Advanced Dichlorotitanocene Catalysis for Efficient Commercial Production of Benzodiazepine Derivatives
Introduction to Next-Generation Benzodiazepine Synthesis
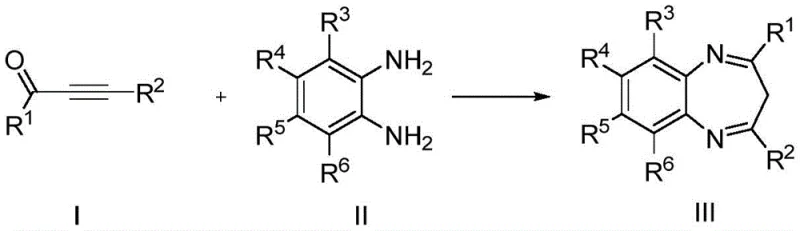
The pharmaceutical industry continuously seeks robust and sustainable methodologies for constructing privileged scaffolds, and the recent disclosure in patent CN110256365B represents a significant leap forward in the synthesis of benzodiazepine derivatives. This intellectual property details a novel catalytic protocol utilizing dichlorotitanocene as a highly efficient promoter for the cyclocondensation of 3-butyn-2-one compounds with o-phenylenediamines. Unlike traditional approaches that often rely on corrosive reagents or energy-intensive conditions, this invention leverages a ligand-accelerated catalytic system that operates under remarkably mild parameters. For R&D directors and process chemists, this technology offers a compelling alternative for generating high-purity intermediates essential for anxiolytic, sedative, and anticonvulsant drug development. The strategic integration of carboxylic acid ligands, such as isophthalic acid, enhances the catalytic turnover, ensuring that the reaction proceeds with exceptional atom economy and minimal byproduct formation. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances is critical for evaluating the long-term viability and cost-effectiveness of adopting this route for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,5-benzodiazepine core has been fraught with synthetic challenges that hinder efficient commercial production. Conventional methodologies typically necessitate the use of harsh reaction environments, including strong mineral acids, potent Lewis acids, or basic conditions that can degrade sensitive functional groups on the substrate. Furthermore, many legacy protocols require microwave irradiation or elevated temperatures to drive the reaction to completion, which not only increases energy consumption but also poses significant safety hazards in a plant setting. The reliance on stoichiometric amounts of expensive metal catalysts in some older methods further exacerbates the cost burden, while the subsequent removal of these metal residues often demands complex purification steps that lower the overall yield. These operational inefficiencies create bottlenecks in the supply chain, leading to extended lead times and inconsistent quality profiles that are unacceptable for modern GMP manufacturing standards. Consequently, there is an urgent demand for a catalytic system that can overcome these thermodynamic and kinetic barriers without compromising on safety or environmental compliance.
The Novel Approach
The methodology described in CN110256365B fundamentally shifts the paradigm by introducing a dichlorotitanocene-catalyzed pathway that operates efficiently at room temperature. This innovative approach utilizes a synergistic combination of a low-loading titanium catalyst and specific organic acid ligands to activate the alkyne and amine functionalities gently yet effectively. By employing ethanol or methanol as the reaction medium, the process aligns perfectly with green chemistry principles, eliminating the need for toxic chlorinated solvents. The reaction demonstrates broad substrate tolerance, accommodating various electron-donating and electron-withdrawing groups on both the alkyne and diamine components without significant loss in efficiency. This versatility allows for the rapid generation of diverse libraries of benzodiazepine analogues, accelerating the drug discovery phase. Moreover, the simplicity of the workup procedure, which often requires only basic column chromatography, streamlines the isolation of the final product, thereby reducing the overall processing time and operational costs associated with purification.
Mechanistic Insights into Dichlorotitanocene-Catalyzed Cyclization
The efficacy of this transformation lies in the unique coordination chemistry of the dichlorotitanocene species when paired with carboxylic acid ligands. Mechanistically, the titanium center acts as a soft Lewis acid that coordinates with the triple bond of the 3-butyn-2-one, increasing its electrophilicity and facilitating nucleophilic attack by the amine groups of the o-phenylenediamine. The presence of ligands like isophthalic acid is believed to stabilize the active catalytic species and prevent the formation of inactive titanium aggregates, thereby maintaining high catalytic activity throughout the reaction cycle. This ligand acceleration effect is crucial for achieving high turnover numbers with catalyst loadings as low as 1% to 3%, which is economically advantageous for large-scale operations. The reaction proceeds through a concerted cyclization pathway that minimizes the formation of oligomeric side products, ensuring a clean impurity profile that simplifies downstream processing. Understanding this mechanistic framework allows process engineers to fine-tune reaction parameters, such as ligand-to-catalyst ratios, to maximize yield and reproducibility across different batches.
Impurity control is another critical aspect where this catalytic system excels, particularly regarding the suppression of polymerization side reactions common in alkyne chemistry. The mild acidic nature of the titanium-ligand complex prevents the excessive protonation of the amine nucleophile, which can otherwise lead to deactivation or non-selective reactions. Additionally, the use of ethanol as a solvent helps to solubilize polar intermediates while keeping the final benzodiazepine product precipitable or easily separable, further enhancing purity. Spectroscopic data from the patent examples confirms the high structural fidelity of the products, with NMR analysis showing sharp signals indicative of well-defined heterocyclic structures free from significant regio-isomers. For quality assurance teams, this level of control translates to reduced testing burdens and higher confidence in batch release specifications. The ability to consistently produce high-purity benzodiazepine derivatives is a key differentiator for suppliers aiming to serve stringent regulatory markets.
