Advanced Catalytic Curtius Rearrangement for Efficient Amine and Urea Manufacturing
Introduction to Novel Catalytic C-N Bond Construction
The construction of carbon-nitrogen bonds remains a cornerstone of modern organic synthesis, particularly for the development of active pharmaceutical ingredients (APIs) and agrochemicals. While traditional transition metal-catalyzed couplings like Buchwald-Hartwig amination have revolutionized arylamine synthesis, they often struggle with aliphatic substrates or require harsh conditions. Patent CN112028814B introduces a groundbreaking methodology that leverages a novel catalytic Curtius rearrangement to efficiently generate alkyl, alkenyl, and aryl amines. This technology utilizes abundant organic carboxylic acids as carbon sources and easily prepared alkane/aryloxyacyl azides as nitrogen sources. By employing a dual catalytic system of DMAP and Copper(II) acetate, the process achieves high efficiency under neutral conditions, releasing only nitrogen and carbon dioxide as byproducts. This represents a significant leap forward for manufacturers seeking reliable pharmaceutical intermediate supplier partnerships that prioritize green chemistry and operational simplicity.
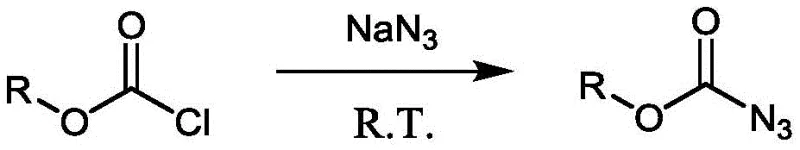
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sp3 hybridized C-N bonds has relied heavily on nucleophilic substitution reactions, Mitsunobu reactions, or reductive amination of carbonyl compounds. These classical approaches frequently suffer from significant drawbacks, including the requirement for stoichiometric amounts of hazardous reagents, poor functional group tolerance, and the generation of substantial chemical waste. Furthermore, methods capable of constructing both sp2 and sp3 C-N bonds simultaneously are relatively underdeveloped, often necessitating distinct synthetic routes for aromatic versus aliphatic amines. The reliance on expensive palladium catalysts in cross-coupling reactions also imposes a heavy financial burden on large-scale manufacturing, complicating the supply chain for cost reduction in API manufacturing. Additionally, the removal of residual heavy metals from the final product to meet stringent regulatory standards adds further complexity and cost to the production workflow.
The Novel Approach
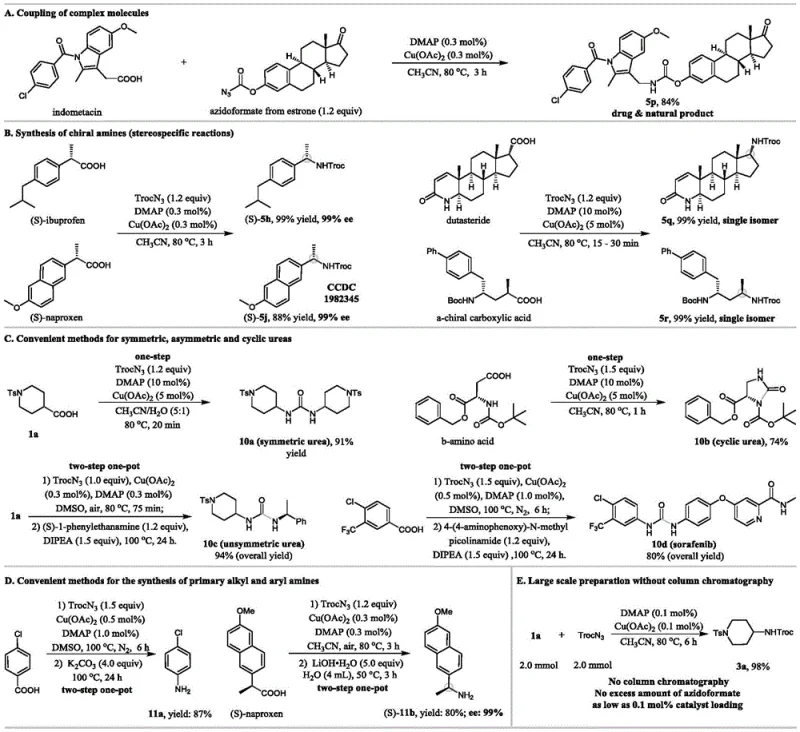
The technology disclosed in CN112028814B overcomes these hurdles by utilizing a catalytic decarboxylative strategy that unifies the synthesis of diverse amine classes. Unlike traditional methods that might require pre-functionalized halides or activated esters, this approach directly activates stable carboxylic acids. The reaction proceeds through a cascade involving carboxylic acid activation, azidation, Curtius rearrangement, and nucleophilic addition, all within a single pot. This streamlined process tolerates a wide array of functional groups, including hydroxyls, carbonyls, and olefins, without the need for protecting groups in many cases. The use of inexpensive copper and DMAP catalysts, combined with the evolution of gaseous byproducts that drive the reaction forward, ensures high atom economy. This novel pathway not only simplifies the synthetic route but also enhances the commercial scale-up of complex polymer additives and pharmaceutical intermediates by reducing step count and purification burdens.
