Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for High-Value Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for fluorinated heterocycles, particularly 3-trifluoromethyl-substituted 1,2,4-triazoles, which serve as critical scaffolds in modern drug discovery. As highlighted in recent intellectual property developments, specifically Patent CN114920707B, a groundbreaking preparation method has emerged that leverages the dual functionality of N,N-dimethylformamide (DMF). This innovation addresses the persistent challenges in constructing nitrogen-rich heterocyclic cores efficiently. The significance of this chemical motif cannot be overstated, as evidenced by its presence in high-profile therapeutic agents ranging from antidiabetic medications like Sitagliptin to potent Factor IXa inhibitors and GlyT1 inhibitors used in neurological disorders. 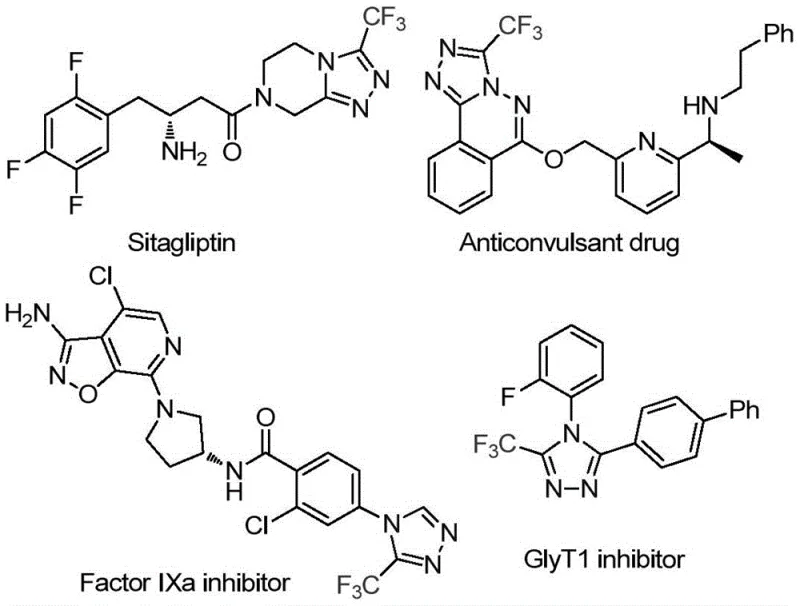 For R&D directors and procurement specialists, understanding this new methodology offers a pathway to more reliable pharmaceutical intermediate supplier partnerships, ensuring that the complex demands of next-generation API manufacturing can be met with greater economic and operational efficiency.
For R&D directors and procurement specialists, understanding this new methodology offers a pathway to more reliable pharmaceutical intermediate supplier partnerships, ensuring that the complex demands of next-generation API manufacturing can be met with greater economic and operational efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of trifluoromethyl-substituted triazoles has been plagued by operational complexities that hinder large-scale production. Conventional routes often necessitate the use of specialized, moisture-sensitive reagents and require stringent anhydrous and anaerobic environments to prevent side reactions or catalyst deactivation. These requirements invariably drive up the cost of goods sold (COGS) due to the need for specialized equipment, inert gases like nitrogen or argon, and highly purified solvents. Furthermore, many existing methods rely on expensive transition metal catalysts or multi-step sequences where the trifluoromethyl group and the triazole ring are installed separately, leading to lower overall yields and increased waste generation. For supply chain heads, these factors translate into longer lead times for high-purity pharmaceutical intermediates and a higher risk of batch-to-batch variability, making the sourcing of these critical building blocks a significant bottleneck in the drug development timeline.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in Patent CN114920707B introduces a remarkably streamlined strategy that utilizes molecular iodine as a promoter and DMF as both the solvent and the carbon source. This approach eliminates the need for exotic reagents or harsh conditions, operating effectively under simple air atmosphere at moderate temperatures between 110°C and 130°C. By integrating the carbon source directly into the solvent matrix, the reaction achieves high atom economy and simplifies the workup procedure significantly. 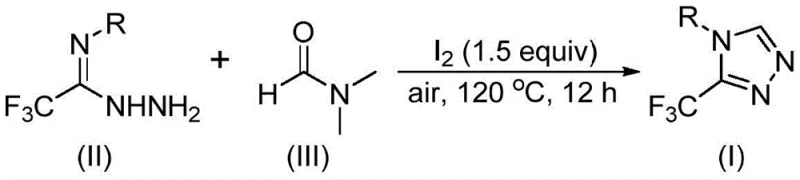 This novel pathway allows for the direct conversion of readily available trifluoroethyliminohydrazides into the desired triazole products with impressive efficiency. For procurement managers, this represents a substantial opportunity for cost reduction in API manufacturing, as it replaces costly specialty chemicals with commodity solvents while simultaneously reducing the environmental footprint associated with solvent disposal and energy consumption.
