Advanced Synthesis of Axial Chiral Naphthalene-Pyrrole Phosphine Catalysts for Industrial Asymmetric Catalysis
The pharmaceutical and fine chemical industries are constantly seeking novel chiral ligands that can unlock new reactivity profiles and improve stereoselectivity in asymmetric synthesis. Patent CN114029086B introduces a groundbreaking class of axial chiral naphthalene-pyrrole phosphine catalysts, represented by Formula 6, which address the limitations of traditional carbon-centered chiral phosphines. Unlike conventional ligands that rely on point chirality, these catalysts leverage axial chirality derived from a rigid naphthalene-pyrrole scaffold, offering superior steric control in catalytic cycles. 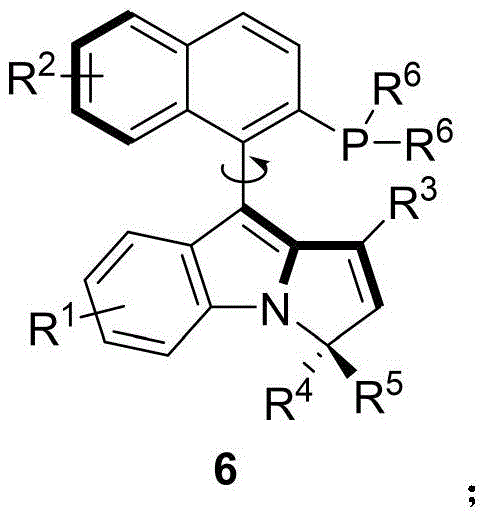 . This innovation is particularly significant for the production of high-purity pharmaceutical intermediates, where precise stereochemical outcomes are critical for drug efficacy and safety. The technology provides a robust platform for developing reliable specialty chemical suppliers capable of delivering complex chiral building blocks.
. This innovation is particularly significant for the production of high-purity pharmaceutical intermediates, where precise stereochemical outcomes are critical for drug efficacy and safety. The technology provides a robust platform for developing reliable specialty chemical suppliers capable of delivering complex chiral building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing chiral phosphine catalysts often rely on the resolution of racemic mixtures or the use of chiral pool starting materials, which can be costly and limit structural diversity. Furthermore, many existing axial chiral ligands, such as BINAP derivatives, are restricted to biaryl scaffolds that may not provide the optimal electronic or steric environment for certain challenging transformations. The synthesis of these traditional ligands frequently involves harsh reaction conditions, including high temperatures and strong bases, which can lead to racemization or decomposition of sensitive functional groups. Additionally, the reliance on precious metal catalysts in early stages without efficient recovery systems can drastically increase the cost reduction in fine chemical manufacturing, making them less attractive for large-scale industrial applications where margin compression is a constant pressure.
The Novel Approach
The patented methodology presents a paradigm shift by utilizing a chiral phosphoric acid-catalyzed assembly strategy to construct the axial chiral backbone under remarkably mild conditions. By reacting a naphthalene-indole derivative with a propargylic alcohol in the presence of a chiral phosphoric acid catalyst at temperatures ranging from 0°C to 40°C, the process achieves high enantioselectivity without the need for cryogenic conditions. This approach not only simplifies the operational complexity but also expands the substrate scope to include diverse aryl and heteroaryl groups. The subsequent functionalization steps, including triflation and palladium-catalyzed phosphorylation, are optimized to maintain the integrity of the chiral axis while introducing the phosphine moiety efficiently. This streamlined route ensures that the commercial scale-up of complex chiral ligands is feasible, offering a sustainable alternative to legacy synthesis pathways that often suffer from low yields and poor atom economy.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Assembly
