Advanced Molybdenum-Copper Catalyzed Route to High-Purity Trifluoromethyl Triazoles for Pharmaceutical Manufacturing
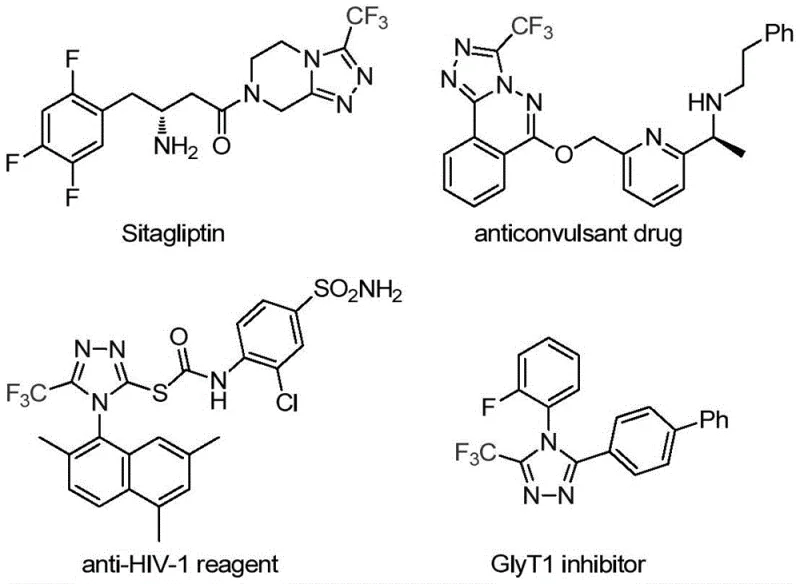
Patent CN113307778A introduces a transformative methodology for synthesizing critical pharmaceutical intermediates specifically targeting the production of three-trifluoromethyl substituted one-two-four-triazole compounds which serve as essential structural motifs in numerous bioactive molecules including blockbuster drugs such as sitagliptin anticonvulsant agents anti-HIV reagents and GlyT1 inhibitors This innovative approach employs a dual catalytic system featuring molybdenum hexacarbonyl and cuprous acetate enabling highly efficient cycloaddition reactions under remarkably mild thermal conditions ranging from seventy to ninety degrees Celsius over eighteen to thirty hours Unlike conventional synthetic routes that frequently demand extreme temperatures or hazardous reagents this patented process utilizes inexpensive commercially accessible starting materials including trifluoroethylimidoyl chloride and functionalized isonitriles The methodology demonstrates exceptional scalability from laboratory gram-scale operations to potential industrial production volumes while maintaining stringent purity specifications required for advanced pharmaceutical applications Furthermore its strategic design permits facile structural diversification through simple modification of the isonitrile component thereby expanding its utility across diverse therapeutic compound libraries representing a significant advancement in fluorinated heterocyclic chemistry with profound implications for drug discovery pipelines
The Limitations of Conventional Methods vs The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic strategies for constructing trifluoromethyl-substituted one-two-four-triazole scaffolds typically involve cyclization reactions between trifluoroacetyl hydrazine and amidine compounds or hydrazinolysis of trifluoromethyl oxazolinones both imposing significant operational constraints including elevated temperatures exceeding one hundred fifty degrees Celsius extended reaction times beyond forty-eight hours that increase energy consumption impurity formation risks and safety hazards Alternative approaches using copper-catalyzed multi-component reactions with diazonium salts often require cryogenic conditions below minus forty degrees Celsius to handle unstable trifluorodiazoethane intermediates creating substantial safety challenges complicating large-scale implementation due to specialized equipment requirements Moreover these established methods frequently exhibit narrow substrate scope with poor tolerance for functional groups commonly present in complex pharmaceutical intermediates necessitating additional protection-deprotection steps that reduce overall yield increase manufacturing costs while failing to provide adequate control over regioselectivity for the critical three-position substitution pattern required in modern drug molecules These cumulative drawbacks severely limit practical utility in commercial pharmaceutical manufacturing where consistent quality cost-effectiveness and regulatory compliance are paramount
The Novel Approach
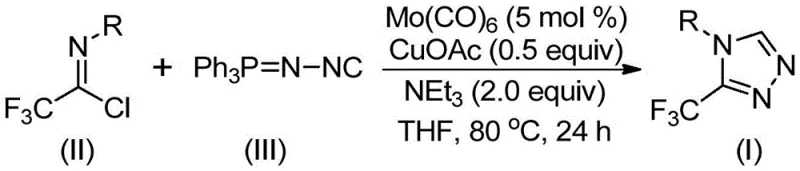
The patented methodology overcomes these challenges through an elegant molybdenum-copper co-catalyzed cycloaddition between trifluoroethylimidoyl chloride and functionalized isonitriles operating under exceptionally mild thermal conditions of seventy to ninety degrees Celsius for eighteen to thirty hours without requiring cryogenic handling or high-pressure equipment This innovative process utilizes readily available catalysts molybdenum hexacarbonyl at only five mol percent loading and cuprous acetate at stoichiometric equivalence both cost-effective commercially abundant compared to precious metal alternatives commonly employed The reaction