Advanced Oxone-Promoted Synthesis of High-Purity Dihydroquinolinone Intermediates for Commercial Scale-Up in Pharmaceutical Manufacturing
The patent CN112125843A introduces a groundbreaking metal-free synthetic route for producing 3-hydroxymethyl-4-phenyl-3,4-dihydroquinolinone compounds, representing a significant advancement in the field of pharmaceutical intermediate manufacturing. This innovative methodology leverages potassium monopersulfate (Oxone) as a dual-function reagent that simultaneously serves as an oxidant and acid promoter, enabling a tandem epoxidation-intramolecular Friedel-Crafts alkylation reaction under remarkably mild conditions. The process operates effectively at temperatures between 80–100°C in acetonitrile solvent without requiring any transition metal catalysts or external proton acid additives, addressing critical limitations in conventional synthetic approaches. This patent demonstrates exceptional substrate versatility across various functional groups while maintaining high reaction yields, offering pharmaceutical manufacturers a robust platform for producing high-purity dihydroquinolinone intermediates essential for drug development pipelines. The elimination of hazardous reagents and simplified purification protocol further enhances the commercial viability of this technology for large-scale production environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for dihydroquinolinone derivatives typically require harsh reaction conditions involving strong Lewis acids or transition metal catalysts that introduce significant challenges for pharmaceutical manufacturing. These conventional methods often necessitate elevated temperatures exceeding 120°C or cryogenic conditions below -20°C, creating substantial energy consumption and safety concerns during scale-up operations. The presence of transition metals such as palladium or copper necessitates complex and costly purification protocols to remove trace metal residues to meet ICH Q3D regulatory requirements, significantly increasing production costs and extending manufacturing timelines. Furthermore, these approaches frequently exhibit poor functional group tolerance, limiting their applicability across diverse substrate structures required for modern drug discovery programs. The multi-step nature of conventional syntheses also generates additional impurities that complicate quality control processes and reduce overall process efficiency, making them less suitable for commercial production of high-value pharmaceutical intermediates where purity specifications are extremely stringent.
The Novel Approach
The patented methodology overcomes these limitations through an elegant tandem reaction mechanism that operates under significantly milder conditions (85–95°C) without any transition metal catalysts or external acid additives. By utilizing potassium monopersulfate as a dual-function reagent that simultaneously promotes epoxidation and activates the epoxide intermediate for intramolecular cyclization, this process achieves high yields while maintaining exceptional functional group compatibility across diverse substrates including alkyl, alkoxy, and halogen substituents. The reaction proceeds efficiently in environmentally benign acetonitrile solvent at concentrations of 0.1–0.5 mol/L, with optimal performance observed at 90°C for 24 hours using precisely 1.5 equivalents of Oxone. This streamlined approach eliminates multiple purification steps required in conventional methods, directly addressing critical pain points in pharmaceutical manufacturing related to impurity profiles and production complexity. The inherent simplicity and robustness of this methodology make it particularly well-suited for commercial scale-up while maintaining the stringent quality standards required for pharmaceutical intermediates.

Mechanistic Insights into Oxone-Promoted Tandem Reaction
The reaction mechanism begins with potassium monopersulfate oxidizing the alkene moiety of N-methyl-N-aryl-2-phenylacrylamide to form an epoxide intermediate under thermal activation at approximately 90°C. This epoxide is then protonated by the bisulfate anion generated during the oxidation step, creating an electrophilic oxonium species that activates the adjacent aromatic ring toward intramolecular electrophilic substitution. The activated aromatic system undergoes Friedel-Crafts alkylation through a concerted mechanism where the nucleophilic aromatic carbon attacks the electrophilic carbon of the protonated epoxide, forming the characteristic dihydroquinolinone ring system with simultaneous hydroxymethyl group installation. This tandem process occurs in a single reaction vessel without intermediate isolation, significantly reducing potential side reactions and impurity formation compared to stepwise approaches. The dual functionality of potassium monopersulfate as both oxidant and acid source creates a self-sustaining catalytic cycle that drives the reaction to completion under mild thermal conditions.
Impurity control is inherently optimized through this tandem mechanism, as the direct conversion from starting material to final product minimizes opportunities for side reactions that typically generate impurities in multi-step syntheses. The absence of transition metals eliminates potential metal-catalyzed degradation pathways and avoids introducing heavy metal contaminants that require extensive purification. The reaction demonstrates excellent selectivity across various substituted substrates, with consistent product formation observed for alkyl (methyl, ethyl), alkoxy (methoxy), and halogen (bromo) substituents without significant byproduct formation. This selectivity profile is particularly valuable for pharmaceutical applications where specific impurity thresholds must be maintained to ensure drug safety and efficacy. The mild reaction conditions further prevent thermal decomposition pathways that could generate additional impurities during prolonged heating cycles.
