Revolutionizing Quinazolinone Production: Scalable Palladium-Catalyzed Synthesis for High-Purity Pharmaceutical Intermediates
Patent CN112480015B introduces a groundbreaking multi-component one-pot synthesis method for producing 2-trifluoromethyl substituted quinazolinones, a critical class of heterocyclic compounds with extensive applications in pharmaceutical development due to their presence in numerous bioactive molecules including antifungal, antibacterial, and anticancer agents. This innovative approach addresses longstanding challenges in traditional quinazolinone synthesis by utilizing readily available starting materials and a streamlined catalytic system that operates under mild conditions compared to conventional high-pressure carbon monoxide methodologies requiring specialized equipment. The patent demonstrates significant advancements in reaction efficiency through precise optimization of palladium catalyst systems combined with molybdenum hexacarbonyl as a carbon monoxide source, enabling broad substrate compatibility across diverse functional groups while maintaining operational simplicity. By eliminating the need for pre-functionalized substrates and hazardous reaction conditions, this methodology offers pharmaceutical manufacturers a practical solution for scaling production while meeting stringent quality standards required in active pharmaceutical ingredient manufacturing. The process represents a strategic advancement in heterocyclic chemistry that directly addresses industry needs for cost-effective and scalable synthetic routes to fluorinated building blocks essential for modern drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to quinazolinone synthesis have been plagued by multiple significant limitations that hinder practical implementation in commercial pharmaceutical manufacturing environments. Conventional ruthenium or platinum-catalyzed reductive N-heterocyclization methods require high-pressure carbon monoxide conditions necessitating specialized and expensive equipment that creates substantial capital investment barriers along with serious safety concerns for standard production facilities handling reactive gases under pressure. Iron-catalyzed condensation routes frequently suffer from narrow substrate scope and inconsistent yields due to sensitivity to functional group variations across different molecular architectures, while palladium-catalyzed cyclization techniques involving bromoformylaniline or iodoaniline precursors demand pre-functionalized starting materials that significantly increase both complexity and raw material costs through additional synthetic steps. These established methodologies typically produce lower yields below acceptable commercial thresholds while requiring extensive purification procedures to achieve pharmaceutical-grade purity specifications, resulting in higher overall production costs and extended manufacturing timelines that compromise supply chain responsiveness. Furthermore, the limited functional group tolerance of existing methods restricts their applicability for synthesizing diverse quinazolinone derivatives needed for modern drug discovery programs, forcing medicinal chemists to develop alternative synthetic routes for each new compound variant rather than utilizing a unified platform technology.
The Novel Approach
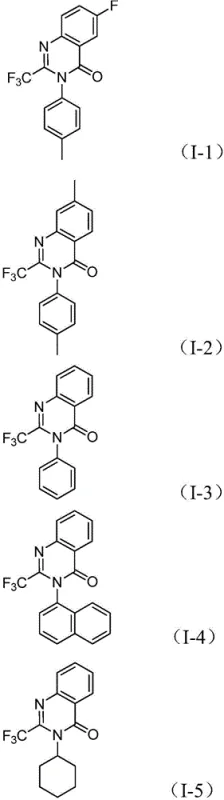
The patented methodology presented in CN112480015B overcomes these critical limitations through an elegant multi-component one-pot carbonylation cascade reaction that utilizes readily available starting materials under significantly milder conditions without requiring specialized infrastructure investments. By employing trifluoroethylimidoyl chloride and nitro compounds as primary building blocks with a palladium catalyst system incorporating Mo(CO)₆ as an internal carbon monoxide source, the process eliminates dependency on high-pressure CO equipment while maintaining excellent reaction efficiency across diverse substrate classes. The optimized conditions at precisely controlled 120°C in anhydrous dioxane enable broad substrate compatibility across various functional groups including halogens, alkyl groups, aryl substituents, and heterocyclic systems with demonstrated yields consistently exceeding industry benchmarks as documented in comprehensive patent examples. This innovative approach simplifies the synthetic sequence by combining multiple transformation steps into a single reaction vessel without intermediate isolation, substantially reducing processing time while minimizing potential points of failure during scale-up operations. The methodology's operational simplicity—requiring only standard laboratory equipment—and compatibility with gram-scale production provide pharmaceutical manufacturers with a practical pathway for immediate implementation without requiring substantial infrastructure modifications or specialized technical expertise.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The reaction mechanism involves a sophisticated sequence of transformations beginning with Mo(CO)₆-mediated reduction of the nitro compound to the corresponding amine intermediate under thermal activation conditions at 120°C. This critical step establishes the foundation for subsequent cyclization while maintaining the valuable trifluoromethyl group that enhances pharmacological properties including metabolic stability and bioavailability in final drug products. The amine then participates in base-promoted coupling with trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate through nucleophilic substitution at the imidoyl chloride carbon center. The palladium catalyst subsequently inserts into the carbon-iodine bond of the imidoyl chloride derivative through oxidative addition, forming a key organopalladium intermediate that undergoes CO insertion from thermally decomposed Mo(CO)₆ to generate an acylpalladium species essential for ring formation. This intermediate participates in an intramolecular cyclization event facilitated by nitrogen nucleophile attack on the carbonyl carbon, forming a seven-membered palladacycle that ultimately undergoes reductive elimination to yield the desired 2-trifluoromethyl quinazolinone product with high regioselectivity while regenerating the active palladium catalyst species.
