Scalable One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones via Palladium-Catalyzed Carbonylation
Scalable One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones via Palladium-Catalyzed Carbonylation
The quinazolinone scaffold represents a cornerstone structure in modern medicinal chemistry, underpinning the efficacy of numerous therapeutic agents ranging from antifungal and antiviral medications to potent anticancer drugs. As illustrated in the structural diversity of known bioactive molecules such as Methaqualone and Afloqualone, the incorporation of fluorine atoms, particularly the trifluoromethyl group, dramatically enhances metabolic stability, lipophilicity, and bioavailability. 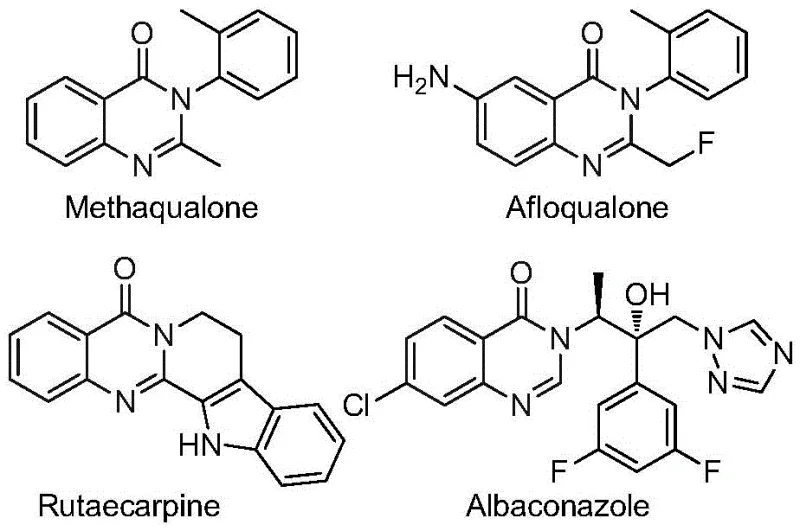 Recognizing the critical demand for efficient access to these privileged scaffolds, recent intellectual property CN112480015B discloses a groundbreaking multi-component one-pot methodology. This patent describes a robust palladium-catalyzed carbonylation cascade that utilizes inexpensive nitro compounds and trifluoroethylimidoyl chlorides as starting materials, effectively bypassing the limitations of traditional synthetic routes while delivering high-purity 2-trifluoromethyl substituted quinazolinones suitable for rigorous pharmaceutical applications.
Recognizing the critical demand for efficient access to these privileged scaffolds, recent intellectual property CN112480015B discloses a groundbreaking multi-component one-pot methodology. This patent describes a robust palladium-catalyzed carbonylation cascade that utilizes inexpensive nitro compounds and trifluoroethylimidoyl chlorides as starting materials, effectively bypassing the limitations of traditional synthetic routes while delivering high-purity 2-trifluoromethyl substituted quinazolinones suitable for rigorous pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has relied on methodologies that impose significant logistical and economic burdens on chemical manufacturing. Traditional pathways often necessitate the use of high-pressure carbon monoxide gas, which requires specialized, expensive reactor infrastructure and stringent safety protocols to mitigate toxicity risks. Alternative strategies involving ruthenium or platinum catalysts frequently suffer from narrow substrate scopes and low turnover numbers, leading to poor atom economy. Furthermore, many established protocols depend on pre-activated substrates such as 2-bromoformylanilines or acid anhydrides, which are not only costly to procure but also generate substantial stoichiometric waste during the coupling process. These factors collectively hinder the scalability and cost-efficiency required for the commercial production of complex heterocyclic intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology detailed in patent CN112480015B introduces a streamlined, one-pot transformation that leverages the reducing power of molybdenum hexacarbonyl [Mo(CO)6] to drive the reaction forward without external CO pressure. This innovative approach couples readily available nitro compounds with trifluoroethylimidoyl chlorides in the presence of a palladium catalyst system. 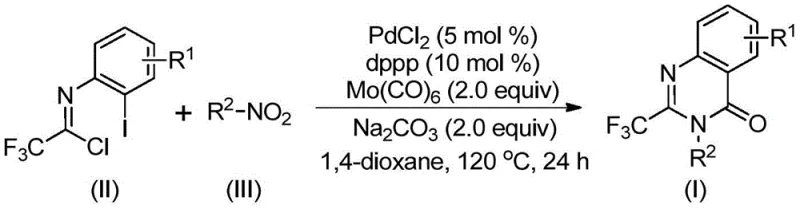 By operating at a moderate temperature of 120°C in 1,4-dioxane, this method achieves excellent conversion rates and broad functional group tolerance. The elimination of high-pressure gas equipment and the use of commodity chemical feedstocks represent a paradigm shift towards safer, more sustainable, and economically superior manufacturing processes for high-value pharmaceutical intermediates.