This novel pathway allows for the direct conversion of readily available trifluoroethyliminohydrazides into the desired triazole products with impressive efficiency. For procurement managers, this represents a substantial opportunity for cost reduction in API manufacturing, as it replaces costly specialty chemicals with commodity solvents while simultaneously reducing the environmental footprint associated with solvent disposal and energy consumption.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic elegance of this transformation lies in the versatile reactivity of DMF under oxidative conditions facilitated by molecular iodine. Detailed analysis suggests that DMF can participate in the reaction through two distinct pathways depending on which functional moiety acts as the carbon synthon. In one pathway, the formyl group of DMF undergoes condensation with the hydrazide substrate to form a hydrazone intermediate, which subsequently cyclizes with the elimination of dimethylamine to yield the triazole core. Alternatively, the N-methyl group of DMF can be activated by iodine to form an amine salt, which then engages in nucleophilic addition followed by oxidative aromatization. This dual-reactivity profile ensures high conversion rates across a broad substrate scope, accommodating various electronic and steric environments on the aromatic ring. For technical teams, understanding this mechanism is crucial for optimizing reaction parameters and troubleshooting potential impurities, ensuring that the final product meets the stringent purity specifications required for clinical applications.
Furthermore, the tolerance of this system to air and moisture is a testament to the robustness of the iodine-mediated activation cycle. Unlike palladium or copper-catalyzed couplings that are sensitive to oxygen quenching, this radical or ionic pathway thrives in the presence of atmospheric oxygen, which may even play a role in the final oxidative aromatization step. This resilience simplifies the engineering controls required for scale-up, allowing the process to be transferred from laboratory glassware to industrial reactors with minimal modification. The ability to synthesize diverse derivatives, including those with electron-withdrawing groups like fluorine and chlorine or electron-donating groups like methoxy and alkyl chains, demonstrates the versatility of this method. Such flexibility is vital for medicinal chemists exploring structure-activity relationships (SAR) who require rapid access to a library of analogues without being constrained by synthetic limitations.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of execution, making it accessible for both laboratory research and commercial production facilities. The protocol involves a straightforward mixing of reagents followed by a thermal treatment period, avoiding the need for slow addition pumps or cryogenic cooling. This simplicity reduces the training burden on operators and minimizes the risk of human error during the manufacturing process, thereby enhancing overall process safety and reliability.
- Combine molecular iodine, trifluoroethyliminohydrazide, and DMF solvent in a reaction vessel under air atmosphere.
- Heat the mixture to 110-130°C and maintain reaction for 10-15 hours to facilitate tandem cyclization.
- Perform post-treatment including filtration, washing, and column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this DMF-based synthesis strategy offers compelling advantages that directly impact the bottom line and supply chain resilience. The shift away from precious metal catalysts and specialized anhydrous solvers towards commodity chemicals creates a more stable and predictable cost structure for long-term supply agreements.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of DMF as a dual-purpose reagent drastically lowers the raw material costs associated with producing these intermediates. By removing the need for rigorous solvent drying and inert gas purging, the process also reduces utility costs and equipment maintenance expenses. This lean manufacturing approach allows suppliers to offer more competitive pricing without compromising on quality, providing significant value to downstream API manufacturers looking to optimize their production budgets.
- Enhanced Supply Chain Reliability: Since the starting materials, including trifluoroethyliminohydrazides and molecular iodine, are commercially available and widely produced, the risk of supply disruption is minimized. The robustness of the reaction conditions means that production can be maintained consistently across different manufacturing sites without the need for highly specialized infrastructure. This reliability ensures that pharmaceutical companies can secure a steady flow of critical intermediates, reducing the risk of project delays caused by material shortages or quality failures.
- Scalability and Environmental Compliance: The simplified post-treatment process, which typically involves filtration and standard chromatography, facilitates easier scale-up from pilot plants to full commercial production. Additionally, the reduced use of hazardous reagents and the potential for solvent recycling align with green chemistry principles, helping manufacturers meet increasingly strict environmental regulations. This sustainability aspect is becoming a key differentiator in vendor selection processes for major multinational corporations committed to reducing their carbon footprint.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis method, we have compiled answers to common inquiries regarding its implementation and benefits. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, ensuring accuracy and relevance for technical decision-makers.
Q: Does this synthesis require strict anhydrous or anaerobic conditions?
A: No, a key advantage of this patented method is that it operates effectively under standard air atmosphere without the need for expensive inert gas protection or rigorous drying of solvents.
Q: What acts as the carbon source in this triazole formation?
A: The organic solvent DMF serves a dual role, acting simultaneously as the reaction medium and the essential carbon synthon (C1 source) for constructing the triazole ring.
Q: Is the purification process complex for these intermediates?
A: The process utilizes standard post-treatment techniques such as filtration and silica gel column chromatography, which are well-established and scalable in industrial settings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in Patent CN114920707B for the production of high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries can be seamlessly translated into reliable industrial supply. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole delivered meets the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this efficient synthesis route for your upcoming projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you accelerate your drug development timeline while optimizing your manufacturing costs.