demonstrates remarkable functional group tolerance across diverse aryl substituents including halogens alkyl groups methoxy functionalities as evidenced by successful synthesis of fifteen distinct derivatives with consistent high conversion rates Crucially it eliminates hazardous diazo compounds unstable intermediates while maintaining excellent regioselectivity for three-position trifluoromethyl substitution Post-reaction workup involves straightforward filtration followed by standard column chromatography purification without specialized metal removal steps significantly streamlining manufacturing while ensuring stringent purity specifications required for pharmaceutical applications
Mechanistic Insights into Molybdenum-Copper Co-Catalyzed Triazole Formation
The reaction mechanism proceeds through a sophisticated dual catalytic cycle where molybdenum hexacarbonyl first activates functionalized isonitrile forming a reactive metal complex that undergoes copper-promoted three-plus-two cycloaddition with trifluoroethylimidoyl chloride This key step generates a five-membered ring intermediate via nucleophilic attack by isonitrile nitrogen on electrophilic carbon of imidoyl chloride moiety facilitated by copper coordination lowering activation energy barriers Subsequent elimination of triphenylphosphine oxide under aqueous conditions drives aromatization yielding final three-trifluoromethyl-substituted triazole product with complete regiocontrol at three-position The synergistic effect between molybdenum copper catalysts proves critical molybdenum enhances isonitrile reactivity while copper accelerates cycloaddition step resulting in significantly improved reaction kinetics compared to single-metal systems This cooperative catalysis enables efficient progression at moderate temperatures without requiring continuous inert atmosphere maintenance throughout reaction duration
Impurity control achieves precision through strict stoichiometric management where optimal ratio of trifluoroethylimidoyl chloride to functionalized isonitrile one-to-one point five minimizes side reactions ensuring complete starting material conversion Molecular sieves effectively scavenge trace moisture preventing hydrolysis of sensitive intermediates while triethylamine functions as both base proton shuttle facilitating key deprotonation steps without promoting unwanted byproducts Mild thermal profile prevents thermal decomposition pathways generating regioisomeric impurities common in high-temperature conventional syntheses Absence of transition metal residues eliminates purification challenges associated with metal-catalyzed reactions ensuring products consistently meet stringent quality standards required for advanced intermediate applications This robust impurity control strategy delivers exceptional purity profiles essential for pharmaceutical manufacturing compliance
How to Synthesize Trifluoromethyl Triazoles Efficiently
This patented methodology provides a streamlined pathway for producing high-purity trifluoromethyl triazoles through carefully optimized dual-metal catalysis addressing longstanding challenges in heterocyclic synthesis The innovative system enables reliable production under practical laboratory conditions while maintaining exceptional selectivity for desired regioisomer critical to pharmaceutical applications By eliminating hazardous reagents extreme temperature requirements found in conventional approaches it significantly enhances operational safety process robustness for scale-up scenarios The following standardized procedure details precise implementation protocol developed through extensive experimental validation ensuring consistent manufacturing outcomes across diverse production environments
- Combine molybdenum hexacarbonyl (5 mol%), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieve, trifluoroethylimidoyl chloride (II), and functionalized isonitrile (III) in THF solvent.
