Advanced Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazines for Commercial Scale-Up
Advanced Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazines for Commercial Scale-Up
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activity. Patent CN116253692A introduces a groundbreaking preparation method for trifluoromethyl substituted 1,2,4-triazine compounds, addressing critical bottlenecks in the production of high-value pharmaceutical intermediates. These heterocyclic structures are ubiquitous in drug discovery, serving as key motifs in agents with anticancer, antifungal, anti-inflammatory, and antimalarial properties. The strategic incorporation of the trifluoromethyl group is not merely a structural variation but a deliberate enhancement of pharmacokinetic profiles, offering superior metabolic stability and lipophilicity compared to non-fluorinated analogs. This technological breakthrough enables the construction of complex triazine skeletons through a streamlined, metal-free protocol that operates under exceptionally mild conditions, marking a significant leap forward for reliable pharmaceutical intermediate supplier networks seeking robust and scalable manufacturing solutions.
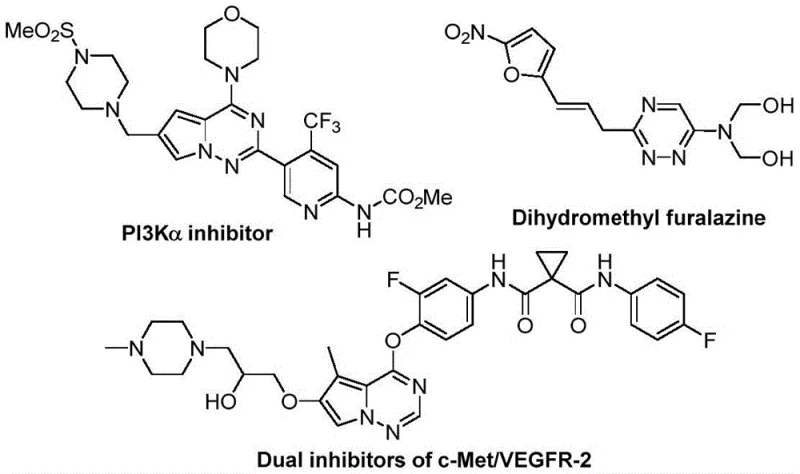
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazine derivatives has relied heavily on classical condensation reactions, such as the interaction between amidrazones and 1,2-diketones or alkynes, as well as multicomponent reactions involving hydrazides and dicarbonyl compounds. While these traditional pathways have served the industry for decades, they are increasingly viewed as inefficient for modern high-throughput demands. Conventional methods often suffer from severe limitations, including the necessity for pre-functionalized and difficult-to-synthesize substrates, which drives up raw material costs and extends lead times. Furthermore, these reactions frequently require harsh thermal conditions or the use of stoichiometric amounts of activating agents, leading to poor atom economy and generating substantial chemical waste. The structural diversity achievable through these older routes is also inherently restricted, limiting the ability of R&D teams to rapidly explore structure-activity relationships (SAR) around the triazine core. Additionally, many legacy processes rely on transition metal catalysts that necessitate rigorous and costly purification steps to meet stringent residual metal specifications required for API manufacturing.
The Novel Approach
In stark contrast to these legacy issues, the methodology disclosed in the patent utilizes a synergistic [3+3] cycloaddition strategy that fundamentally reshapes the synthetic landscape for these compounds. By employing readily accessible chlorohydrazones and trifluoroacetyl sulfur ylides as building blocks, the process bypasses the need for complex substrate preparation. The reaction proceeds efficiently in the presence of potassium carbonate, an inexpensive and non-toxic inorganic base, eliminating the dependency on precious metal catalysts entirely. This novel approach allows the transformation to occur at room temperature (20-40°C) in an open air atmosphere, removing the operational burden of inert gas protection and specialized pressure equipment. The result is a drastically simplified workflow that not only accelerates the timeline from bench to kilogram scale but also ensures a cleaner reaction profile with fewer byproducts. This shift represents a paradigm change in cost reduction in pharmaceutical intermediates manufacturing, offering a direct path to higher purity products with minimal downstream processing.