The core of this synthesis lies in the initial enantioselective construction of the axial chirality, driven by the chiral phosphoric acid catalyst. This organocatalyst activates the propargylic alcohol through hydrogen bonding interactions, facilitating a nucleophilic attack by the electron-rich indole ring of the naphthalene derivative. The rigid pocket created by the bulky substituents on the phosphoric acid (such as 2-naphthyl or 4-chlorophenyl groups) dictates the trajectory of the incoming nucleophile, thereby establishing the axial configuration with high fidelity. Following this key step, the hydroxyl group is converted into a triflate leaving group using trifluoromethanesulfonic anhydride and DIPEA at 0°C, preparing the molecule for C-P bond formation. The subsequent palladium-catalyzed coupling with diphenylphosphine oxide in DMSO at 120°C utilizes DPPB as a bidentate ligand to stabilize the Pd(0) species, ensuring efficient oxidative addition and reductive elimination cycles. 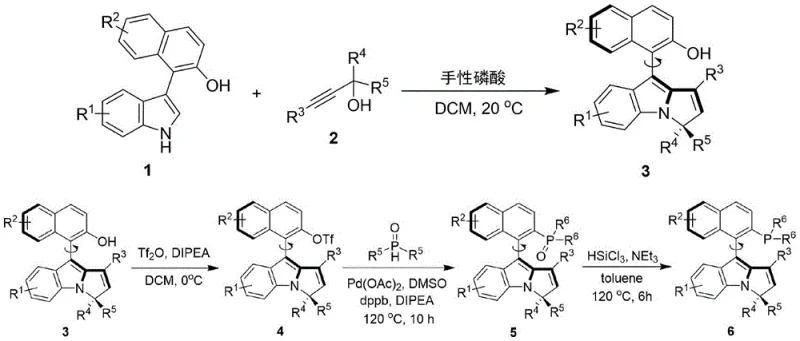 . Finally, the phosphine oxide is reduced to the active phosphine using trichlorosilane and triethylamine in toluene, completing the catalytic cycle preparation.
. Finally, the phosphine oxide is reduced to the active phosphine using trichlorosilane and triethylamine in toluene, completing the catalytic cycle preparation.
Impurity control is meticulously managed throughout this multi-step sequence to ensure the final catalyst meets stringent purity specifications required for GMP manufacturing. The mild conditions of the initial organocatalytic step minimize the formation of side products associated with thermal degradation or non-selective background reactions. In the palladium-catalyzed step, the use of specific ligands like DPPB helps suppress homocoupling of the phosphine oxide, a common impurity in P-C bond formations. Furthermore, the final reduction step is conducted under inert atmosphere to prevent oxidation of the sensitive phosphine product back to the oxide. Rigorous purification protocols, including silica gel column chromatography with optimized eluent systems (e.g., petroleum ether/ethyl acetate gradients), are employed at each stage to remove residual metals, organic byproducts, and unreacted starting materials. This comprehensive approach to impurity profiling guarantees that the resulting axial chiral naphthalene-pyrrole phosphine catalyst possesses the high optical purity necessary for inducing asymmetry in downstream pharmaceutical syntheses.
How to Synthesize Axial Chiral Naphthalene-Pyrrole Phosphine Catalyst Efficiently
The synthesis of this advanced catalyst involves a logical four-step sequence that balances reactivity with stereochemical control, making it accessible for both laboratory research and pilot plant operations. The process begins with the chiral phosphoric acid catalyzed coupling, followed by activation, phosphorylation, and final reduction. Each step has been optimized for yield and selectivity, utilizing common laboratory solvents and reagents that are readily available in the global supply chain. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which outlines the critical process parameters for each transformation.
- React naphthalene-indole derivative with propargylic alcohol using chiral phosphoric acid catalyst in DCM at 0-40°C to form the axial chiral intermediate.
- Convert the hydroxyl group of the intermediate to a triflate using trifluoromethanesulfonic anhydride and DIPEA at 0°C.
- Perform Pd-catalyzed C-P bond formation with diphenylphosphine oxide in DMSO at 120°C using DPPB as ligand.
