Advanced Synthesis of Remdesivir Intermediate for Scalable API Manufacturing
Advanced Synthesis of Remdesivir Intermediate for Scalable API Manufacturing
The global pharmaceutical landscape has witnessed an unprecedented demand for antiviral therapeutics, placing immense pressure on the supply chains of critical active pharmaceutical ingredients (APIs) and their precursors. Patent CN111205294A, published in May 2020, introduces a groundbreaking preparation method for a key Remdesivir intermediate, specifically (2R,3R,4R,5R)-2-(4-aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-3,4-bis(benzyloxy)-5-((benzyloxy)methyl)tetrahydrofuran-2-carbonitrile. This technical disclosure represents a significant paradigm shift from legacy multi-step syntheses, offering a streamlined, two-step pathway that prioritizes stereochemical control and operational simplicity. For R&D directors and procurement strategists, this innovation addresses the twin challenges of chiral purity and manufacturing cost, providing a robust foundation for securing reliable supplies of high-value nucleoside analogues in a volatile market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Remdesivir and its intermediates has been plagued by inefficient linear sequences that compromise overall yield and escalate production costs. The original route disclosed in patent WO2016069826A1, for instance, relies on a tedious sequence involving oxidation, addition, substitution, resolution, debenzylation, and protection steps before finally achieving the desired substitution. This approach not only consumes excessive amounts of reagents and solvents but also necessitates the use of chiral columns for purification, a technique that is notoriously difficult to scale and economically prohibitive for commercial manufacturing. Furthermore, the late-stage introduction of the chiral center in these conventional pathways often results in poor diastereoselectivity, generating complex mixtures of isomers that require resource-intensive separation protocols, thereby severely impacting the throughput and sustainability of the supply chain.
The Novel Approach
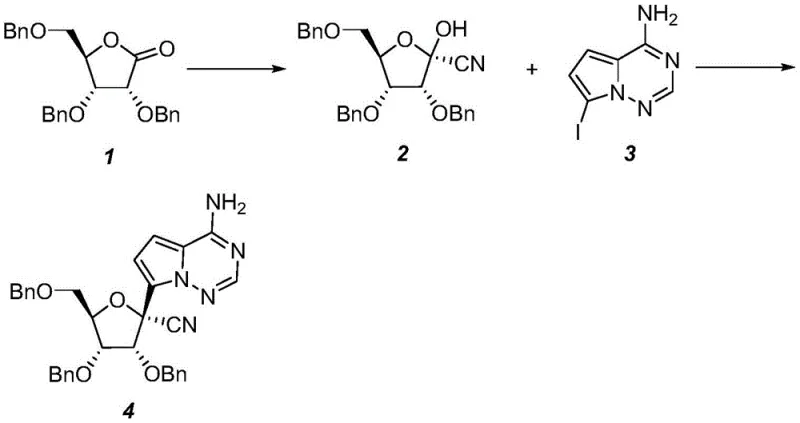
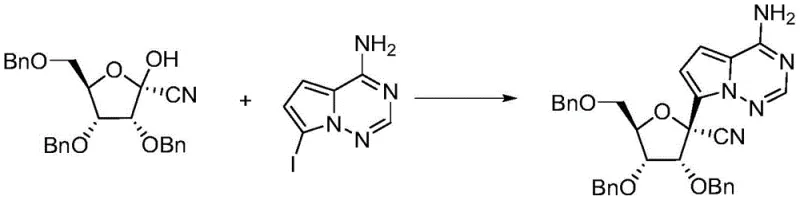
In stark contrast, the methodology outlined in CN111205294A inverts the traditional synthetic logic by establishing the critical chiral nitrile functionality at the very beginning of the sequence. By performing an asymmetric addition on 2,3,5-tribenzyloxy-D-ribono-1,4-lactone first, the process generates a highly pure hydroxy-nitrile intermediate which can be effectively purified via simple recrystallization using solvent systems like ethyl acetate and petroleum ether. This strategic rearrangement of steps eliminates the need for chiral chromatography entirely. The subsequent coupling with the pyrrolo-triazine moiety is then performed on a pre-validated chiral scaffold, ensuring that the final product retains high optical purity. This novel approach drastically reduces the number of unit operations and minimizes the consumption of the expensive heterocyclic coupling partner, delivering substantial economic advantages.
Mechanistic Insights into Asymmetric Cyanation and Substitution
The cornerstone of this improved synthesis lies in the precise control of the asymmetric cyanation step, which dictates the stereochemical outcome of the entire process. The reaction utilizes a Lewis acid catalyst, such as titanium tetrachloride (TiCl4) or aluminum trichloride (AlCl3), to activate the carbonyl group of the ribono-lactone towards nucleophilic attack by a cyano source like trimethylsilyl cyanide (TMSCN). Conducted at controlled temperatures ranging from 0°C to 50°C, this step proceeds through a highly organized transition state that favors the formation of the desired (2R,3R,4R,5R) configuration. The use of specific molar ratios, typically between 1:0.2:1 and 1:2:5 for lactone, Lewis acid, and cyano reagent respectively, ensures complete conversion while suppressing side reactions. Following the reaction, the crude product is subjected to a targeted crystallization process, which acts as a powerful purification engine to remove minor stereoisomers and impurities before the molecule proceeds to the next stage.