- Heat mixture to 80°C under inert atmosphere with stirring for exactly 24 hours.
- Filter reaction mixture through silica gel followed by column chromatography purification using standard elution protocols.
Commercial Advantages for Procurement and Supply Chain Teams
This advanced synthetic route directly addresses critical pain points faced by procurement supply chain professionals through inherent design features enhancing economic viability operational reliability The methodology's reliance on commercially abundant starting materials eliminates dependency on specialized scarce reagents causing supply chain disruptions in traditional triazole synthesis pathways Operating under mild conditions without cryogenic equipment high-pressure reactors significantly reduces capital expenditure requirements improving facility compatibility across diverse manufacturing sites These fundamental improvements translate into tangible benefits strengthening procurement strategies supply chain resilience for organizations committed to sustainable pharmaceutical intermediate production
- Cost Reduction in Manufacturing: Elimination of expensive transition metal catalysts hazardous reagents substantially lowers raw material expenses while simplifying purification workflows through reduced metal contamination concerns Streamlined approach minimizes solvent consumption during workup procedures decreases energy requirements by avoiding extreme temperature conditions typically needed Consequent reduction in processing steps lowers overall production costs without compromising quality standards required for pharmaceutical applications
- Enhanced Supply Chain Reliability: Broad availability of all starting materials from multiple global suppliers mitigates single-source dependency risks enabling flexible sourcing strategies maintaining consistent production schedules during market fluctuations Process tolerance for minor raw material quality variations ensures stable output regardless of supplier batch differences reducing quality-related delays common in complex heterocyclic syntheses Robustness allows manufacturers to maintain reliable delivery timelines even when facing unexpected supply chain disruptions through alternative sourcing options without requiring process revalidation
- Scalability and Environmental Compliance: Methodology's successful demonstration from gram-scale to potential multi-ton production volumes proves readiness for commercial implementation maintaining consistent product specifications throughout scale-up transitions Absence of toxic byproducts reduced solvent usage aligns with green chemistry principles satisfying increasingly stringent environmental regulations Simplified waste streams lower disposal costs enhance corporate sustainability profiles without requiring additional capital investment in specialized treatment systems supporting global regulatory compliance initiatives
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding this patented synthesis method based on detailed analysis experimental validation data practical implementation considerations for industrial adoption These responses provide essential insights supporting informed decision-making during procurement evaluations manufacturing planning phases
Q: What limitations do conventional methods face in synthesizing trifluoromethyl triazoles?
A: Traditional approaches require harsh conditions exceeding 150°C or cryogenic temperatures below -40°C with hazardous intermediates like diazo compounds causing safety risks and limited substrate scope that complicates pharmaceutical intermediate production.
Q: How does this novel catalytic method improve process efficiency?
A: The molybdenum-copper co-catalyzed system operates at mild temperatures (70–90°C) with exceptional functional group tolerance enabling gram-scale synthesis without specialized equipment while maintaining precise regioselectivity for pharmaceutical applications.
Q: What supply chain benefits does this methodology provide?
A: By utilizing commercially abundant starting materials and eliminating multi-step purification requirements this approach delivers enhanced reliability through simplified logistics and reduced vulnerability to raw material shortages common in traditional triazole synthesis routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazole Supplier
Our patented methodology represents a significant leap forward in producing high-value fluorinated heterocyclic compounds essential for modern pharmaceutical development pipelines NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from one hundred kgs to one hundred MT annual commercial production while maintaining stringent purity specifications through state-of-the-art QC labs equipped with advanced analytical instrumentation Our dedicated technical teams specialize in optimizing complex synthetic routes like this molybdenum-copper co-catalyzed process ensuring seamless technology transfer from laboratory discovery to full-scale manufacturing operations with minimal disruption to development timelines
We invite you to contact our technical procurement team requesting specific COA data route feasibility assessments tailored to requirements through our Customized Cost-Saving Analysis service identifying potential efficiency improvements within current supply chain operations