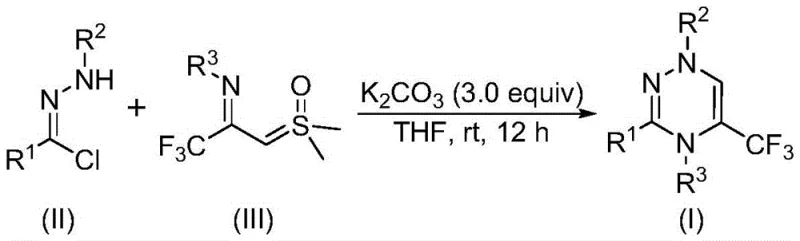
Mechanistic Insights into Potassium Carbonate-Promoted [3+3] Cycloaddition
The elegance of this synthesis lies in its mechanistic simplicity and high selectivity, driven by the unique reactivity of the sulfur ylide species. Under the promotion of potassium carbonate, the chlorohydrazone precursor undergoes dehydrohalogenation to generate a reactive nitrile imine intermediate in situ. This transient species then engages in a concerted [3+3] cycloaddition with the trifluoroacetyl sulfur ylide, effectively stitching together the six-membered triazine ring in a single operational step. The driving force for this cyclization is further enhanced by the elimination of dimethyl sulfoxide (DMSO), a thermodynamically favorable process that pushes the equilibrium toward the desired product. This mechanism avoids the formation of stable metallacycles often seen in transition-metal catalyzed couplings, thereby sidestepping issues related to catalyst poisoning or deactivation. From a quality control perspective, the absence of metal residues simplifies the impurity profile, making it easier to achieve the high-purity standards demanded by regulatory bodies for clinical trial materials.
Furthermore, the tolerance of this reaction system towards various functional groups is exceptional, allowing for the introduction of diverse substituents at the R1, R2, and R3 positions without compromising yield. Whether incorporating electron-withdrawing groups like halogens and trifluoromethyl moieties or electron-donating groups such as methoxy and alkyl chains, the reaction maintains robust performance. This versatility is crucial for medicinal chemists aiming to optimize the electronic properties of the final drug candidate. The mild basic conditions provided by potassium carbonate ensure that sensitive functional groups remain intact, preventing unwanted side reactions such as hydrolysis or elimination that might occur under stronger acidic or basic regimes. Consequently, this method provides a reliable platform for the commercial scale-up of complex heterocyclic intermediates, ensuring consistent batch-to-batch reproducibility essential for supply chain stability.
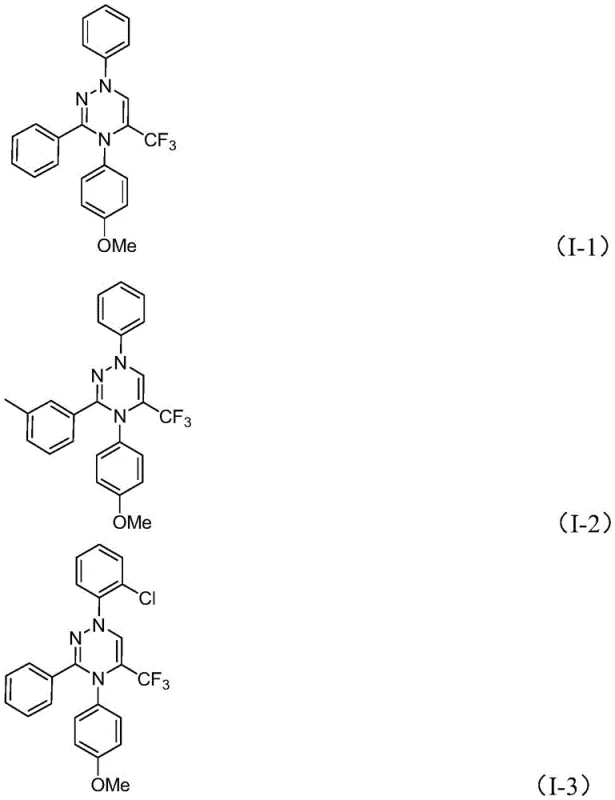
How to Synthesize Trifluoromethyl Substituted 1,2,4-Triazine Efficiently
To implement this advanced synthesis in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and mixing protocols to maximize conversion rates. The process begins with the careful selection of high-quality starting materials, specifically ensuring the chlorohydrazone and sulfur ylide are free from moisture which could interfere with the ylide stability. The reaction is typically conducted in aprotic solvents like tetrahydrofuran (THF), which effectively solubilizes both organic reactants and the inorganic base while facilitating the necessary ion-pairing interactions. Detailed standardized operating procedures regarding addition rates, stirring speeds, and temperature monitoring are critical to maintaining the exothermic balance during the initial mixing phase. For a comprehensive breakdown of the exact experimental parameters, reagent quantities, and workup techniques validated across multiple examples, please refer to the technical guide below.
