Transforming Pharmaceutical Intermediate Manufacturing: Scalable Copper-Catalyzed Synthesis of High-Purity Benzoindolizine Compounds
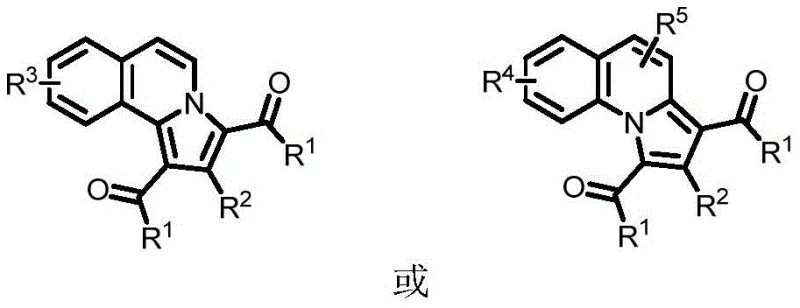
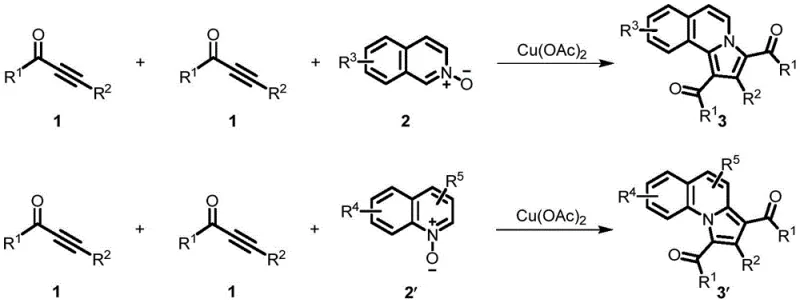
Patent CN11166290B introduces a groundbreaking methodology for synthesizing benzoindolizine compounds, a critical class of N-heterocyclic structures widely found in biologically active organic and pharmaceutical molecules essential for drug discovery pipelines. This innovative approach utilizes alkynone compounds and quinoline or isoquinoline nitrogen oxides as starting materials, employing copper(II) acetate as a catalyst under ambient air conditions to efficiently construct the benzoindolizine skeleton without requiring inert gas protection systems typically needed in conventional syntheses. The patented process represents a significant advancement over traditional synthetic routes by operating under milder reaction conditions while maintaining high product yields across diverse substrate combinations, thereby enhancing both operational simplicity and economic viability for industrial-scale production environments where reliability is paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing benzoindolizine compounds have been constrained by multiple significant limitations that hinder their industrial applicability and economic viability within pharmaceutical manufacturing contexts where cost-effectiveness is critical. Conventional methodologies typically rely on expensive transition metal catalysts such as palladium or gold complexes that substantially increase raw material costs while requiring complex removal procedures to meet stringent pharmaceutical purity specifications required by regulatory authorities worldwide. These processes often demand inert atmosphere conditions with specialized equipment that adds operational complexity and capital expenditure burdens to manufacturing facilities already operating under tight budget constraints. Furthermore, many established routes operate at extreme temperatures exceeding safe industrial processing limits or require multiple synthetic steps with low overall yields that complicate scale-up efforts essential for commercial production volumes where consistent supply is non-negotiable.
The Novel Approach
The patented methodology overcomes these limitations through an elegant copper-catalyzed cyclization strategy that operates efficiently under ambient air conditions without requiring inert gas protection systems or expensive transition metal catalysts typically associated with heterocyclic compound synthesis. By utilizing readily available copper(II) acetate as the catalyst at loadings between 0.2 to 2 equivalents relative to nitrogen oxide substrates, this approach significantly reduces raw material costs while maintaining high reaction efficiency across diverse substrate combinations including challenging functionalized derivatives required in modern drug development programs. The process operates within a moderate temperature range of 80°C to 120°C using standard laboratory equipment without specialized modifications, eliminating energy-intensive thermal requirements that increase operational costs in traditional approaches while enhancing process safety profiles essential for large-scale manufacturing operations where risk mitigation is critical.
Mechanistic Insights into Copper-Catalyzed Benzoindolizine Formation
The core innovation lies in a [2+2+1] cyclization mechanism where copper(II) acetate facilitates coupling between alkynone compounds and quinoline/isoquinoline nitrogen oxides through well-defined coordination chemistry that activates both reaction partners simultaneously without requiring additional oxidants or reductants typically needed in alternative methodologies. The copper catalyst initially coordinates with the nitrogen oxide functionality through oxygen atom interaction, activating the heterocyclic system toward nucleophilic attack by the alkyne moiety of the alkynone compound through π-complexation that lowers activation energy barriers significantly compared to uncatalyzed processes observed in literature precedents.
This catalytic cycle proceeds through copper-stabilized intermediates that undergo sequential bond formation events with precise regiocontrol dictated by electronic effects within both substrate classes, ultimately delivering the benzoindolizine scaffold with exceptional selectivity that minimizes formation of regioisomeric byproducts problematic in traditional syntheses requiring extensive purification efforts that compromise overall process economics essential for commercial viability in competitive markets where margins are increasingly tight.