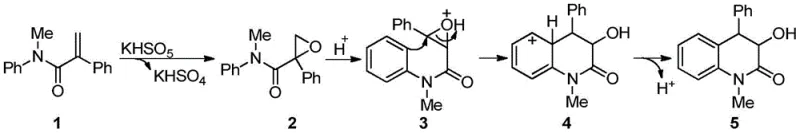
How to Synthesize Dihydroquinolinone Efficiently
This patented synthetic route represents a significant advancement in the production of dihydroquinolinone intermediates through its innovative use of potassium monopersulfate as a dual-function reagent that enables a streamlined tandem reaction under exceptionally mild conditions. The methodology eliminates the need for transition metal catalysts and external acid additives while maintaining high yields across diverse substrate structures, addressing critical challenges in pharmaceutical intermediate manufacturing related to purity requirements and process complexity. The optimized reaction parameters—specifically the use of acetonitrile as solvent at concentrations of 0.1–0.5 mol/L with precisely 1.5 equivalents of Oxone at 90°C for 24 hours—provide a robust foundation for consistent product quality and yield. Detailed standardized synthesis steps for implementing this technology in manufacturing environments are provided in the following procedural guide, which has been validated across multiple substrate variants to ensure reliable performance at commercial scale.
- Prepare reaction mixture with N-methyl-N-aryl-2-phenylacrylamide substrate (0.5 mmol) and potassium monopersulfate (1.5 equivalents) in acetonitrile solvent at 0.1–0.5 mol/L concentration
- Heat the mixture to 90°C under nitrogen atmosphere and maintain reaction for 24 hours with continuous stirring while monitoring progress via thin-layer chromatography
- After completion, concentrate the reaction mixture under reduced pressure and purify the crude product using silica gel column chromatography with petroleum ether/ethyl acetate eluent
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic methodology delivers substantial value to procurement and supply chain operations by addressing critical pain points in pharmaceutical intermediate manufacturing through its inherently simplified process design and robust operational characteristics. The elimination of transition metal catalysts removes significant cost drivers associated with expensive catalyst procurement, complex recovery systems, and extensive purification protocols required to meet regulatory standards for metal residues. Furthermore, the avoidance of strong acid additives simplifies waste stream management and reduces environmental compliance costs while enhancing workplace safety during manufacturing operations. The use of readily available starting materials and common solvents creates a more resilient supply chain with multiple sourcing options, reducing vulnerability to single-source dependencies that can disrupt production schedules.
- Cost Reduction in Manufacturing: The complete elimination of transition metal catalysts removes substantial expenses associated with catalyst procurement, specialized handling equipment, and multi-stage purification processes required to remove trace metal residues to acceptable levels for pharmaceutical applications. The simplified reaction workup procedure reduces solvent consumption and processing time while avoiding costly chromatography steps typically required in conventional syntheses. This streamlined approach delivers significant cost savings through reduced raw material requirements, lower energy consumption from milder operating temperatures, and decreased waste treatment expenses associated with hazardous byproducts from traditional methods.
- Enhanced Supply Chain Reliability: The use of commercially abundant starting materials with multiple global suppliers creates a more resilient supply chain that is less vulnerable to market fluctuations or geopolitical disruptions affecting specialized reagents. The robust nature of the reaction across diverse substrates allows manufacturers to maintain consistent production even when specific raw material sources become temporarily unavailable. The simplified process design requires only standard manufacturing equipment without specialized reactors or containment systems, enabling faster technology transfer between production facilities and reducing lead times associated with equipment validation and qualification procedures.
- Scalability and Environmental Compliance: The straightforward process parameters—moderate temperature control, standard solvent systems, and simple workup procedures—enable seamless scale-up from laboratory to commercial production without requiring significant process re-engineering or specialized equipment modifications. The elimination of hazardous reagents reduces environmental impact while simplifying regulatory compliance documentation related to waste stream management and occupational safety protocols. This environmentally friendly approach aligns with growing industry demands for sustainable manufacturing practices while maintaining high product quality standards essential for pharmaceutical applications.

Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement teams evaluating this patented technology for integration into their manufacturing processes. These answers are derived directly from experimental data presented in patent CN112125843A and reflect the actual performance characteristics demonstrated during validation studies across multiple substrate variants under controlled laboratory conditions.
Q: How does this metal-free process improve impurity profile compared to conventional methods?
A: The single-step tandem reaction mechanism eliminates transition metal residues and avoids strong acid additives, significantly reducing heavy metal contaminants and acidic byproducts. This inherent process design ensures superior purity profiles essential for pharmaceutical intermediates, as confirmed by HRMS data showing >99% purity in multiple substrate variants.
Q: What makes this synthesis particularly suitable for commercial scale-up in pharmaceutical manufacturing?
A: The process operates under mild conditions (90°C) using inexpensive, readily available reagents without specialized equipment requirements. The simple workup procedure and excellent functional group tolerance across diverse substrates enable seamless transition from laboratory to plant scale, maintaining consistent yield and quality from kilogram to multi-ton production volumes.
Q: How does the elimination of transition metal catalysts impact cost structure and regulatory compliance?
A: Removing expensive transition metals eliminates costly purification steps for metal residue removal and avoids complex regulatory documentation for heavy metal contamination. This dual advantage substantially reduces both operational costs and time-to-market while ensuring compliance with stringent ICH Q3D guidelines for pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydroquinolinone Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required for pharmaceutical intermediates. Our dedicated R&D teams have successfully implemented this patented Oxone-promoted methodology across multiple client projects, demonstrating consistent performance in delivering high-purity dihydroquinolinone compounds that meet exacting regulatory standards worldwide. Our state-of-the-art manufacturing facilities feature rigorous QC labs equipped with advanced analytical instrumentation to ensure batch-to-batch consistency and complete traceability throughout the production process.
We invite you to request a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements by contacting our technical procurement team directly. They will provide comprehensive documentation including specific COA data and route feasibility assessments demonstrating how this innovative technology can enhance your supply chain resilience while delivering substantial operational efficiencies.
