Advanced Nickel-Catalyzed Carbonylation for High-Purity Thioester Intermediates
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing complex molecular scaffolds, particularly those containing sulfur functionalities which are pivotal in drug design. Patent CN112239384B, published in May 2023, introduces a groundbreaking preparation method for thioester compounds that addresses long-standing challenges in synthetic efficiency and safety. This innovation utilizes a transition metal-catalyzed carbonylation strategy, specifically leveraging a nickel-based catalytic system to couple arylboronic acids with sulfonyl chlorides. Unlike traditional approaches that rely on volatile and malodorous thiols, this novel pathway employs stable and commercially available sulfonyl chlorides as the sulfur source. The significance of this development cannot be overstated for manufacturers of active pharmaceutical ingredients (APIs) and agrochemical intermediates, as thioester motifs are prevalent in bioactive molecules such as Timobesone and Spironolactone. By establishing a reliable protocol that operates under relatively mild conditions without the need for external oxidants or reductants, this technology offers a streamlined route for producing high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioester compounds has predominantly relied on the direct acylation of thiols with carboxylic acids or their activated derivatives. While chemically straightforward, this classical approach presents severe logistical and environmental drawbacks for industrial scale-up. Thiols are notoriously difficult to handle due to their extremely unpleasant and pervasive odors, which pose significant occupational health risks and require specialized containment infrastructure. Furthermore, the sulfur atoms in free thiols have a high affinity for transition metals, often leading to catalyst poisoning in metal-mediated processes, thereby reducing turnover numbers and increasing production costs. Alternative strategies involving transition metal-catalyzed carbonylation have emerged, yet many still depend on thiols as the sulfur source, inheriting the same toxicity and stability issues. Additionally, some carbonylation protocols require the use of high-pressure carbon monoxide gas, introducing substantial safety hazards related to gas storage and leakage, which complicates the regulatory compliance and insurance aspects of chemical manufacturing facilities.
The Novel Approach
The methodology disclosed in CN112239384B represents a paradigm shift by replacing problematic thiols with sulfonyl chlorides, which are odorless, stable, and widely accessible commodity chemicals. This new route utilizes a sophisticated catalytic system comprising nickel triflate, a bipyridine ligand, and molybdenum hexacarbonyl to facilitate the coupling reaction. The use of sulfonyl chlorides not only mitigates the odor and toxicity concerns but also enhances the designability of the substrate, allowing for a broader scope of functional group compatibility. Crucially, the reaction proceeds efficiently at 120°C in polar aprotic solvents like N-methylpyrrolidone (NMP), achieving high yields without the necessity for external oxidants or reductants. This simplification of the reagent profile translates directly into cost reduction in API manufacturing, as it reduces the number of unit operations and waste streams generated during the process. The ability to synthesize diverse thioester structures from readily available starting materials makes this method highly attractive for a reliable pharmaceutical intermediate supplier aiming to optimize their production portfolio.
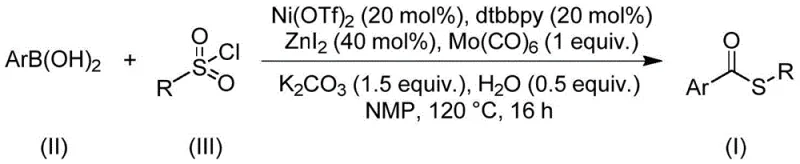
Mechanistic Insights into Nickel-Catalyzed Carbonylation
The core of this innovative synthesis lies in the intricate interplay between the nickel catalyst and the molybdenum carbonyl source. The reaction initiates with the activation of the nickel precatalyst, likely Ni(OTf)2, by the dtbbpy ligand to form an active low-valent nickel species. Simultaneously, molybdenum hexacarbonyl (Mo(CO)6) acts as a solid-state carbon monoxide reservoir, releasing CO in situ under thermal conditions. This controlled release of CO is critical for maintaining a steady concentration of the carbonylating agent without the risks associated with gaseous CO handling. The arylboronic acid undergoes transmetallation with the nickel center, while the sulfonyl chloride serves as the electrophilic sulfur source, potentially undergoing desulfitative insertion or reductive elimination pathways to forge the C-S bond. The presence of zinc iodide and water as additives plays a synergistic role, likely facilitating the activation of the sulfonyl chloride or stabilizing key intermediates within the catalytic cycle. This mechanistic elegance ensures that the reaction proceeds with high atom economy and selectivity, minimizing the formation of side products such as biaryls or homocoupling impurities that often plague cross-coupling reactions.
From an impurity control perspective, the absence of external oxidants or reductants is a significant advantage. In many redox-neutral or oxidative couplings, over-oxidation of the sulfur moiety to sulfones or sulfoxides is a common degradation pathway that complicates purification. By carefully tuning the redox potential of the nickel catalyst through ligand selection and additive optimization, this method effectively suppresses such over-oxidation side reactions. Furthermore, the use of arylboronic acids, which are generally stable and easy to purify, contributes to a cleaner reaction profile compared to organometallic reagents like Grignard or organolithium species that are sensitive to moisture and air. The resulting crude reaction mixtures are amenable to standard workup procedures, such as filtration and silica gel chromatography, yielding thioester products with high purity specifications. This level of control over the impurity profile is essential for meeting the stringent quality standards required for clinical-grade pharmaceutical intermediates.
