Advanced Synthesis of Indole-Derived Piperidines for Oncology Drug Development
Advanced Synthesis of Indole-Derived Piperidines for Oncology Drug Development
The pharmaceutical industry is constantly seeking novel scaffolds that combine proven pharmacophores to unlock new therapeutic potentials, particularly in the challenging field of oncology. Patent CN115785122A introduces a groundbreaking class of indole-derived piperidine compounds, represented generally by Formula 3, which merge the biological significance of indole and piperidine skeletons into a single, potent molecular architecture. These compounds have demonstrated remarkable cytotoxic activity against human breast cancer MCF-7 cells, positioning them as high-value candidates for next-generation anticancer drug discovery. The synthesis method described represents a significant leap forward in heterocyclic chemistry, offering a direct, one-pot route that bypasses the cumbersome multi-step sequences traditionally associated with constructing such complex fused ring systems. By leveraging advanced Lewis acid catalysis, this technology provides a robust platform for generating diverse libraries of bioactive molecules with high structural complexity and purity.
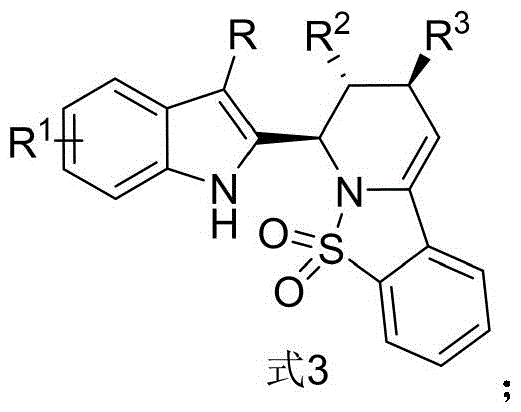
For research directors and process chemists, the ability to access these complex architectures efficiently is paramount. The general structure shown above highlights the versatility of the scaffold, where R can be varied from alkyl to aryl groups, and the aromatic systems can be functionalized with halogens or alkoxy groups. This modularity allows for extensive Structure-Activity Relationship (SAR) studies, enabling medicinal chemists to fine-tune the potency and selectivity of the lead compounds. The integration of the sulfonyl group within the piperidine ring further enhances the metabolic stability and solubility profile of the final drug candidates, addressing common liabilities in early-stage drug development. As a reliable pharmaceutical intermediate supplier, understanding the nuances of this synthetic pathway is crucial for ensuring a steady supply of high-quality materials for preclinical evaluation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of piperidine rings fused with indole moieties has been a formidable challenge in organic synthesis, often requiring harsh reaction conditions and multiple protection-deprotection steps. Traditional approaches frequently rely on transition metal catalysts that are not only expensive but also pose significant challenges regarding residual metal contamination in the final Active Pharmaceutical Ingredient (API). Furthermore, achieving high diastereoselectivity in the formation of multiple chiral centers within the piperidine ring typically necessitates the use of chiral auxiliaries or resolution techniques, which drastically reduce the overall atom economy and increase production costs. These conventional routes often suffer from low yields and poor scalability, making them unsuitable for the rapid generation of material needed for biological testing or commercial manufacturing. The reliance on toxic reagents and difficult purification processes also creates substantial environmental burdens, conflicting with the modern principles of green chemistry that govern sustainable pharmaceutical manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a highly efficient Lewis acid-catalyzed cascade reaction that constructs the complex piperidine core in a single operational step. As illustrated in the reaction scheme below, the coupling of 3-alkyl-2-indole methanols with alpha,beta-unsaturated N-sulfonyl imines proceeds smoothly under mild thermal conditions. This novel approach eliminates the need for pre-functionalized starting materials and avoids the use of stoichiometric amounts of hazardous reagents. The reaction is driven by the activation of the imine electrophile by the Lewis acid, facilitating a concerted cyclization that establishes the stereocenters with exceptional control. This streamlining of the synthetic route not only accelerates the timeline from bench to kilogram scale but also significantly simplifies the downstream processing, as the crude reaction mixtures are often cleaner and easier to purify than those from traditional multi-step syntheses.
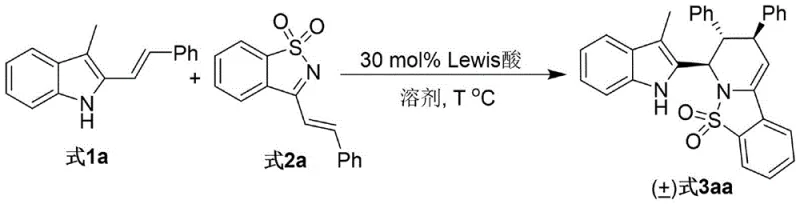
Mechanistic Insights into Sc(OTf)3-Catalyzed Cyclization
The success of this transformation hinges on the precise selection of the Lewis acid catalyst, with Scandium(III) triflate (Sc(OTf)3) emerging as the superior choice among various metals tested. The mechanism likely involves the coordination of the scandium center to the nitrogen atom of the N-sulfonyl imine, thereby increasing the electrophilicity of the beta-carbon and activating the system for nucleophilic attack by the electron-rich indole ring. This activation lowers the energy barrier for the initial C-C bond formation, which is the rate-determining step in the cascade. Following the initial addition, an intramolecular cyclization occurs, closing the piperidine ring and establishing the rigid bicyclic framework. The bulky nature of the sulfonyl group and the specific coordination geometry of the scandium complex play a critical role in directing the facial selectivity of the attack, resulting in the observed high diastereoselectivity (>95:5 dr). This level of stereocontrol is essential for pharmaceutical applications, as different diastereomers can exhibit vastly different biological activities and toxicity profiles.