Mechanistic Insights into Catalytic Decarboxylative Amination
The mechanistic pathway of this transformation is a sophisticated interplay of activation and rearrangement steps that ensure high fidelity and yield. Initially, the carboxylic acid is activated by the copper catalyst and DMAP, facilitating the nucleophilic attack by the acyl azide to form a mixed anhydride or activated acyl species. This intermediate undergoes a Curtius rearrangement, losing nitrogen gas to generate a highly reactive isocyanate species. Crucially, this rearrangement occurs with retention of configuration at the migrating carbon center, which is vital for the synthesis of chiral amines from enantiopure carboxylic acids. The resulting isocyanate is then trapped by the alcohol moiety of the azide reagent (or an external nucleophile in urea synthesis variants) to form the final carbamate or urea product. This mechanism explains the observed stereospecificity, where chiral centers adjacent to the carboxyl group are preserved with excellent enantiomeric excess, making it ideal for producing high-purity OLED material precursors or chiral drug fragments.
Impurity control in this system is inherently robust due to the clean nature of the byproducts. Since nitrogen and carbon dioxide are the sole gaseous emissions, there is minimal formation of salt waste or organic side products typically associated with coupling reagents like EDC or HATU. The catalytic cycle regenerates the active copper species, allowing for turnover numbers that support low catalyst loading. In scenarios where aryl carboxylic acids are used, the stability of the aromatic ring requires slightly elevated temperatures or extended reaction times, but the fundamental mechanism remains consistent. The tolerance for heteroatoms and sensitive functional groups suggests that the catalytic species does not promote unwanted side reactions such as beta-hydride elimination or over-oxidation, ensuring a clean impurity profile that simplifies downstream processing for regulatory compliance.
How to Synthesize Protected Amines Efficiently
Implementing this catalytic Curtius rearrangement in a laboratory or pilot plant setting requires careful attention to reagent ratios and thermal conditions to maximize yield. The process is designed to be operationally simple, utilizing common solvents like acetonitrile or DMSO and commercially available catalysts. The protocol allows for the direct conversion of readily available carboxylic acids into valuable protected amine intermediates, which can serve as versatile building blocks for further derivatization. For detailed procedural specifics regarding reagent addition sequences and workup procedures, please refer to the standardized synthesis guide below.
- Combine organic carboxylic acid, DMAP (0.3 mol%), and Cu(OAc)2 (0.3 mol%) in a reaction flask with acetonitrile solvent.
- Add alkane/aryloxy acyl azide (1.2 equiv) to the mixture and heat to 80°C for 3 hours under air.
