Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Pharmaceutical Applications
Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Pharmaceutical Applications
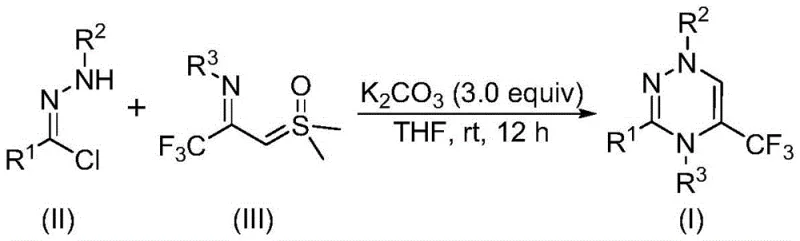
The rapid evolution of medicinal chemistry demands robust and efficient pathways for constructing nitrogen-rich heterocyclic scaffolds, particularly those incorporating fluorine motifs that enhance metabolic stability and bioavailability. A groundbreaking advancement in this domain is detailed in patent CN116253692A, which discloses a novel preparation method for trifluoromethyl-substituted 1,2,4-triazine compounds. This technology represents a significant leap forward for the industry, offering a streamlined approach to accessing these valuable pharmacophores without the logistical burdens associated with traditional transition-metal catalysis. By leveraging a unique [3+3] cycloaddition strategy between chlorohydrazones and trifluoroacetyl sulfur ylides, this method achieves high yields under exceptionally mild conditions. For R&D directors and process chemists, this translates to a reliable pathway for generating diverse libraries of bioactive molecules, while supply chain leaders benefit from the operational simplicity that facilitates seamless transition from gram-scale discovery to multi-ton commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazine core has relied on classical condensation reactions involving amidrazones and 1,2-diketones or alkynes, as well as multicomponent reactions utilizing hydrazides and dicarbonyl compounds. While these methods have served the community for decades, they are increasingly viewed as suboptimal for modern high-throughput drug discovery and manufacturing. Conventional routes often suffer from poor atom economy, requiring harsh reaction conditions such as high temperatures or strong acidic environments that limit functional group tolerance. Furthermore, the structural diversity achievable through these traditional pathways is frequently restricted, making it difficult to introduce specific substituents like the trifluoromethyl group at precise positions without extensive protecting group manipulation. The reliance on specialized substrates that are themselves difficult to synthesize adds layers of complexity and cost, creating bottlenecks that hinder the rapid iteration required in lead optimization campaigns.
The Novel Approach
In stark contrast to these legacy techniques, the methodology described in CN116253692A introduces a disruptive synthetic logic that bypasses these historical inefficiencies. The core innovation lies in the direct coupling of readily accessible chlorohydrazones with trifluoroacetyl sulfur ylides, mediated by a simple inorganic base. This approach eliminates the need for pre-functionalized diketones or complex multicomponent setups, drastically reducing the step count and overall process mass intensity. The reaction proceeds smoothly in common organic solvents like tetrahydrofuran at near-ambient temperatures, demonstrating remarkable compatibility with a wide array of electronic and steric environments on the aromatic rings. This versatility allows for the rapid generation of analogues with varied substitution patterns, empowering medicinal chemists to explore chemical space more effectively. Moreover, the avoidance of sensitive reagents means the process is far more forgiving and easier to control, setting a new standard for efficiency in heterocycle synthesis.
Mechanistic Insights into Potassium Carbonate-Promoted [3+3] Cycloaddition
The elegance of this transformation is rooted in its mechanistic simplicity yet profound utility. The reaction initiates with the deprotonation of the chlorohydrazone by potassium carbonate, triggering the elimination of hydrogen chloride to generate a highly reactive nitrile imine intermediate in situ. This transient species acts as a 1,3-dipole, poised for a concerted [3+3] cycloaddition with the trifluoroacetyl sulfur ylide, which serves as the three-carbon synthon. The synergy between these two components drives the formation of the six-membered triazine ring with high regioselectivity. Crucially, the final aromatization step involves the extrusion of dimethyl sulfoxide (DMSO), a thermodynamically favorable process that propels the equilibrium toward the desired product. This mechanism avoids the formation of stable off-cycle intermediates that often plague metal-catalyzed cycles, ensuring that the reaction flux remains directed toward the target molecule with minimal diversion into side products.
From an impurity control perspective, this metal-free pathway offers distinct advantages for pharmaceutical manufacturing. Traditional methods involving transition metals often leave behind trace residues that require costly and time-consuming scavenging steps to meet regulatory limits. By utilizing potassium carbonate, an inexpensive and non-toxic inorganic salt, the impurity profile is significantly cleaner, consisting primarily of organic byproducts that are easily removed via standard chromatographic techniques or crystallization. The mild reaction temperature of 20-40°C further suppresses thermal degradation pathways and polymerization side reactions that can occur under more vigorous conditions. This inherent selectivity ensures that the final trifluoromethyl-substituted 1,2,4-triazine compounds are obtained with high purity, reducing the burden on downstream purification units and enhancing the overall yield of the process.
