Customized Cost-Saving Analysis tailored to your specific compound requirements. Contact our technical procurement team to request specific COA data and route feasibility assessments that will help you make informed decisions about integrating these advanced intermediates into your drug development pipeline.