- Upon completion, purify the crude product via column chromatography or recrystallization to isolate the protected amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers transformative benefits that address key pain points in chemical sourcing and manufacturing. The shift from precious metal catalysts to base metal systems like copper significantly lowers the raw material cost baseline, while the use of commodity carboxylic acids ensures a stable and diversified supply base. The elimination of stoichiometric activating agents reduces the volume of hazardous waste generated per kilogram of product, aligning with increasingly strict environmental regulations and reducing disposal costs. Furthermore, the ability to perform these reactions under air and in a one-pot fashion minimizes equipment downtime and cleaning cycles, thereby enhancing overall plant throughput and reliability for reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with inexpensive copper acetate and DMAP results in a drastic reduction in catalyst costs, which is particularly impactful for large-volume production runs. Additionally, the reaction generates gaseous byproducts that spontaneously leave the reaction mixture, eliminating the need for complex aqueous workups to remove salt byproducts, thus saving on solvent usage and labor. The high yields reported across a broad substrate scope mean that less starting material is wasted, directly improving the cost of goods sold (COGS) for the final active ingredient. This economic efficiency makes the process highly attractive for cost reduction in electronic chemical manufacturing and other high-value sectors.
- Enhanced Supply Chain Reliability: By utilizing organic carboxylic acids as the primary carbon source, manufacturers can leverage a vast global supply chain of stable, non-toxic, and shelf-stable raw materials. Unlike sensitive organometallic reagents that require cold chain logistics and inert atmosphere handling, these substrates are robust and easy to transport, mitigating risks associated with supply disruptions. The mild reaction conditions and tolerance to moisture and air further reduce the dependency on specialized infrastructure, allowing for more flexible manufacturing locations. This resilience ensures a continuous supply of critical intermediates, safeguarding production schedules against external logistical volatility.
- Scalability and Environmental Compliance: The process has been validated at gram scales with catalyst loadings as low as 0.1 mol%, demonstrating excellent potential for ton-scale production without proportional increases in catalyst cost. The clean reaction profile, producing only CO2 and N2 as byproducts, simplifies environmental permitting and waste treatment, as there are no heavy metal residues or toxic organic sludge to manage. This alignment with green chemistry principles not only reduces the environmental footprint but also future-proofs the manufacturing process against tightening global sustainability mandates. The ease of product isolation via recrystallization further supports scalable operations by avoiding resource-intensive chromatographic separations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this catalytic method. These insights are derived directly from the experimental data and scope studies presented in the patent documentation, providing clarity on substrate compatibility and operational parameters. Understanding these nuances is essential for R&D teams evaluating this technology for integration into existing synthetic routes.
Q: Does this catalytic method preserve stereochemistry in chiral amines?
A: Yes, the patent demonstrates complete chiral transfer. Reactions with enantiomerically pure carboxylic acids like (S)-ibuprofen and (S)-naproxen yielded corresponding amines with 99% ee, confirming the stereospecific nature of the rearrangement.
Q: What are the primary byproducts of this decarboxylative amination?
A: The reaction is atom-economical and generates only nitrogen gas (N2) and carbon dioxide (CO2) as sole byproducts, simplifying downstream purification and waste management.
Q: Can this method be scaled for industrial production of drug intermediates?
A: Absolutely. The patent reports successful gram-scale preparations with catalyst loading as low as 0.1 mol%, yielding products up to 98% purity after simple recrystallization, indicating high scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amine Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic methodologies like the catalytic Curtius rearrangement to enhance our portfolio of fine chemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are seamlessly translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of amine compound or urea derivative meets the exacting standards required by the global pharmaceutical and agrochemical industries. Our commitment to quality and consistency makes us a trusted partner for long-term supply agreements.
We invite you to collaborate with us to explore how this advanced amination technology can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating the economic advantages of switching to this catalytic route. Please contact us to request specific COA data and route feasibility assessments, and let us help you accelerate your development timeline with our reliable supply of high-performance chemical intermediates.