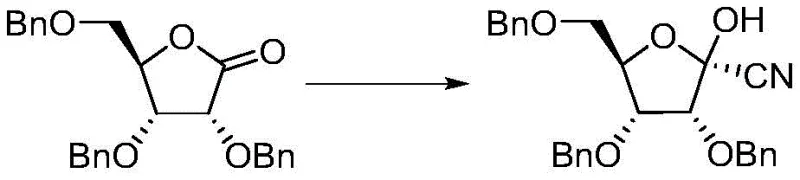
The second phase of the synthesis involves the activation of the newly formed hydroxyl group followed by nucleophilic substitution with 7-iodopyrrolo[2,1-f][1,2,4]triazin-4-amine. The hydroxyl group is first converted into a superior leaving group, such as a mesylate or tosylate, using reagents like methanesulfonyl chloride (MsCl) or p-toluenesulfonyl chloride (TsCl) in the presence of a base like triethylamine or DIPEA. This activation is performed at low temperatures, typically between -10°C and 20°C, to prevent degradation of the sensitive nitrile functionality. The subsequent displacement by the iodide-substituted heterocycle proceeds smoothly in polar aprotic solvents such as tetrahydrofuran (THF) or acetonitrile. This mechanistic pathway ensures that the fragile glycosidic bond is formed with high fidelity, preserving the integrity of the sugar backbone and the attached base, which is critical for the biological activity of the final antiviral agent.
How to Synthesize Remdesivir Intermediate Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly temperature control and stoichiometry, to replicate the high yields reported in the patent examples. The process is designed to be operationally straightforward, utilizing standard laboratory and plant equipment without the need for specialized high-pressure or cryogenic reactors. Operators should focus on maintaining the specified temperature windows during the addition of Lewis acids and activating agents to maximize selectivity. The workup procedures involve standard aqueous quenches and organic extractions, followed by crystallization steps that are crucial for achieving the required purity specifications. For a comprehensive breakdown of the standardized operating procedures and safety protocols required for this synthesis, please refer to the detailed guide below.
- Perform asymmetric addition on 2,3,5-tribenzyloxy-D-ribono-1,4-lactone using a Lewis acid (e.g., TiCl4) and a cyano reagent (e.g., TMSCN) in DCM at 0-10°C to form the hydroxy-nitrile intermediate.
- Activate the hydroxyl group of the intermediate using MsCl or TsCl and a base like triethylamine in THF at low temperature (-10 to 20°C).
- React the activated species with 7-iodopyrrolo[2,1-f][1,2,4]triazin-4-amine to yield the final nitrile intermediate, followed by purification via crystallization or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers transformative benefits for procurement managers and supply chain leaders tasked with optimizing the cost of goods sold (COGS) and ensuring supply continuity. The primary advantage stems from the significant reduction in process complexity; by condensing the synthesis into fewer steps and eliminating the need for chiral column chromatography, the method drastically lowers both capital expenditure on equipment and operational expenditure on consumables. The ability to purify intermediates via crystallization rather than chromatography is a major cost driver, as it allows for processing much larger batches with higher throughput and lower solvent consumption. Furthermore, the early establishment of chirality reduces the risk of batch failure due to isomer contamination, thereby enhancing overall manufacturing reliability.
- Cost Reduction in Manufacturing: The elimination of expensive chiral separation technologies and the reduction in the number of synthetic steps directly translate to lower production costs. By forming the chiral center early, the process avoids the loss of valuable advanced intermediates that often occurs during late-stage resolutions in traditional routes. Additionally, the use of commodity chemicals like TiCl4 and TMSCN, rather than exotic catalysts, ensures that raw material costs remain stable and predictable. This efficiency allows manufacturers to offer competitive pricing for high-purity pharmaceutical intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials, such as 2,3,5-tribenzyloxy-D-ribono-1,4-lactone, mitigates the risk of supply bottlenecks associated with specialized reagents. The robustness of the reaction conditions, which tolerate standard industrial solvents like DCM and THF, means that production can be easily transferred between different manufacturing sites or scaled up rapidly to meet surging demand. This flexibility is crucial for maintaining a steady flow of materials to downstream API producers, ensuring that drug development timelines are not delayed by intermediate shortages.
- Scalability and Environmental Compliance: The simplified workup procedures, which rely on filtration and crystallization rather than complex distillations or chromatographic separations, generate less hazardous waste and reduce the environmental footprint of the manufacturing process. This aligns with increasingly stringent global regulations regarding solvent emissions and waste disposal. The process is inherently scalable, capable of transitioning seamlessly from pilot plant quantities to multi-ton commercial production, making it an ideal candidate for long-term supply agreements with major pharmaceutical companies seeking sustainable manufacturing partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and beneficial effects described in patent CN111205294A, providing clarity on the practical aspects of adopting this technology. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing supply chains.
Q: How does this new route improve chiral purity compared to traditional methods?
A: By introducing the cyano group early in the synthesis (Step 1) rather than late, the method establishes the chiral center prior to the bulky coupling step. This allows for effective purification via recrystallization (e.g., Ethyl Acetate/Petroleum Ether) before the complex heterocycle is attached, avoiding the need for expensive chiral column separation required in older routes.
Q: What are the critical reaction conditions for the cyanation step?
A: The asymmetric addition requires strict temperature control between 0°C and 50°C, preferably 0-10°C, using Lewis acids like Titanium Tetrachloride or Aluminum Trichloride. The molar ratio of lactone to Lewis acid to cyano reagent is optimized around 1:0.2:1 to 1:2:5 to maximize yield and stereoselectivity.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes common industrial solvents (DCM, THF, Acetonitrile) and avoids cryogenic conditions or exotic catalysts. The workup involves standard aqueous quenching and crystallization, making it highly amenable to scale-up from kilogram to multi-ton production without significant engineering hurdles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Remdesivir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving antiviral therapies depends on a secure and high-quality supply of critical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the global pharmaceutical market. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our expertise in nucleoside chemistry allows us to navigate the complexities of chiral synthesis, guaranteeing that the intermediates we supply are perfectly suited for downstream API manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume and quality requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this more efficient pathway. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive antiviral market.