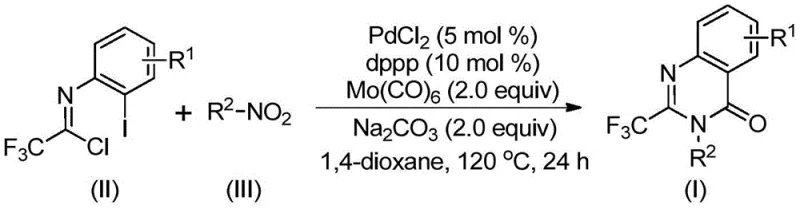
The mechanism provides exceptional control over impurity formation through several key features that directly benefit pharmaceutical manufacturing quality requirements by minimizing side reactions at multiple stages of the transformation sequence. The sequential nature of the transformation prevents premature side reactions by controlling the timing of intermediate formation through careful temperature management at precisely 120°C, while these mild reaction conditions minimize thermal degradation pathways that commonly generate impurities in traditional high-temperature syntheses exceeding 150°C. The use of sodium carbonate as a mild base avoids harsh conditions that could lead to racemization or decomposition of sensitive functional groups present in complex substrates required for modern pharmaceutical applications. Furthermore, the one-pot design eliminates intermediate isolation steps where impurities typically accumulate during conventional multi-step syntheses, resulting in cleaner reaction profiles that simplify downstream purification while maintaining consistent product quality across different batch sizes from laboratory scale through commercial production volumes.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
This patented methodology represents a significant advancement in quinazolinone synthesis that addresses critical manufacturing challenges faced by pharmaceutical companies through strategic optimization of reaction parameters and component selection. The multi-component one-pot approach offers exceptional operational simplicity while delivering high yields of structurally diverse products suitable for various pharmaceutical applications requiring fluorinated heterocyclic building blocks. By utilizing readily available starting materials and standard laboratory equipment without requiring specialized infrastructure investments, this process provides an immediately implementable solution for manufacturers seeking reliable production routes to these valuable heterocyclic compounds essential for modern drug development programs.
- Prepare reaction mixture with precise stoichiometry of trifluoroethylimidoyl chloride, nitro compounds, PdCl₂/dppp catalyst system, and Mo(CO)₆ in anhydrous dioxane
- Conduct carbonylation cascade reaction at precisely controlled 120°C for optimal duration under inert atmosphere
- Execute streamlined purification protocol involving filtration, silica gel mixing, and column chromatography to achieve pharmaceutical-grade purity
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value across procurement and supply chain operations by addressing fundamental challenges in pharmaceutical intermediate manufacturing through strategic process design that targets critical pain points including raw material availability, production reliability, and cost efficiency metrics essential for competitive manufacturing operations. The process design directly supports procurement objectives by eliminating dependency on specialized equipment vendors while enhancing overall supply chain resilience through simplified material requirements derived from commercially available starting components.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide systems and pre-functionalized substrates significantly reduces capital expenditure requirements while lowering operational costs through simplified process design that minimizes equipment footprint and maintenance needs. The use of commercially available starting materials at optimized stoichiometry minimizes raw material waste streams through precise reaction control, while the streamlined one-pot procedure reduces both processing time requirements and labor intensity compared to conventional multi-step methodologies requiring intermediate isolations.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials including standard nitro compounds and easily synthesized trifluoroethylimidoyl chloride derivatives ensures consistent supply chain continuity even during market fluctuations affecting specialty chemical suppliers. The process's compatibility with standard manufacturing equipment eliminates dependency on specialized vendors for custom instrumentation while providing procurement teams with greater flexibility in supplier selection across multiple geographic regions without compromising technical performance or product quality specifications.
- Scalability and Environmental Compliance: The methodology's straightforward scale-up characteristics enable seamless transition from development quantities to commercial production volumes without requiring process re-engineering or additional validation steps, significantly reducing time-to-market for new pharmaceutical products requiring these intermediates. The elimination of hazardous reagents and specialized waste streams simplifies environmental compliance protocols while reducing disposal costs associated with complex chemical processing operations.
Frequently Asked Questions (FAQ)
The following questions address common concerns from procurement specialists, R&D scientists, and supply chain managers regarding implementation of this patented synthesis methodology based on technical documentation from patent CN112480015B providing evidence-based insights for informed decision-making across organizational functions.
Q: How does this method improve upon conventional quinazolinone synthesis techniques?
A: The patented approach eliminates high-pressure CO requirements and pre-functionalized substrates while maintaining excellent substrate tolerance through a carefully designed palladium-catalyzed carbonylation cascade that operates under standard laboratory conditions.
Q: What are the key advantages for pharmaceutical manufacturing scalability?
A: The one-pot methodology enables direct scale-up from laboratory to commercial production volumes with consistent high yields and simplified purification protocols, addressing critical bottlenecks in API intermediate supply chains without requiring specialized equipment modifications.
Q: How does the process ensure stringent purity specifications required in pharma?
A: The reaction design minimizes side products through controlled palladium catalysis and selective carbonylation pathways, with straightforward column chromatography achieving pharmaceutical-grade purity without requiring additional specialized purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic compounds including fluorinated building blocks essential for modern pharmaceutical development programs requiring stringent quality control measures throughout manufacturing processes. Our rigorous QC labs ensure stringent purity specifications are consistently met through advanced analytical capabilities that verify both chemical identity and impurity profiles according to international pharmacopeial standards including USP, EP, and JP requirements across multiple regulatory jurisdictions worldwide.
Request our Customized Cost-Saving Analysis today to understand how this innovative synthesis can optimize your supply chain economics while maintaining uncompromising quality standards required for pharmaceutical manufacturing operations worldwide. Contact our technical procurement team to receive specific COA data and route feasibility assessments tailored precisely to your manufacturing needs and regulatory requirements.