How to Synthesize Thioester Compounds Efficiently
Implementing this synthesis route requires precise adherence to the optimized reaction parameters to ensure maximum yield and reproducibility. The process begins with the careful weighing and charging of the catalytic components, including nickel triflate, the bipyridine ligand, and the carbonyl source, into a suitable reaction vessel capable of withstanding elevated temperatures. The choice of solvent is critical, with N-methylpyrrolidone (NMP) identified as the optimal medium due to its ability to dissolve all reactants and stabilize the catalytic species effectively. Once the reaction mixture is homogenized, the substrates—arylboronic acid and sulfonyl chloride—are introduced, followed by the addition of inorganic bases and promoters. The detailed standardized synthesis steps for this process are outlined in the guide below.
- Charge a reaction vessel with nickel triflate catalyst, dtbbpy ligand, molybdenum hexacarbonyl, zinc iodide, potassium carbonate, and water in an organic solvent like NMP.
- Add the arylboronic acid and sulfonyl chloride substrates to the mixture under stirring to ensure homogeneous dissolution and interaction.
- Heat the reaction mixture to 120°C for 16 hours, then perform filtration and column chromatography to isolate the pure thioester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers tangible strategic benefits beyond mere chemical novelty. The shift from thiols to sulfonyl chlorides fundamentally alters the cost structure and risk profile of thioester production. By eliminating the need for specialized odor-control scrubbing systems and hazardous gas handling infrastructure, capital expenditure (CAPEX) for new production lines can be significantly reduced. Moreover, the reliance on commodity chemicals like arylboronic acids and sulfonyl chlorides ensures a stable and diversified supply base, reducing the risk of raw material shortages that often plague niche reagent markets. This stability is crucial for maintaining continuous manufacturing operations and meeting tight delivery schedules for downstream clients in the pharmaceutical sector.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive and abundant starting materials. Sulfonyl chlorides are produced on a massive industrial scale for various applications, ensuring competitive pricing and long-term availability. Furthermore, the catalytic system, while utilizing nickel, operates with high efficiency, minimizing the loading of precious metals or expensive ligands required per kilogram of product. The simplified workup procedure, which avoids complex extraction or distillation steps often needed to remove volatile thiols, further lowers operational expenditures (OPEX) by reducing energy consumption and labor hours. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain or reinvested into R&D initiatives.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the commercial availability of reagents. Unlike methods requiring custom-synthesized organometallic reagents or unstable intermediates, the inputs for this process are shelf-stable and can be sourced from multiple global suppliers. This redundancy mitigates the risk of single-source dependency and allows for flexible inventory management strategies. Additionally, the reaction's tolerance to various functional groups means that a single platform technology can be adapted to produce a wide range of derivatives, allowing manufacturers to respond quickly to changing market demands without retooling entire production lines.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard batch reactors and the absence of high-pressure gas feeds. The solid nature of the CO source (Mo(CO)6) simplifies dosing and control, making the transition to larger vessels more predictable and safer. From an environmental standpoint, the method aligns with green chemistry principles by avoiding toxic thiols and minimizing waste generation. The reduced hazard profile simplifies waste disposal and regulatory reporting, ensuring compliance with increasingly stringent environmental regulations. This sustainability angle is increasingly important for corporate social responsibility (CSR) goals and can enhance the marketability of the final products to eco-conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thioester synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating the feasibility of this route for their specific projects. Understanding these nuances is key to making informed decisions about process adoption and resource allocation.
Q: Why is sulfonyl chloride preferred over thiols in this synthesis?
A: Traditional thiol-based methods often suffer from unpleasant odors and catalyst poisoning issues. Using sulfonyl chlorides as the sulfur source eliminates these handling hazards and improves catalyst longevity, making the process more suitable for large-scale industrial applications.
Q: What is the specific role of Molybdenum Hexacarbonyl in this reaction?
A: Molybdenum hexacarbonyl (Mo(CO)6) serves as a solid, safe, and controllable source of carbon monoxide (CO) in situ. This avoids the need for handling hazardous high-pressure CO gas cylinders, significantly enhancing operational safety and simplifying the equipment requirements for the carbonylation step.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the patented protocol demonstrates excellent functional group tolerance. It successfully accommodates various substituents such as alkyl, alkoxy, and halogen groups on both the arylboronic acid and the sulfonyl chloride, allowing for the synthesis of a wide library of structurally diverse thioester intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN112239384B for the future of fine chemical synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle the specific requirements of nickel-catalyzed reactions, including rigorous QC labs that enforce stringent purity specifications to guarantee the quality of every batch. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of the global healthcare industry.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this route can optimize your budget and timeline. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the market with reliable, high-quality thioester solutions.