Furthermore, the choice of solvent and temperature profoundly influences the reaction outcome, with acetonitrile proving to be the optimal medium for stabilizing the charged intermediates involved in the catalytic cycle. The reaction temperature of 70°C provides sufficient thermal energy to overcome the activation barrier without promoting side reactions such as polymerization or decomposition of the sensitive imine substrate. Impurity control is inherently built into this mechanism; the high specificity of the Lewis acid for the imine functionality minimizes non-specific alkylation of the indole nitrogen or other undesired pathways. Consequently, the impurity profile of the crude product is significantly cleaner compared to acid-catalyzed methods using protic acids, which often lead to complex mixtures of regioisomers. This mechanistic clarity allows process chemists to confidently scale the reaction, knowing that the critical quality attributes of the product are governed by well-understood chemical principles rather than empirical trial and error.
How to Synthesize Indole-Derived Piperidine Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory research and pilot plant production. The protocol involves simply mixing the indole methanol and the unsaturated imine in the solvent, adding the catalyst, and heating the mixture. Detailed standard operating procedures for this synthesis, including precise stoichiometry and workup instructions, are provided in the guide below to ensure reproducibility and safety.
- Combine 3-alkyl-2-indole methanol and alpha,beta-unsaturated N-sulfonyl imine derivatives in an organic solvent such as acetonitrile.
- Add 30 mol% Scandium(III) triflate (Sc(OTf)3) as the Lewis acid catalyst to the reaction mixture.
- Stir the reaction at 70°C until completion monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The shift from multi-step, low-yielding processes to this convergent one-pot strategy fundamentally alters the cost structure of manufacturing these valuable intermediates. By reducing the number of unit operations, the process inherently lowers labor costs, energy consumption, and equipment occupancy time, leading to substantial cost savings in pharmaceutical intermediate manufacturing. The use of commercially available starting materials ensures a stable supply chain, mitigating the risks associated with sourcing exotic or custom-synthesized precursors that often plague complex drug development programs.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in solvent usage per kilogram of product significantly lower the raw material costs. Additionally, the simplified purification process reduces the consumption of silica gel and eluents, which are major cost drivers in chromatographic separations. The high yield and selectivity mean that less starting material is wasted, maximizing the return on investment for every batch produced. This efficiency translates into a more competitive pricing structure for the final API, allowing pharmaceutical companies to bring life-saving treatments to market at a more accessible price point.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a wide range of substituents on both the indole and imine components, ensures consistent production quality even with slight variations in raw material grades. This tolerance reduces the likelihood of batch failures and supply disruptions, providing a reliable source of high-purity pharmaceutical intermediates. The ability to synthesize a diverse array of analogs using the same core protocol allows for rapid response to changing clinical demands, ensuring that the supply chain can adapt quickly to the evolving needs of drug development pipelines without requiring extensive re-validation of new processes.
- Scalability and Environmental Compliance: The mild reaction temperature and the use of relatively benign solvents like acetonitrile align well with environmental, health, and safety (EHS) regulations, simplifying the permitting process for large-scale production facilities. The high atom economy of the reaction minimizes the generation of hazardous waste, reducing the costs associated with waste disposal and environmental remediation. This green chemistry profile not only enhances the corporate sustainability image but also future-proofs the manufacturing process against increasingly stringent global environmental regulations, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these indole-derived piperidine compounds. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a transparent view of the technology's capabilities and limitations for potential partners and stakeholders.
Q: What is the biological activity of these indole-derived piperidine compounds?
A: According to patent CN115785122A, these compounds exhibit high sensitivity and strong cytotoxic activity against human breast cancer cells (MCF-7), indicating significant potential for oncology drug development.
Q: What are the optimal reaction conditions for this synthesis?
A: The optimal conditions involve using Scandium(III) triflate (Sc(OTf)3) as the catalyst in acetonitrile solvent at a temperature of 70°C, achieving high yields and excellent diastereoselectivity (>95:5 dr).
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the process utilizes conventional reaction conditions, mild temperatures, and readily available substrates, making it highly suitable for large-scale industrial production with simplified post-treatment procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole-Derived Piperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this novel synthetic methodology in accelerating oncology drug discovery. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from gram-scale research to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of indole-derived piperidine intermediate meets the highest standards required for clinical trials and commercial distribution. We are committed to being a reliable pharmaceutical intermediate supplier that supports your innovation with unwavering quality and dependability.
We invite you to collaborate with us to leverage this cutting-edge technology for your next drug development program. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your overall development costs.
