Scalable Synthesis of Bioactive Indole-Derived Piperidines via Efficient Lewis Acid Catalysis
Scalable Synthesis of Bioactive Indole-Derived Piperidines via Efficient Lewis Acid Catalysis
The pharmaceutical industry is constantly seeking robust synthetic methodologies to access complex heterocyclic scaffolds that serve as privileged structures in drug discovery. Patent CN115785122A introduces a groundbreaking approach to synthesizing a novel class of indole-derived piperidine compounds, designated as Formula 3, which have demonstrated potent cytotoxic activity against human breast cancer cell lines. This technology represents a significant leap forward in the construction of fused nitrogen-containing heterocycles, utilizing a highly efficient Lewis acid-catalyzed cascade reaction. The core innovation lies in the direct coupling of 3-alkyl-2-indole carbinols with alpha,beta-unsaturated N-sulfonyl imine derivatives, bypassing traditional multi-step protections and deprotections. For R&D directors and process chemists, this pathway offers a streamlined route to high-value pharmaceutical intermediates with exceptional stereocontrol.
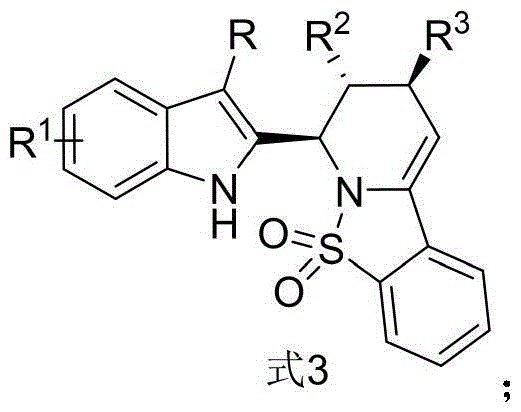
The structural versatility of this scaffold is immediately apparent, allowing for extensive SAR (Structure-Activity Relationship) studies crucial for lead optimization in oncology programs. The ability to introduce diverse substituents at the R, R1, R2, and R3 positions enables the rapid generation of chemical libraries. Furthermore, the reported biological data indicates that these derivatives possess strong sensitivity towards MCF-7 cells, suggesting a promising future as potential anticancer agents or key building blocks for API synthesis. As a reliable pharmaceutical intermediates supplier, understanding the nuances of such patented methodologies is essential for ensuring supply chain continuity and intellectual property compliance in commercial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of complex piperidine-fused indole systems has been fraught with synthetic challenges that hinder scalability and cost-efficiency. Traditional routes often rely on stepwise assembly involving multiple protection-deprotection cycles, harsh reaction conditions, and the use of stoichiometric amounts of toxic reagents. These conventional methods frequently suffer from poor atom economy and low diastereoselectivity, necessitating difficult and yield-losing chromatographic separations to isolate the desired stereoisomer. Moreover, the requirement for extreme temperatures or sensitive organometallic reagents often limits the functional group tolerance, thereby restricting the diversity of analogues that can be practically synthesized. For procurement managers, these inefficiencies translate into higher raw material costs, extended production timelines, and increased waste disposal burdens, making the cost reduction in pharmaceutical intermediates manufacturing a critical pain point.
The Novel Approach
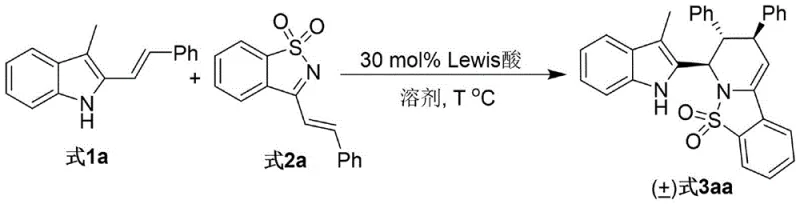
In stark contrast, the methodology disclosed in CN115785122A utilizes a convergent one-pot strategy that dramatically simplifies the synthetic landscape. By employing a Lewis acid catalyst, specifically Scandium triflate (Sc(OTf)3), the reaction proceeds under mild thermal conditions ranging from 50°C to 90°C, with an optimal setpoint at 70°C. This approach not only tolerates a wide array of functional groups on both the indole and imine components but also achieves remarkable diastereoselectivity, often exceeding a ratio of >95:5. The operational simplicity of this process—simply mixing the substrates in a solvent like acetonitrile with the catalyst—eliminates the need for inert atmosphere techniques or cryogenic cooling. This shift towards milder, catalytic processes directly addresses the industry's demand for greener chemistry and facilitates the commercial scale-up of complex pharmaceutical intermediates by reducing unit operations and energy consumption.
Mechanistic Insights into Sc(OTf)3-Catalyzed Cycloaddition
The success of this transformation hinges on the unique activation mode provided by the Scandium triflate catalyst. Mechanistically, the Lewis acid coordinates with the nitrogen and oxygen atoms of the alpha,beta-unsaturated N-sulfonyl imine, significantly increasing the electrophilicity of the beta-carbon. This activation facilitates a nucleophilic attack by the electron-rich C3-position of the indole ring (or a related reactive intermediate derived from the 3-alkyl-2-indole carbinol), initiating a cascade cyclization sequence. The rigid transition state imposed by the catalyst-substrate complex is responsible for the high level of stereocontrol observed, ensuring the formation of the thermodynamically favored diastereomer. Understanding this mechanistic pathway is vital for process chemists aiming to troubleshoot potential side reactions or optimize the reaction further for kilogram-scale production.
