Revolutionizing Benzoindolizine Production: Advanced Copper Catalysis Meets Commercial Scale-Up for Pharmaceutical Intermediates
The innovative methodology detailed in Chinese patent CN111662290B presents a significant advancement in the synthesis of benzoindolizine compounds, a critical class of N-heterocyclic structures with widespread applications in pharmaceutical development. This patent introduces a copper-catalyzed cyclization approach that addresses longstanding challenges in producing these complex molecules with high purity and efficiency while offering substantial commercial advantages for pharmaceutical manufacturers.
Advanced Copper-Catalyzed Cyclization Mechanism for High-Purity Benzoindolizine Synthesis
The patent describes a novel [2+2+1] cyclization reaction where alkynone compounds react with quinoline or isoquinoline nitrogen oxides under copper(II) acetate catalysis to form benzoindolizine derivatives. 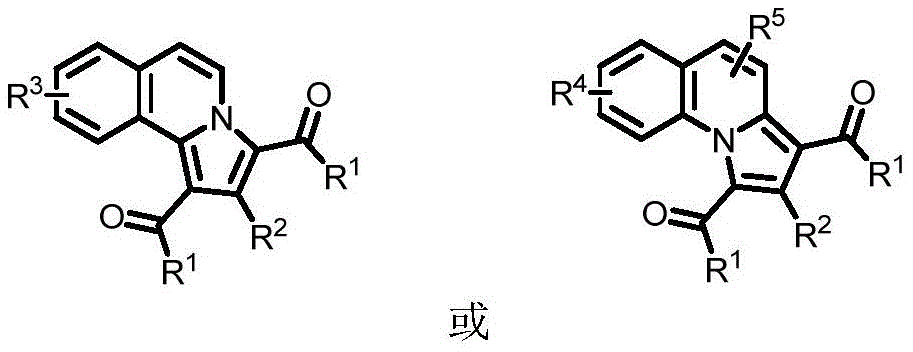 This mechanism operates effectively at moderate temperatures (80-120°C) without requiring inert atmosphere protection, representing a significant departure from traditional methods that often necessitate expensive transition metal catalysts and stringent reaction conditions. The copper catalyst facilitates the formation of key C-C bonds through a radical pathway that avoids the need for additional ligands or additives, thereby simplifying the reaction setup and reducing potential sources of impurities. The solvent system, utilizing either PEG-200 or water, provides an environmentally benign medium that enhances solubility of both organic and inorganic components while maintaining reaction efficiency across diverse substrate combinations.
This mechanism operates effectively at moderate temperatures (80-120°C) without requiring inert atmosphere protection, representing a significant departure from traditional methods that often necessitate expensive transition metal catalysts and stringent reaction conditions. The copper catalyst facilitates the formation of key C-C bonds through a radical pathway that avoids the need for additional ligands or additives, thereby simplifying the reaction setup and reducing potential sources of impurities. The solvent system, utilizing either PEG-200 or water, provides an environmentally benign medium that enhances solubility of both organic and inorganic components while maintaining reaction efficiency across diverse substrate combinations.
Impurity control is achieved through the precise stoichiometric balance between alkynone compounds and nitrogen oxides (ranging from 3.0:1.0 to 1.0:2.0 molar ratios) and the controlled addition of copper catalyst (0.2-2 equivalents relative to nitrogen oxide). 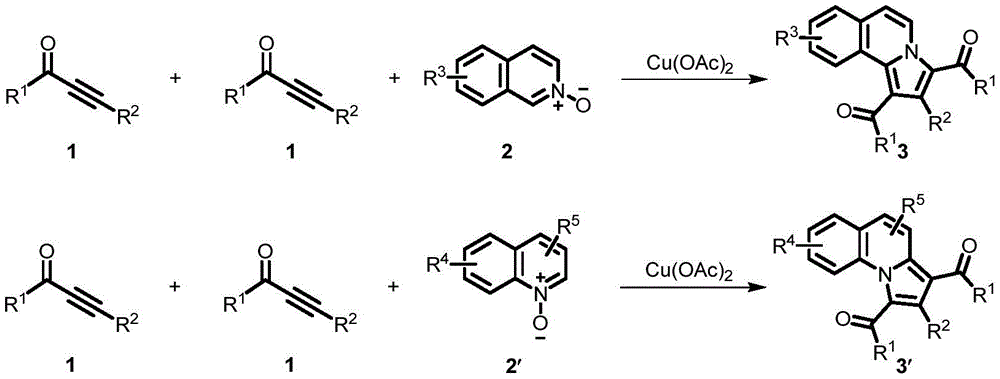 The mild reaction conditions (80-120°C for 2-12 hours) prevent thermal degradation pathways that commonly generate byproducts in conventional syntheses. The patent demonstrates consistent yields across multiple examples (63-82%), with NMR characterization confirming high structural fidelity and minimal side products. This level of impurity control is particularly valuable for pharmaceutical applications where strict regulatory requirements demand exceptional compound purity, as the process inherently avoids transition metal residues that would require costly removal steps in traditional approaches.