- Reduce the phosphine oxide to the final phosphine catalyst using trichlorosilane and triethylamine in toluene at 120°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel catalyst synthesis route offers substantial strategic benefits regarding cost stability and supply continuity. The reliance on economically obtained raw materials, such as substituted naphthalenes and propargylic alcohols, mitigates the risk of price volatility often associated with exotic chiral pool reagents. Moreover, the elimination of extreme reaction conditions, such as cryogenic temperatures or high-pressure hydrogenation, translates to significantly reduced energy consumption and lower capital expenditure requirements for reactor infrastructure. The high yields reported in the patent examples indicate a robust process that maximizes material throughput, thereby reducing the overall cost of goods sold (COGS) for the final catalyst. This efficiency is crucial for maintaining competitive pricing in the market for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The synthetic pathway avoids the use of stoichiometric amounts of expensive chiral auxiliaries that need to be attached and removed, instead employing a catalytic amount of chiral phosphoric acid which drives the stereochemistry. This catalytic nature inherently lowers the material cost per kilogram of product. Additionally, the final reduction step uses trichlorosilane, a cost-effective reductant compared to more expensive hydride sources, further contributing to substantial cost savings. The high enantiomeric excess achieved directly reduces the burden on downstream purification processes like chiral HPLC or recrystallization, which are typically the most expensive unit operations in chiral manufacturing.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are commodity chemicals or easily synthesized derivatives, ensuring a stable and diversified supply base that is not dependent on single-source natural extracts. The reaction conditions are compatible with standard stainless steel reactors, allowing for seamless technology transfer from R&D to commercial production facilities without the need for specialized glass-lined or Hastelloy equipment. This compatibility reduces lead time for high-purity chiral ligands by accelerating the scale-up timeline and minimizing the risk of campaign delays due to equipment availability or compatibility issues.
- Scalability and Environmental Compliance: The process utilizes solvents like dichloromethane, toluene, and DMSO, which have well-established recovery and recycling protocols in modern chemical plants, supporting green chemistry initiatives. The absence of heavy metal waste in the initial organocatalytic step simplifies effluent treatment, while the palladium used in the coupling step can be recovered using standard scavengers, minimizing environmental impact. The mild reaction temperatures reduce the thermal load on cooling systems, enhancing the overall energy efficiency of the plant and aligning with corporate sustainability goals for reducing carbon footprint in specialty chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and sourcing of this axial chiral catalyst technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and performance characteristics of the ligand in industrial settings. Understanding these details is essential for R&D teams evaluating this catalyst for new process development and for procurement teams assessing its long-term viability.
Q: What are the key advantages of this axial chiral catalyst over traditional biaryl phosphines?
A: This naphthalene-pyrrole scaffold offers a distinct steric environment compared to traditional BINAP-type ligands, enabling unique selectivity in asymmetric hydrosilylation and coupling reactions. The synthesis utilizes mild chiral phosphoric acid catalysis, avoiding harsh conditions often required for atroposelective biaryl formation.
Q: Is the synthesis scalable for industrial production?
A: Yes, the process employs commercially available raw materials and standard solvents like DCM, DMSO, and toluene. The reaction conditions are mild (0-120°C) and do not require extreme pressures, making it highly suitable for commercial scale-up of complex chiral ligands.
Q: What level of enantiomeric excess (ee) can be achieved?
A: The patented method demonstrates high stereocontrol, achieving enantiomeric excess values up to 96% in various examples. This high optical purity minimizes the need for extensive downstream purification, significantly enhancing process efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Phosphine Catalyst Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chiral technology, leveraging deep expertise in asymmetric synthesis to deliver cutting-edge solutions like the axial chiral naphthalene-pyrrole phosphine catalyst. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and risk-free. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral analysis capabilities to guarantee that every batch meets the exacting standards required for API synthesis. Our commitment to quality ensures that the high enantiomeric excess demonstrated in the patent is consistently reproduced in our commercial batches.
We invite you to collaborate with us to explore how this novel catalyst can enhance your asymmetric synthesis workflows and drive down your overall production costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals and accelerate your time to market for critical pharmaceutical intermediates.