Impurity control is achieved through the reaction's inherent selectivity combined with mild operating conditions that minimize unwanted side reactions commonly observed in harsher synthetic methodologies relying on elevated temperatures or aggressive reagents that generate difficult-to-remove impurities requiring sophisticated purification techniques incompatible with cost-effective manufacturing requirements essential for sustainable business operations within today's competitive pharmaceutical landscape where quality must be balanced against economic realities.
How to Synthesize Benzoindolizine Compounds Efficiently
This patented synthesis represents a significant advancement in heterocyclic chemistry methodology offering pharmaceutical manufacturers practical access to high-value benzoindolizine intermediates through an economically viable route that eliminates multiple costly steps required by conventional approaches while maintaining excellent product quality suitable for demanding regulatory environments where documentation standards are increasingly rigorous across global markets requiring consistent quality profiles regardless of production location or scale requirements essential for multinational operations.
- Prepare reaction mixture by combining alkynone compounds with quinoline or isoquinoline nitrogen oxides in PEG-200 or water solvent at concentration of 0.1-0.2 mol/L.
- Add copper(II) acetate catalyst (0.2-2 equivalents) under ambient air conditions without requiring inert gas protection.
- Heat reaction mixture to 80-120°C and stir for 2-12 hours followed by standard workup and purification procedures.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points faced by procurement professionals through its inherently more efficient process design that reduces raw material complexity while enhancing supply chain resilience through simplified sourcing requirements essential for maintaining consistent inventory levels required by just-in-time manufacturing systems increasingly adopted across the pharmaceutical industry where inventory carrying costs represent significant financial burdens requiring strategic mitigation approaches supported by reliable supplier partnerships.
- Cost Reduction in Manufacturing: Elimination of expensive transition metal catalysts represents substantial cost advantages over conventional methodologies as copper(II) acetate provides comparable catalytic efficiency at significantly lower material costs while operating under ambient air conditions without requiring inert gas protection systems that add both capital expenditure and ongoing operational expenses associated with gas handling infrastructure maintenance where every dollar saved contributes directly to improved profit margins essential for business sustainability in competitive markets.
- Enhanced Supply Chain Reliability: Utilization of readily available starting materials with broad commercial sourcing options significantly improves supply chain resilience compared to methodologies dependent on specialized or restricted reagents that create single points of failure within complex global supply networks where geopolitical factors increasingly impact material availability requiring diversified sourcing strategies supported by robust supplier relationships capable of adapting quickly to changing market conditions without compromising quality standards required by regulatory authorities worldwide.
- Scalability and Environmental Compliance: Methodology demonstrates excellent scalability from laboratory development stages through full commercial production volumes due to straightforward reaction profile compatible with standard manufacturing infrastructure enabling seamless technology transfer between facilities without requiring major process re-engineering investments that would otherwise delay time-to-market objectives critical for competitive advantage where speed is increasingly valued alongside cost considerations within rapidly evolving therapeutic landscapes demanding accelerated development timelines.
Frequently Asked Questions (FAQ)
The following questions address common concerns from procurement specialists regarding implementation timelines while providing technical insights valuable for R&D teams evaluating process feasibility alongside supply chain professionals assessing reliability metrics essential for comprehensive vendor evaluation processes where multiple stakeholder perspectives must be harmonized before final supplier selection decisions are made within complex organizational structures typical of multinational corporations.
Q: How does this copper-catalyzed method overcome limitations of traditional benzoindolizine synthesis?
A: The patented methodology eliminates need for expensive transition metal catalysts and inert gas protection while operating at milder temperatures (80-120°C), significantly improving process economics and operational simplicity compared to conventional approaches requiring costly catalysts and specialized equipment.
Q: What are key advantages of using copper(II) acetate instead of precious metal catalysts?
A: Copper(II) acetate offers substantial cost savings over precious metal catalysts while maintaining high efficiency, operates effectively under ambient air conditions without inert gas protection, eliminating need for additional additives that complicate purification processes.
Q: How does this synthesis ensure high substrate versatility and product purity?
A: Reaction demonstrates excellent substrate tolerance across diverse alkynone and (iso)quinoline nitrogen oxide structures with optimized conditions enabling straightforward product separation resulting in high-purity benzoindolizine compounds suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoindolizine Compound Supplier
Our patented copper-catalyzed synthesis methodology represents a significant advancement in producing high-value benzoindolizine intermediates with exceptional purity profiles suitable for demanding pharmaceutical applications requiring stringent quality control measures throughout manufacturing processes where consistency is non-negotiable across all production scales from early development through commercial launch phases where regulatory compliance must be maintained without exception across all batches produced globally regardless of facility location or scale requirements essential for multinational operations seeking reliable supply partners capable of meeting exacting standards consistently over time.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your particular application requirements where detailed technical documentation will be provided upon request following standard confidentiality protocols protecting proprietary information while enabling informed decision-making processes essential for establishing long-term supplier relationships built on mutual trust and technical excellence where every interaction contributes toward strengthening collaborative partnerships focused on shared business objectives requiring reliable access to high-quality intermediates supporting successful drug development programs worldwide.