By operating at a moderate temperature of 120°C in 1,4-dioxane, this method achieves excellent conversion rates and broad functional group tolerance. The elimination of high-pressure gas equipment and the use of commodity chemical feedstocks represent a paradigm shift towards safer, more sustainable, and economically superior manufacturing processes for high-value pharmaceutical intermediates.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a sophisticated interplay of reduction, coupling, and cyclization events orchestrated by the palladium catalyst. The process likely initiates with the reduction of the nitro group on the aromatic ring to the corresponding amine by Mo(CO)6, which simultaneously serves as the carbon monoxide source. Following this in situ generation of the amine, a base-promoted intermolecular coupling occurs between the amine and the trifluoroethylimidoyl chloride, forming a trifluoroacetamidine derivative. Subsequently, the palladium catalyst undergoes oxidative addition into the carbon-iodine bond of the imidoyl chloride moiety, generating a reactive divalent palladium intermediate.
As the reaction progresses, thermal decomposition of Mo(CO)6 releases carbon monoxide, which inserts into the carbon-palladium bond to form an acyl-palladium species. This key intermediate then undergoes intramolecular nucleophilic attack by the nitrogen atom, facilitated by the base, to close the seven-membered cyclic palladium intermediate. The final step involves reductive elimination, which releases the desired 2-trifluoromethyl substituted quinazolinone product and regenerates the active palladium catalyst. This elegant cascade ensures high atom efficiency and minimizes the formation of side products, thereby simplifying downstream purification and ensuring the delivery of high-purity quinazolinone derivatives essential for drug development.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The execution of this synthesis is designed for operational simplicity, making it highly attractive for process chemistry teams aiming to rapidly access diverse libraries of quinazolinone analogs. The protocol requires the precise combination of palladium chloride, the bidentate ligand 1,3-bis(diphenylphosphino)propane (dppp), sodium carbonate, and Mo(CO)6 in an anhydrous organic solvent. Detailed standardized synthetic steps, including specific molar ratios and workup procedures validated across multiple substrates, are provided in the guide below to ensure reproducibility and optimal yield.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in 1,4-dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours under inert atmosphere to facilitate the carbonylation cascade.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling strategic advantages that directly impact the bottom line and operational resilience. By shifting away from hazardous high-pressure gases and expensive pre-functionalized building blocks, manufacturers can significantly reduce capital expenditure on specialized equipment and lower the overall cost of goods sold. The reliance on nitro compounds, which are abundant commodity chemicals with stable global supply chains, mitigates the risk of raw material shortages that often plague the production of specialty fine chemicals.
- Cost Reduction in Manufacturing: The substitution of high-pressure carbon monoxide with solid Mo(CO)6 eliminates the need for expensive autoclaves and complex gas handling systems, drastically lowering infrastructure maintenance costs. Additionally, the use of cheap nitro compounds instead of costly acid anhydrides or bromo-anilines reduces raw material expenses substantially. The one-pot nature of the reaction also minimizes solvent usage and labor hours associated with intermediate isolation, leading to comprehensive operational cost savings.
- Enhanced Supply Chain Reliability: Nitro compounds and trifluoroethylimidoyl chlorides are widely available from multiple global suppliers, ensuring a robust and diversified supply base that protects against market volatility. The high functional group tolerance of this method allows for the use of varied substrate grades without compromising reaction efficiency, providing flexibility in sourcing strategies. This reliability is critical for maintaining continuous production schedules and meeting tight delivery deadlines for pharmaceutical clients.
- Scalability and Environmental Compliance: The reaction conditions are mild and utilize standard organic solvents like 1,4-dioxane, facilitating seamless scale-up from laboratory benchtop to multi-ton commercial production without significant process re-engineering. The avoidance of toxic CO gas and the generation of manageable solid byproducts simplify waste treatment protocols, ensuring compliance with increasingly stringent environmental regulations. This green chemistry profile enhances the sustainability credentials of the final product, aligning with the ESG goals of modern pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of this synthesis method for industrial partners seeking reliable solutions.
Q: What are the key advantages of using nitro compounds over traditional amines in this synthesis?
A: Nitro compounds are significantly cheaper and more readily available than pre-functionalized amines or acid anhydrides. Furthermore, this method avoids the need for hazardous high-pressure carbon monoxide gas by utilizing solid Mo(CO)6 as a safe CO surrogate, enhancing operational safety and reducing infrastructure costs.
Q: What is the substrate scope and functional group tolerance of this Pd-catalyzed method?
A: The protocol demonstrates excellent compatibility with various substituents including halogens (F, Cl, Br), alkyl groups (methyl, n-propyl), and electron-withdrawing groups (CF3). It successfully accommodates both aryl and alkyl nitro compounds, yielding products with efficiencies ranging up to 96%.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the one-pot nature of the reaction simplifies processing by eliminating intermediate isolation steps. The use of standard organic solvents like 1,4-dioxane and commercially available catalysts makes the transition from gram-scale laboratory synthesis to multi-kilogram industrial production straightforward and economically viable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN112480015B for accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from concept to market. Our state-of-the-art facilities are equipped to handle complex palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that guarantee the highest quality standards for every batch of 2-trifluoromethyl quinazolinone delivered.
We invite you to collaborate with our technical team to explore how this cost-effective synthesis route can optimize your specific supply chain requirements. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, along with specific COA data and route feasibility assessments tailored to your project needs. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
