Innovative Metal-Free Triazole Synthesis: Scaling Pharmaceutical Intermediates from Lab to Commercial Production
The patent CN113105402A introduces a groundbreaking methodology for synthesizing trifluoromethyl-containing triazole intermediates critical to modern pharmaceutical development. This innovation addresses longstanding industry challenges in heterocyclic chemistry by establishing a robust metal-free pathway that eliminates conventional constraints such as anhydrous conditions and heavy metal catalysts. The process leverages iodine-promoted Kornblum oxidation in dimethyl sulfoxide to generate key aryl diketone intermediates before cyclization with trifluoroethylimine hydrazides. Notably, this approach achieves high functional group tolerance across diverse aryl substitutions while maintaining operational simplicity that facilitates seamless transition from laboratory discovery to commercial manufacturing environments. The strategic incorporation of trifluoromethyl groups enhances physicochemical properties like metabolic stability and lipophilicity—attributes essential for next-generation drug candidates including antiviral and antidiabetic therapeutics. This patent represents a significant advancement in sustainable heterocyclic synthesis with direct implications for accelerating API development timelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional syntheses of polysubstituted triazoles frequently require stringent anhydrous and oxygen-free conditions that complicate large-scale implementation due to specialized equipment needs and increased operational costs. Many established routes depend on transition metal catalysts like copper or palladium systems which introduce significant purification challenges related to heavy metal residues that must be reduced below parts-per-million levels for pharmaceutical applications. These methods often exhibit narrow substrate scope with poor tolerance for electron-donating or withdrawing functional groups on aromatic rings—limiting their utility for complex API intermediates requiring specific substitution patterns. Additionally, conventional cyclization techniques typically demand multi-step sequences with intermediate isolations that reduce overall atom economy while increasing solvent consumption and waste generation. The reliance on expensive ligands and sensitive reagents further elevates production costs while creating supply chain vulnerabilities through single-source dependencies on specialized chemical suppliers.
The Novel Approach
The patented methodology overcomes these limitations through an elegant iodine-catalyzed cascade reaction that operates under ambient atmospheric conditions without requiring inert gas protection or moisture control. By utilizing elemental iodine as a sustainable catalyst in dimethyl sulfoxide solvent system—both commercially available at low cost—the process eliminates all transition metal dependencies while maintaining high regioselectivity across diverse substrates. The reaction sequence integrates Kornblum oxidation with tandem cyclization in a single reaction vessel through carefully optimized temperature staging between two distinct thermal regimes. This design achieves exceptional functional group tolerance where substituents like methyl, methoxy, chlorine or trifluoromethyl groups on aryl rings do not compromise yield or purity. Crucially, the methodology demonstrates inherent scalability from gram-scale validation to pilot plant quantities without reoptimization—providing immediate transferability to manufacturing environments while reducing process development timelines by eliminating complex catalyst recovery steps.
Mechanistic Insights into Iodine-Promoted Triazole Cyclization
The reaction mechanism initiates with iodine-mediated oxidation of arylethanone in DMSO to form α-halo ketone intermediates that rapidly hydrolyze into aryl diketones under thermal conditions. This Kornblum oxidation step occurs efficiently at moderate temperatures between 90°C and 110°C without requiring additional oxidants or catalysts beyond elemental iodine. The generated diketone then undergoes condensation with trifluoroethylimine hydrazide through nucleophilic attack at the carbonyl carbon—forming a hydrazone intermediate that subsequently cyclizes via intramolecular nucleophilic substitution. The second thermal phase at elevated temperatures (110°C–130°C) facilitates ring closure where pyridine acts as a mild base while sodium dihydrogen phosphate modulates reaction kinetics through buffer effects. Iodine plays a dual role throughout this cascade—first promoting oxidation then facilitating dehydrogenation during cyclization—without being consumed in stoichiometric quantities.
Impurity control is achieved through precise stoichiometric balancing of iodine relative to sodium dihydrogen phosphate and pyridine at ratios of 2.5:4:1 equivalents respectively. This optimization minimizes side reactions such as over-halogenation or hydrolysis by maintaining controlled redox potential throughout both reaction phases. The absence of transition metals eliminates common impurities like palladium black or copper residues that typically require extensive chromatographic purification in conventional routes. Furthermore, the solvent system's polarity profile selectively dissolves target intermediates while precipitating byproducts during cooling—enabling straightforward filtration-based separation before final chromatographic polishing. This inherent selectivity produces compounds with consistently high purity profiles suitable for direct use in subsequent API manufacturing steps without additional recrystallization requirements.
