Scalable Metal-Free Synthesis of 3-Hydroxymethyl-4-Phenyl-3,4-Dihydroquinolinone for Advanced Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust, scalable, and environmentally benign pathways for constructing complex heterocyclic scaffolds essential for modern drug discovery. Patent CN112125843B introduces a groundbreaking methodology for the preparation of 3-hydroxymethyl-4-phenyl-3,4-dihydroquinolinone compounds, a privileged structural motif found in numerous bioactive molecules. This innovation addresses critical bottlenecks in traditional synthesis by utilizing potassium monopersulfate (Oxone) as a dual-function promoter in an acetonitrile medium. The process facilitates a tandem epoxidation-intramolecular Friedel-Crafts alkylation sequence without the need for external transition metal catalysts or added protonic acids. For R&D directors and process chemists, this represents a significant leap forward in atom economy and operational simplicity, offering a direct route from readily available N-methyl-N-aryl-2-phenylacrylamides to high-value intermediates. The mild reaction conditions and high functional group tolerance make this technology particularly attractive for the rapid generation of diverse compound libraries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 3,4-dihydroquinolinone core has often relied on multi-step sequences involving harsh reagents and expensive catalytic systems. Traditional approaches frequently necessitate the use of precious transition metals such as palladium, rhodium, or ruthenium to facilitate C-H activation or cross-coupling reactions, which introduces significant cost burdens and regulatory hurdles regarding residual metal limits in final API products. Furthermore, many existing protocols require stoichiometric amounts of strong Lewis or Brønsted acids to drive cyclization, leading to corrosion issues, difficult waste stream management, and poor compatibility with acid-sensitive functional groups. These conventional methods often suffer from narrow substrate scopes, where electron-deficient or sterically hindered substrates fail to react efficiently, limiting their utility in medicinal chemistry campaigns. The reliance on toxic solvents and complex purification steps to remove metal residues further exacerbates the environmental footprint and production costs associated with these legacy processes.
The Novel Approach
In stark contrast, the methodology disclosed in CN112125843B utilizes a metal-free strategy driven by the inexpensive and commercially available oxidant, potassium monopersulfate. This novel approach streamlines the synthesis into a single pot operation where the oxidant simultaneously effects the epoxidation of the alkene moiety and, through its acidic byproduct potassium bisulfate, catalyzes the subsequent intramolecular cyclization.  As illustrated in the reaction scheme, the transformation proceeds smoothly in acetonitrile at 90°C, achieving yields up to 80% for optimized substrates. This eliminates the need for separate oxidation and cyclization steps, drastically reducing processing time and solvent consumption. The absence of heavy metals simplifies the downstream purification process, as there is no need for specialized scavengers or extensive chromatography to meet strict pharmaceutical purity standards. This streamlined workflow not only enhances the overall throughput but also aligns perfectly with green chemistry principles, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
As illustrated in the reaction scheme, the transformation proceeds smoothly in acetonitrile at 90°C, achieving yields up to 80% for optimized substrates. This eliminates the need for separate oxidation and cyclization steps, drastically reducing processing time and solvent consumption. The absence of heavy metals simplifies the downstream purification process, as there is no need for specialized scavengers or extensive chromatography to meet strict pharmaceutical purity standards. This streamlined workflow not only enhances the overall throughput but also aligns perfectly with green chemistry principles, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Oxone-Mediated Epoxidation-Intramolecular Friedel-Crafts Alkylation
A deep understanding of the reaction mechanism is crucial for process optimization and scale-up. The transformation begins with the electrophilic attack of the active oxygen species from potassium monopersulfate on the electron-rich double bond of the N-methyl-N-aryl-2-phenylacrylamide substrate. This initial step generates an epoxide intermediate along with potassium bisulfate as a byproduct. Uniquely, this byproduct serves as an in situ source of protons, eliminating the requirement for外加 (added) acid. The protonation of the epoxide oxygen activates the three-membered ring towards nucleophilic attack. 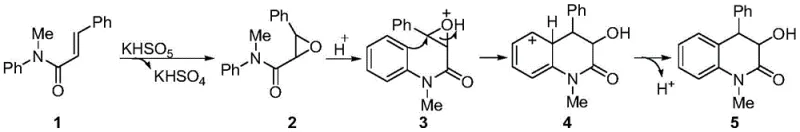 Following protonation, the electron-rich aromatic ring performs an intramolecular Friedel-Crafts alkylation at the benzylic position of the opened epoxide. This cyclization step constructs the six-membered dihydroquinolinone ring system with high regioselectivity. The final stage involves a dehydroaromatization or rearrangement that stabilizes the system into the observed 3-hydroxymethyl-4-phenyl-3,4-dihydroquinolinone product. This mechanistic pathway highlights the elegant dual role of Oxone, acting first as an oxygen transfer agent and subsequently providing the acidic environment necessary for ring closure. Such a self-sufficient catalytic cycle minimizes reagent complexity and reduces the potential for side reactions often associated with exogenous acid additives.