How to Synthesize Trifluoromethyl 1,2,4-Triazine Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires attention to stoichiometry and mixing to maximize the efficiency of the cycloaddition. The protocol dictates a molar ratio where the trifluoroacetyl sulfur ylide and potassium carbonate are used in excess relative to the chlorohydrazone, typically favoring a 1:2:3 ratio to drive the reaction to completion. The choice of solvent is also critical, with tetrahydrofuran (THF) identified as the optimal medium due to its ability to dissolve both organic precursors and the inorganic base effectively. Operators should maintain the reaction under an air atmosphere, as no inert gas protection is necessary, which simplifies the equipment requirements significantly. Following the 10 to 14-hour reaction window, the workup involves a straightforward filtration to remove inorganic salts, followed by standard purification protocols.
- Combine chlorohydrazone, trifluoroacetyl sulfur ylide, and potassium carbonate (3.0 equivalents) in an organic solvent such as tetrahydrofuran (THF).
- Stir the reaction mixture at room temperature (20-40°C) under an air atmosphere for 10 to 14 hours to allow the [3+3] cycloaddition to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target trifluoromethyl-substituted 1,2,4-triazine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond mere chemical yield. The elimination of precious metal catalysts such as palladium, rhodium, or iridium removes a major cost driver from the bill of materials, while simultaneously mitigating the supply risk associated with these geopolitically sensitive commodities. Furthermore, the use of commodity chemicals like potassium carbonate and common solvents ensures that raw material sourcing is stable and predictable, shielding production schedules from the volatility often seen in the specialty reagent market. The operational simplicity of running reactions in air at room temperature also translates to reduced energy consumption and lower capital expenditure on specialized reactor infrastructure, contributing to substantial cost savings in API manufacturing.
- Cost Reduction in Manufacturing: The most significant financial advantage stems from the complete removal of expensive transition metal catalysts and the associated ligands, which often account for a disproportionate share of raw material costs in fine chemical synthesis. By replacing these with cheap, bulk inorganic bases, the direct material cost is drastically simplified. Additionally, the absence of heavy metals negates the need for specialized metal scavenging resins or complex aqueous workups designed to lower metal content to ppm levels, thereby reducing waste disposal costs and shortening the overall cycle time per batch.
- Enhanced Supply Chain Reliability: The starting materials for this process, including various acyl chlorides, hydrazines, and aromatic amines, are widely available from multiple global suppliers, ensuring a robust and competitive supply base. This diversity in sourcing options prevents single-point failures and allows procurement teams to negotiate better terms. The stability of the reagents also means they can be stored for extended periods without significant degradation, allowing for strategic stockpiling and buffering against market fluctuations or logistics disruptions.
- Scalability and Environmental Compliance: The benign nature of the reagents and the mild reaction conditions make this process inherently safer and easier to scale from kilogram to multi-ton quantities. The lack of pyrophoric reagents or high-pressure requirements reduces the safety footprint of the manufacturing facility. Moreover, the generation of dimethyl sulfoxide as a byproduct, rather than toxic heavy metal waste, aligns with increasingly stringent environmental regulations, facilitating smoother permitting processes and enhancing the sustainability profile of the final pharmaceutical intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl-triazine synthesis platform. These insights are derived directly from the experimental data and scope analysis provided in the patent documentation, aiming to clarify the practical aspects of adopting this technology for industrial applications. Understanding these nuances is essential for project managers evaluating the feasibility of integrating this route into existing production lines.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process explicitly avoids the use of heavy metal catalysts. It utilizes inexpensive and non-toxic potassium carbonate as a promoter, which significantly simplifies downstream purification and reduces heavy metal residue concerns in pharmaceutical intermediates.
Q: What are the optimal reaction conditions for this cycloaddition?
A: The reaction operates efficiently under mild conditions, specifically at temperatures between 20°C and 40°C. It does not require an inert atmosphere and can be conducted in air using tetrahydrofuran (THF) as the preferred solvent for 10 to 14 hours.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the process is highly scalable. The use of cheap, commercially available starting materials, combined with the absence of stringent moisture or oxygen exclusion requirements, makes it ideal for commercial scale-up of complex heterocyclic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of next-generation therapeutics. Our technical team has thoroughly analyzed the potential of the metal-free [3+3] cycloaddition pathway described in CN116253692A and is fully prepared to leverage this chemistry for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of trifluoromethyl-substituted 1,2,4-triazine delivered meets the highest industry standards for quality and consistency.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can optimize your specific supply chain requirements. Whether you require a Customized Cost-Saving Analysis for your current API roadmap or need to evaluate the feasibility of new analogues, our experts are ready to provide detailed route assessments. Please contact us today to request specific COA data for our available inventory or to initiate a feasibility study for your custom synthesis needs, ensuring your project stays on schedule and within budget.