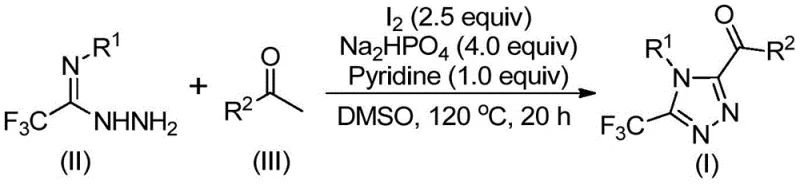
How to Synthesize Trifluoromethyl Triazole Intermediates Efficiently
This innovative synthesis route represents a paradigm shift in heterocyclic chemistry by replacing traditional metal-catalyzed methods with a sustainable iodine-mediated cascade process that maintains high efficiency across diverse substrate combinations. The methodology demonstrates exceptional robustness through its tolerance of various functional groups including halogens and electron-donating moieties on aromatic rings—enabling customization for specific pharmaceutical applications without process revalidation. Detailed standardized synthesis steps are provided below to facilitate immediate implementation in R&D laboratories seeking reliable production of high-purity triazole intermediates.
- Dissolve arylethanone and elemental iodine in dimethyl sulfoxide (DMSO) and heat the mixture to 90-110°C for 4-6 hours to form the aryl diketone intermediate via iodination/Kornblum oxidation.
- Add trifluoroethylimine hydrazide, sodium dihydrogen phosphate, pyridine, and additional iodine to the reaction mixture, then heat to 110-130°C for 12-20 hours to facilitate tandem cyclization and form the triazole core.
- Perform post-treatment by filtering the reaction mixture, mixing with silica gel, and purifying via column chromatography to obtain high-purity 3,4,5-trisubstituted 1,2,4-triazole compounds.
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial value across procurement and supply chain functions by addressing critical pain points associated with traditional triazole synthesis routes. The elimination of transition metal catalysts removes significant cost drivers related to catalyst procurement and specialized waste treatment protocols while enhancing supply chain resilience through reduced dependency on volatile precious metal markets. Furthermore, the use of commercially available starting materials from multiple global suppliers mitigates single-source risks that commonly disrupt intermediate production schedules in pharmaceutical manufacturing.
- Cost Reduction in Manufacturing: The complete avoidance of expensive transition metal catalysts eliminates both procurement costs and downstream purification expenses associated with metal residue removal—creating substantial cost savings through simplified process streams and reduced analytical testing requirements. The utilization of inexpensive reagents like elemental iodine and DMSO further optimizes raw material expenditures while maintaining high yields across diverse substrate combinations without requiring specialized equipment investments.
- Enhanced Supply Chain Reliability: Sourcing flexibility is significantly improved through reliance on widely available starting materials that can be procured from numerous global suppliers without long lead times or geopolitical constraints. The ambient condition operation removes dependencies on specialized infrastructure like gloveboxes or cryogenic systems—enabling rapid deployment across existing manufacturing facilities worldwide while minimizing facility qualification timelines for new production sites.
- Scalability and Environmental Compliance: The inherent scalability from gram-scale validation directly to multi-ton production eliminates time-consuming reoptimization phases typically required when transitioning between development stages. The process generates minimal hazardous waste streams due to its metal-free nature while utilizing recyclable solvents—aligning with increasingly stringent environmental regulations without requiring additional capital investments in waste treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement teams evaluating this technology for integration into their supply chains. Each response is derived directly from experimental data within patent CN113105402A regarding process robustness and commercial viability.
Q: How does this method eliminate the need for anhydrous and oxygen-free conditions compared to traditional triazole syntheses?
A: The iodine/DMSO system enables Kornblum oxidation under ambient conditions without moisture or oxygen sensitivity. Unlike conventional metal-catalyzed routes requiring rigorous inert atmospheres, this method leverages DMSO's dual role as solvent and oxidant precursor while maintaining stability across diverse substrates.
Q: What are the environmental and cost benefits of avoiding heavy metal catalysts in this synthesis?
A: Eliminating transition metals removes costly purification steps for metal residue removal and reduces hazardous waste generation. This directly lowers EHS compliance costs while aligning with green chemistry principles through simplified waste streams and reduced catalyst procurement expenses.
Q: How does substrate flexibility support scalable production of diverse pharmaceutical intermediates?
A: The broad functional group tolerance accommodates varied aryl substitutions at R¹ and R² positions without yield penalties. This design flexibility allows rapid adaptation to specific API requirements while maintaining consistent scalability from gram-scale validation to metric ton production runs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazole Supplier
Our patented methodology represents a significant advancement in sustainable heterocyclic synthesis with direct implications for accelerating API development timelines while maintaining stringent quality standards required by global regulatory bodies. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production using rigorously validated processes that consistently meet stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical instrumentation for comprehensive impurity profiling.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team which will provide specific COA data and route feasibility assessments tailored to your unique manufacturing requirements—enabling data-driven decisions about integrating this innovative technology into your supply chain infrastructure.