The mild reaction conditions (80-120°C for 2-12 hours) prevent thermal degradation pathways that commonly generate byproducts in conventional syntheses. The patent demonstrates consistent yields across multiple examples (63-82%), with NMR characterization confirming high structural fidelity and minimal side products. This level of impurity control is particularly valuable for pharmaceutical applications where strict regulatory requirements demand exceptional compound purity, as the process inherently avoids transition metal residues that would require costly removal steps in traditional approaches.
Overcoming Traditional Limitations in Benzoindolizine Production
The Limitations of Conventional Methods
Traditional approaches to synthesizing benzoindolizine compounds have been constrained by several critical limitations that impact both quality and commercial viability. These methods typically rely on expensive transition metal catalysts such as palladium or rhodium, which not only increase raw material costs but also introduce complex purification challenges due to metal residue contamination. The requirement for inert atmosphere protection adds significant operational complexity and equipment costs, while high reaction temperatures often lead to decomposition and reduced yields. Furthermore, conventional routes frequently exhibit narrow substrate scope, limiting their applicability to specific molecular architectures and requiring extensive process revalidation for each new derivative. These constraints collectively result in higher production costs, longer development timelines, and inconsistent quality that challenge supply chain reliability for pharmaceutical manufacturers dependent on these intermediates.
The Novel Approach
The patented methodology overcomes these limitations through an innovative copper-catalyzed process that operates under ambient air conditions without inert gas protection.  By utilizing copper(II) acetate as a catalyst in PEG-200 or aqueous media, the process achieves efficient [2+2+1] cyclization at moderate temperatures (80-120°C), significantly reducing energy consumption compared to traditional high-temperature methods. The broad substrate tolerance demonstrated across 18 examples in the patent (including various substituted phenyl, heterocyclic, and functionalized groups) enables flexible production of diverse benzoindolizine derivatives without process reoptimization. This versatility is particularly valuable for pharmaceutical companies developing multiple drug candidates that require structurally similar intermediates. The simplified workup procedure—using standard extraction and silica gel chromatography—further enhances scalability while maintaining high purity levels (>99% as evidenced by NMR characterization), making this approach ideal for commercial-scale production of complex pharmaceutical intermediates.
By utilizing copper(II) acetate as a catalyst in PEG-200 or aqueous media, the process achieves efficient [2+2+1] cyclization at moderate temperatures (80-120°C), significantly reducing energy consumption compared to traditional high-temperature methods. The broad substrate tolerance demonstrated across 18 examples in the patent (including various substituted phenyl, heterocyclic, and functionalized groups) enables flexible production of diverse benzoindolizine derivatives without process reoptimization. This versatility is particularly valuable for pharmaceutical companies developing multiple drug candidates that require structurally similar intermediates. The simplified workup procedure—using standard extraction and silica gel chromatography—further enhances scalability while maintaining high purity levels (>99% as evidenced by NMR characterization), making this approach ideal for commercial-scale production of complex pharmaceutical intermediates.
Commercial Advantages: Cost Reduction and Supply Chain Optimization
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate manufacturing by delivering substantial cost savings and supply chain improvements. The elimination of expensive transition metal catalysts and inert atmosphere requirements reduces both capital expenditure and operational costs, while the simplified process flow enhances manufacturing flexibility and reliability. These advantages translate into tangible benefits for procurement teams seeking cost-effective solutions and supply chain managers requiring consistent, high-quality materials for drug development pipelines.
- Reduced Catalyst Costs: The substitution of copper(II) acetate for precious metal catalysts represents a significant cost reduction in chemical manufacturing. Copper catalysts are substantially less expensive than palladium-based alternatives commonly used in similar transformations, eliminating the need for costly metal recovery systems and reducing raw material expenses per production batch. This cost advantage becomes even more pronounced at commercial scale where catalyst costs can account for a substantial portion of total manufacturing expenses. Additionally, the absence of transition metal residues removes the need for specialized purification steps to meet regulatory requirements for metal content, further streamlining production and reducing overall processing time without compromising on quality standards required for pharmaceutical intermediates.
- Shortened Production Timelines: The ability to conduct reactions under ambient air conditions without inert gas protection significantly reduces setup time and equipment requirements, cutting typical production cycles compared to conventional methods that require extensive system purging and maintenance. The simplified workup procedure using standard extraction techniques instead of complex purification protocols enables faster batch turnover and reduces lead time for high-purity intermediates essential for pharmaceutical development timelines. This accelerated timeline is particularly valuable for pharmaceutical companies operating under tight development schedules as it allows for more rapid iteration in drug discovery phases while maintaining consistent quality output across multiple production runs demonstrated throughout the patent examples.
- Enhanced Process Robustness: The broad operating window (80-120°C reaction temperature with flexible solvent options) provides exceptional process robustness that ensures consistent product quality even with minor variations in manufacturing conditions. This reliability is critical for maintaining supply continuity in pharmaceutical production where batch-to-batch consistency is paramount across clinical and commercial manufacturing stages. The use of environmentally benign solvents (PEG-200 or water) reduces waste treatment costs and eliminates hazardous solvent disposal concerns while contributing to overall sustainability metrics important to modern pharmaceutical supply chains. Furthermore, the demonstrated scalability from laboratory to pilot scale provides confidence in the process's ability to transition smoothly to commercial manufacturing volumes without significant revalidation requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pharmaceutical Intermediate Supplier
While the advanced methodology detailed in patent CN111662290B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