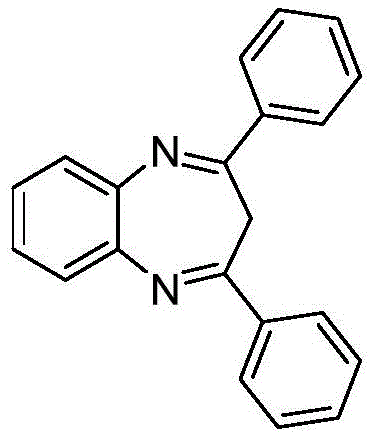
How to Synthesize 2,4-Diphenyl-3-H-1,5-benzodiazepine Efficiently
Implementing this synthesis route requires precise adherence to the optimized molar ratios and reaction conditions outlined in the patent data to ensure maximum efficiency. The process begins with the preparation of the catalytic solution, where dichlorotitanocene and the chosen ligand are dissolved in the alcohol solvent prior to the addition of substrates. Maintaining an inert atmosphere is generally good practice, although the catalyst itself exhibits reasonable stability to air, adding to the operational robustness of the method. Reaction monitoring via TLC or HPLC is recommended to determine the exact endpoint, which typically falls within the 6 to 24-hour window depending on the electronic nature of the substituents. Once the reaction is complete, the solvent is removed under reduced pressure, and the crude residue is subjected to flash chromatography using a petroleum ether and ethyl acetate gradient. Detailed standardized synthesis steps and specific parameter adjustments for different substrates are provided in the technical guide below.
- Combine 3-butyn-2-one compounds and o-phenylenediamine compounds in ethanol solvent with a molar ratio of 1: 1.1 to 1.5.
- Add dichlorotitanocene catalyst (1-3 mol%) and a ligand such as isophthalic acid (2-6 mol%) to the reaction mixture.
- Stir the reaction at room temperature for 6 to 24 hours, then purify the product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this dichlorotitanocene-catalyzed route offers substantial opportunities for cost reduction in API manufacturing without sacrificing quality. The primary economic driver is the replacement of expensive and hazardous reagents with inexpensive, commercially available titanium salts and simple organic acids. Since the catalyst loading is extremely low, the raw material cost per kilogram of product is significantly minimized, directly impacting the bottom line. Furthermore, the elimination of harsh acidic or basic workups reduces the consumption of neutralizing agents and the volume of aqueous waste generated, leading to lower waste disposal fees. The use of ethanol, a green and recyclable solvent, further aligns with sustainability goals and reduces the regulatory burden associated with volatile organic compound (VOC) emissions. These factors collectively contribute to a more resilient and cost-effective supply chain model that can withstand market fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, starting with the drastic reduction in catalyst costs due to the low molar percentage required for effective conversion. By avoiding the use of precious metals like palladium or rhodium, which are subject to volatile market prices, manufacturers can stabilize their production costs over the long term. Additionally, the mild reaction conditions eliminate the need for specialized high-pressure or high-temperature reactors, allowing production to occur in standard glass-lined or stainless steel vessels. This compatibility with existing infrastructure reduces capital expenditure requirements for new facility setups. The simplified purification process also lowers labor and utility costs associated with extended distillation or recrystallization steps, resulting in a leaner manufacturing operation.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the high availability and stability of the key reagents used in this protocol. Dichlorotitanocene and isophthalic acid are bulk chemicals with established global supply networks, minimizing the risk of shortages that often plague specialized catalysts. The robustness of the reaction to ambient conditions means that transportation and storage of intermediates are less critical, reducing logistics complexity. Moreover, the short reaction time of 6 to 24 hours allows for faster batch turnover, enabling suppliers to respond more agilely to sudden spikes in demand from downstream pharmaceutical clients. This agility is crucial for maintaining just-in-time inventory levels and preventing production delays that could impact drug launch timelines.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the absence of exothermic runaway risks associated with strong acid catalysis. The green solvent system facilitates easier solvent recovery and recycling, which is increasingly important for meeting stringent environmental regulations in major manufacturing hubs. The high atom economy of the reaction ensures that the majority of the starting material mass is incorporated into the final product, minimizing the generation of hazardous byproducts. This environmental friendliness not only reduces the carbon footprint of the manufacturing process but also enhances the corporate social responsibility profile of the supplier, making it a more attractive partner for eco-conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology in industrial settings. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a transparent view of the process capabilities. Understanding these details helps stakeholders make informed decisions about integrating this method into their existing production portfolios. The answers reflect the consensus on performance metrics such as yield, purity, and operational safety observed during the validation studies.
Q: What are the advantages of using dichlorotitanocene over traditional acid catalysts?
A: Dichlorotitanocene offers mild reaction conditions at room temperature, avoiding the harsh strong acids or bases required by conventional methods, which significantly reduces equipment corrosion and safety risks while improving atom economy.
Q: Which ligands are most effective for this catalytic system?
A: Isophthalic acid is the preferred ligand for optimal yield, though 2-hydroxynicotinic acid and salicylic acid are also effective alternatives that promote the cyclization efficiency.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process uses inexpensive, air-stable catalysts and green solvents like ethanol, making it highly suitable for commercial scale-up with simplified downstream processing and waste treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzodiazepine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the dichlorotitanocene-catalyzed synthesis route for producing high-value benzodiazepine intermediates. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch. By leveraging this advanced catalytic technology, we can offer our partners a competitive edge through superior product quality and consistent supply reliability. Our dedication to green chemistry and process optimization aligns perfectly with the evolving needs of the global pharmaceutical industry.
We invite you to collaborate with us to explore how this efficient synthesis method can enhance your drug development pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate the viability of this approach for your projects. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing and driving your success in the marketplace.