Following protonation, the electron-rich aromatic ring performs an intramolecular Friedel-Crafts alkylation at the benzylic position of the opened epoxide. This cyclization step constructs the six-membered dihydroquinolinone ring system with high regioselectivity. The final stage involves a dehydroaromatization or rearrangement that stabilizes the system into the observed 3-hydroxymethyl-4-phenyl-3,4-dihydroquinolinone product. This mechanistic pathway highlights the elegant dual role of Oxone, acting first as an oxygen transfer agent and subsequently providing the acidic environment necessary for ring closure. Such a self-sufficient catalytic cycle minimizes reagent complexity and reduces the potential for side reactions often associated with exogenous acid additives.
Furthermore, the robustness of this mechanism is evidenced by its exceptional tolerance to various electronic environments on the substrate. The electrophilic nature of the epoxidation step is sufficiently potent to proceed even with moderately deactivated alkenes, while the intramolecular cyclization is driven by the proximity effect and the stability of the resulting heterocycle. Experimental data confirms that substituents such as methyl, methoxy, and bromine groups on the aniline ring do not inhibit the reaction, allowing for the synthesis of a diverse array of derivatives. 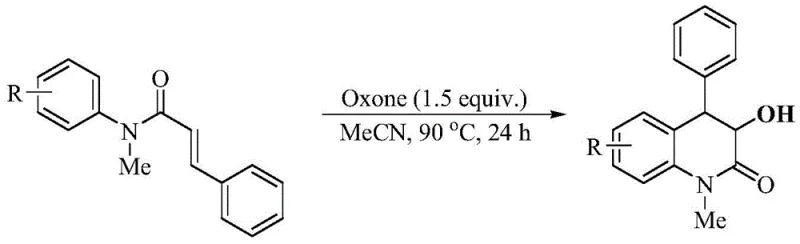 This broad compatibility is vital for medicinal chemists who need to explore structure-activity relationships (SAR) without being constrained by synthetic limitations. The ability to tolerate halogens like bromine is particularly valuable, as it leaves a handle for further functionalization via cross-coupling chemistry in later stages of drug development. The control over impurities is inherently better in this metal-free system, as there are no metal-ligand complexes to decompose into hard-to-remove byproducts, ensuring a cleaner crude profile and higher final purity.
This broad compatibility is vital for medicinal chemists who need to explore structure-activity relationships (SAR) without being constrained by synthetic limitations. The ability to tolerate halogens like bromine is particularly valuable, as it leaves a handle for further functionalization via cross-coupling chemistry in later stages of drug development. The control over impurities is inherently better in this metal-free system, as there are no metal-ligand complexes to decompose into hard-to-remove byproducts, ensuring a cleaner crude profile and higher final purity.
How to Synthesize 3-Hydroxymethyl-4-Phenyl-3,4-Dihydroquinolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and safety. The protocol dictates the use of acetonitrile as the sole reaction medium, as screening of other common solvents like dichloroethane, toluene, and DMF resulted in no reaction, highlighting the unique solvation properties required for this specific oxidant-substrate interaction. The concentration of the starting material should be maintained between 0.1 and 0.5 mol/L to balance reaction kinetics with heat dissipation. The stoichiometry of potassium monopersulfate is critical; using 1.5 equivalents relative to the substrate has been identified as the optimal ratio. Deviating from this, either by reducing the oxidant load or increasing it excessively, leads to diminished returns in yield. The reaction is thermally driven, requiring a temperature of 90°C to proceed to completion within a 24-hour window. Lower temperatures result in incomplete conversion, while significantly higher temperatures may promote degradation. Detailed standardized operating procedures for this synthesis are provided below.
- Dissolve N-methyl-N-aryl-2-phenylacrylamide in acetonitrile to achieve a concentration of 0.1-0.5 mol/L.
- Add 1.5 equivalents of potassium monopersulfate (Oxone) to the reaction mixture under heating conditions.