- Prepare the reaction mixture by adding potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide into an organic solvent such as tetrahydrofuran.
- Stir the reaction mixture at room temperature (20-40°C) in an air atmosphere for 10 to 14 hours to allow the [3+3] cycloaddition to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final trifluoromethyl substituted 1,2,4-triazine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route offers transformative benefits that extend far beyond simple yield improvements. By eliminating the requirement for expensive transition metal catalysts such as palladium, copper, or rhodium, the direct material costs associated with each production batch are significantly reduced. Moreover, the removal of heavy metals from the process flow negates the need for specialized scavenging resins or complex extraction protocols designed to lower metal content to ppm levels, which traditionally adds substantial time and expense to the manufacturing cycle. This streamlining of the downstream process translates directly into faster turnaround times and lower overall production costs, providing a competitive edge in the pricing of high-purity pharmaceutical intermediates. The reliance on commodity chemicals like potassium carbonate and common organic solvents further insulates the supply chain from the volatility often seen in the market for specialty catalysts.
- Cost Reduction in Manufacturing: The economic impact of switching to this protocol is profound, primarily driven by the substitution of costly catalytic systems with inexpensive inorganic salts. Traditional methods often incur hidden costs related to catalyst recovery, recycling, or disposal of metal-laden waste streams, all of which are completely avoided here. The high atom economy of the [3+3] cycloaddition ensures that a greater proportion of the input mass is converted into valuable product, minimizing waste disposal fees. Additionally, the ability to run the reaction at ambient temperature reduces energy consumption associated with heating or cooling reactors, contributing to lower utility bills and a smaller carbon footprint for the facility.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of starting materials that are commercially available in bulk quantities from multiple global vendors. Chlorohydrazones and sulfur ylides can be synthesized from basic feedstocks like acyl chlorides and hydrazines, which are produced on a massive industrial scale, ensuring consistent availability even during market fluctuations. The robustness of the reaction conditions, which do not require stringent exclusion of oxygen or moisture, reduces the risk of batch failures due to minor environmental deviations. This reliability allows for more accurate forecasting and inventory planning, reducing the need for excessive safety stock and freeing up working capital for other strategic investments.
- Scalability and Environmental Compliance: Scaling this process from gram to multi-ton quantities is straightforward due to the absence of hazardous reagents and the simplicity of the reaction engineering. The use of non-toxic potassium carbonate aligns with green chemistry principles, facilitating easier permitting and compliance with increasingly strict environmental regulations regarding heavy metal discharge. The simplified workup, involving basic filtration and chromatography, is easily adaptable to continuous flow processing or large-scale batch reactors, enabling rapid response to surges in demand. This scalability ensures that the technology can support the entire lifecycle of a drug candidate, from early-stage discovery through to commercial API production, without the need for disruptive process redevelopment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl triazine synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this route into their existing pipelines and for procurement specialists assessing the long-term viability of the supply source.
Q: What are the key advantages of this new synthesis method over traditional condensation reactions?
A: Unlike traditional methods that often require harsh conditions and expensive substrates, this novel approach utilizes cheap, readily available starting materials like chlorohydrazones and trifluoroacetyl sulfur ylides. It operates under mild conditions (room temperature, air atmosphere) without the need for toxic heavy metal catalysts, significantly simplifying post-treatment and reducing environmental impact.
Q: How does the incorporation of the trifluoromethyl group benefit the final pharmaceutical compound?
A: The introduction of a trifluoromethyl group into the 1,2,4-triazine scaffold significantly enhances the physicochemical properties of the molecule. This modification improves metabolic stability, lipophilicity, and bioavailability, which are critical parameters for developing effective anticancer, antifungal, and anti-inflammatory agents.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is highly scalable. The use of inexpensive inorganic bases like potassium carbonate, combined with the ability to run the reaction in air without inert gas protection, makes it ideal for commercial scale-up. The simple workup procedure involving filtration and chromatography further supports efficient large-batch production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. We are committed to delivering trifluoromethyl substituted 1,2,4-triazine compounds with stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorine chemistry, guaranteeing the safety and consistency needed for global pharmaceutical supply chains.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. We encourage potential partners to contact us directly to obtain specific COA data for our catalog compounds and to discuss route feasibility assessments for custom derivatives. Let us help you accelerate your timeline to market with a supply partner dedicated to quality, efficiency, and scientific excellence.