Furthermore, the impurity profile of this reaction is exceptionally clean, largely due to the high specificity of the catalytic cycle. The patent data indicates that competing pathways, such as polymerization of the unsaturated imine or decomposition of the indole substrate, are effectively suppressed under the optimized conditions (30 mol% catalyst in acetonitrile). This high fidelity in product formation minimizes the burden on downstream purification processes, which is a key factor in determining the overall cost of goods sold (COGS). For quality assurance teams, the consistency of the diastereomeric ratio (dr) across various substrates suggests a robust process that is less susceptible to minor fluctuations in raw material quality, thereby enhancing the reliability of the supply chain for high-purity pharmaceutical intermediates.
How to Synthesize Indole-Derived Piperidine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the precise weighing of 3-alkyl-2-indole carbinol and the alpha,beta-unsaturated N-sulfonyl imine derivative, typically in a molar ratio of 1:2 to drive the equilibrium towards product formation. These reactants are dissolved in dry acetonitrile, and the Sc(OTf)3 catalyst is added under standard stirring conditions. The reaction mixture is then heated to 70°C and monitored via Thin Layer Chromatography (TLC) until the starting materials are fully consumed, usually within 8 hours. Following completion, the workup involves simple filtration and concentration, followed by purification via silica gel column chromatography using a petroleum ether/ethyl acetate system. Detailed standardized synthesis steps are provided in the guide below.
- Combine 3-alkyl-2-indole carbinol (Formula 1) and alpha,beta-unsaturated N-sulfonyl imine derivative (Formula 2) in an organic solvent such as acetonitrile.
- Add 30 mol% of Lewis acid catalyst, specifically Sc(OTf)3, to the reaction mixture under stirring.
- Heat the reaction to 70°C and monitor progress via TLC until completion, followed by filtration, concentration, and silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The elimination of harsh reagents and the use of common organic solvents like acetonitrile significantly lower the barrier to entry for manufacturing partners. The mild reaction temperatures reduce energy overheads, while the high yields reported (often exceeding 70-90% for optimized substrates) ensure maximum throughput from raw materials. For supply chain heads, the reliance on commercially available starting materials—substituted indoles and sulfonyl imines—mitigates the risk of supply bottlenecks associated with exotic or custom-synthesized precursors. This accessibility is crucial for maintaining reducing lead time for high-purity pharmaceutical intermediates in a fast-paced drug development environment.
- Cost Reduction in Manufacturing: The catalytic nature of the Sc(OTf)3 system, despite the metal cost, allows for high turnover and eliminates the need for stoichiometric promoters that generate massive amounts of salt waste. The simplified workup procedure, which avoids complex extractions or recrystallizations in favor of straightforward chromatography, reduces labor hours and solvent consumption. Qualitatively, this translates to a leaner manufacturing process where resources are focused on value-added steps rather than waste management, driving down the overall production cost per kilogram of the active intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that the process is less sensitive to environmental variables, leading to consistent batch-to-batch quality. The broad substrate scope demonstrated in the patent implies that the supply chain is not dependent on a single, narrowly defined precursor; rather, a variety of substituted indoles and imines can be utilized to access the same core scaffold. This flexibility allows procurement teams to source materials from multiple vendors, hedging against market volatility and ensuring uninterrupted production schedules for critical oncology research programs.
- Scalability and Environmental Compliance: The absence of toxic heavy metals or pyrophoric reagents aligns well with modern environmental, health, and safety (EHS) standards. Scaling this reaction from gram to kilogram quantities does not require specialized pressure vessels or cryogenic infrastructure, making it suitable for existing multipurpose reactor suites. The reduced generation of hazardous byproducts simplifies waste treatment protocols, facilitating easier regulatory approval for commercial manufacturing sites and supporting the company's sustainability goals without compromising on output volume.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Whether you are evaluating the feasibility of in-house production or assessing the quality of external suppliers, understanding these details is paramount for successful project execution.
Q: What is the optimal catalyst and temperature for this synthesis?
A: According to patent CN115785122A, Scandium triflate (Sc(OTf)3) at 30 mol% loading provides the best results, with an optimal reaction temperature of 70°C in acetonitrile solvent.
Q: What is the biological activity profile of these compounds?
A: The synthesized indole-derived piperidine compounds exhibit significant cytotoxic activity against human breast cancer cells (MCF-7), with IC50 values demonstrating high sensitivity in biological assays.
Q: How does this method improve upon traditional synthesis routes?
A: This method offers a one-pot synthesis with mild reaction conditions (50-90°C), high diastereoselectivity (>95:5 dr), and excellent yields, eliminating the need for complex multi-step sequences often required for constructing fused heterocyclic systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole-Derived Piperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced heterocyclic scaffolds like the indole-derived piperidines described in CN115785122A for the next generation of anticancer therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to leverage our technical expertise to optimize your supply chain for these high-value compounds. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your R&D and commercial goals efficiently and reliably.