- Maintain the reaction temperature at 90°C for 24 hours to complete the epoxidation-intramolecular Friedel-Crafts alkylation cascade.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this metal-free protocol offers tangible strategic benefits beyond mere chemical elegance. The primary driver for cost optimization lies in the replacement of expensive noble metal catalysts with commodity-grade potassium monopersulfate. Transition metals like palladium are subject to volatile market pricing and supply chain disruptions, whereas Oxone is produced on a massive industrial scale with stable availability. By eliminating the need for these precious metals, manufacturers can significantly reduce the raw material cost per kilogram of the intermediate. Moreover, the removal of heavy metals from the process flow negates the need for costly purification steps such as metal scavenging resins or repeated recrystallizations designed to meet ppm-level residual metal specifications. This simplification of the downstream processing directly translates to reduced labor hours, lower solvent usage, and decreased waste disposal costs, contributing to a leaner and more profitable manufacturing operation.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the drastic simplification of the reagent profile. By utilizing a single, inexpensive oxidant that performs multiple chemical functions, the process reduces the total number of input materials required. This consolidation lowers inventory holding costs and simplifies procurement logistics. Additionally, the absence of transition metals means that the expensive infrastructure often required for handling air- or moisture-sensitive catalysts is unnecessary. The reaction can be run in standard glass-lined or stainless steel reactors without special passivation, further lowering capital expenditure requirements for production facilities. The high atom efficiency of the tandem reaction ensures that a larger proportion of the starting mass ends up in the final product, minimizing waste generation fees.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, non-specialized raw materials. N-methyl-N-aryl-2-phenylacrylamides are easily synthesized from commodity anilines and cinnamoyl chlorides, ensuring a robust upstream supply base. Unlike proprietary ligands or custom-synthesized catalysts that may have long lead times and single-source risks, the reagents for this process are stocked by multiple global chemical suppliers. This diversification of supply sources mitigates the risk of production stoppages due to vendor shortages. Furthermore, the stability of the reagents allows for bulk purchasing and long-term storage without significant degradation, enabling manufacturers to take advantage of favorable market pricing and maintain healthy safety stocks to buffer against market volatility.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process is exceptionally well-suited for commercial scale-up of complex pharmaceutical intermediates. The use of acetonitrile, a solvent with well-established recovery and recycling protocols, facilitates closed-loop solvent management systems that minimize VOC emissions. The reaction operates at atmospheric pressure and moderate temperatures (90°C), removing the need for high-pressure autoclaves or cryogenic cooling systems, which simplifies engineering controls and improves operational safety. The generation of inorganic salts (potassium sulfate/bisulfate) as the primary byproduct is environmentally benign compared to the heavy metal waste streams associated with traditional catalysis. This aligns with increasingly stringent global environmental regulations, reducing the compliance burden and potential liability for the manufacturing site while enhancing the company's sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Oxone-mediated synthesis. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for large-scale production. The answers provided reflect the specific advantages of the metal-free cascade mechanism and its impact on process efficiency and product quality.
Q: Why is acetonitrile critical for this Oxone-mediated cyclization?
A: Experimental data indicates that acetonitrile is the only effective solvent among those tested (including DCM, toluene, DMF, and ethyl acetate). Other solvents resulted in no reaction, suggesting acetonitrile plays a unique role in stabilizing the transition state or solubilizing the oxidant for this specific cascade.
Q: Does this method require expensive transition metal catalysts?
A: No. A key advantage of Patent CN112125843B is the complete elimination of transition metal catalysts. Potassium monopersulfate serves a dual function as both the oxidant for epoxidation and the source of protons (via KHSO4 byproduct) for the subsequent Friedel-Crafts cyclization.
Q: What is the functional group tolerance of this synthesis?
A: The process demonstrates excellent compatibility with various substituents on the aryl ring, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing halogens like bromine, yielding products with efficiencies ranging from 65% to 80%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxymethyl-4-Phenyl-3,4-Dihydroquinolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthetic methodologies in modern pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale discoveries like the one described in CN112125843B are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of this Oxone-mediated reaction, guaranteeing consistent batch-to-batch quality. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of 3-hydroxymethyl-4-phenyl-3,4-dihydroquinolinone meets the highest international standards for API intermediates, free from problematic metal residues.
We invite global partners to leverage our technical expertise to optimize their supply chains. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Our technical procurement team is ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this green chemistry approach can enhance your project's bottom line. Contact us today to discuss how we can support your development goals with reliable, high-quality intermediates delivered with the speed and precision your business demands.
