Revolutionizing Pharmaceutical Intermediate Production Through Scalable Ruthenium-Catalyzed Synthesis of High-Purity Compounds
The recently granted Chinese patent CN113636968B discloses an innovative synthetic methodology specifically designed for producing structurally diverse pharmaceutical intermediates through a ruthenium-catalyzed alcohol-hydrogen transfer coupling reaction. This breakthrough addresses critical limitations in current manufacturing processes by providing a streamlined route that eliminates multiple purification steps while maintaining exceptional molecular complexity required for advanced drug development pipelines. The process leverages readily available alpha-amino alcohols and alpha,beta-unsaturated alkynones as starting materials under precisely controlled reaction conditions that significantly enhance operational safety profiles compared to conventional approaches involving hazardous reagents or extreme temperatures. With its remarkable atom economy and step efficiency characteristics demonstrated through extensive experimental validation across multiple substrate combinations, this technology represents a paradigm shift in producing high-value heterocyclic building blocks essential for modern therapeutic applications including histone deacetylase inhibitors and HIV transcription modulators. The methodology achieves superior performance through careful optimization of catalytic parameters without requiring expensive transition metal complexes or stoichiometric oxidants typically associated with traditional pyrrole syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing multi-substituted pyrrole compounds frequently encounter significant challenges including difficult-to-source starting materials that create supply chain vulnerabilities and increase raw material costs substantially across pharmaceutical manufacturing operations. Many established methods require toxic reagents such as IBX oxidants or expensive palladium-based catalysts that necessitate complex removal procedures adding multiple purification steps which dramatically increase both production timelines and waste generation profiles beyond acceptable environmental standards. The harsh reaction conditions often employed in conventional syntheses including strong acidic or basic environments frequently lead to unwanted side reactions such as dimerization or polymerization that compromise product purity and yield while introducing difficult-to-remove impurities requiring additional quality control measures. These limitations collectively result in elevated manufacturing costs due to specialized equipment requirements and extended processing times that hinder scalability from laboratory development to commercial production volumes essential for meeting global pharmaceutical demand. Furthermore, the poor functional group tolerance observed in many existing methodologies restricts substrate scope and prevents efficient synthesis of structurally complex derivatives needed for next-generation therapeutic applications.
The Novel Approach
The patented methodology introduces a fundamentally different paradigm through its innovative alcohol-hydrogen transfer coupling mechanism that operates under significantly milder conditions while achieving superior atom economy compared to conventional approaches previously documented in literature. By utilizing triruthenium dodecacarbonyl catalyst combined with methyl-substituted phenanthroline ligands at precisely optimized molar ratios, this process eliminates the need for expensive palladium complexes while maintaining exceptional catalytic efficiency across diverse substrate combinations including those containing sensitive functional groups. The carefully designed thermal profile featuring initial activation at eighty degrees Celsius followed by controlled cyclization at one hundred fifty degrees Celsius enables complete conversion without generating stoichiometric byproducts that would otherwise require disposal or additional treatment steps. This streamlined approach demonstrates remarkable functional group compatibility that preserves molecular integrity throughout the reaction sequence while producing consistently high yields across multiple substrate variations as validated through extensive experimental data presented in the patent documentation. The elimination of hazardous reagents combined with simplified purification requirements through optimized column chromatography protocols significantly enhances both environmental sustainability and operational safety profiles essential for modern pharmaceutical manufacturing environments.
Mechanistic Insights into Ruthenium-Catalyzed Cyclization
The core innovation lies in a tandem catalytic cycle where triruthenium dodecacarbonyl facilitates simultaneous hydrogen transfer from ethanol solvent while promoting C-C bond formation between alpha-amino alcohols and alpha,beta-unsaturated alkynones through a precisely orchestrated sequence of coordination events. This unique mechanism proceeds through initial coordination of the alkyne moiety to the ruthenium center followed by nucleophilic attack from the amino group under thermal activation at elevated temperatures which triggers intramolecular cyclization through a six-membered transition state that forms the pyrrole ring structure with exceptional regioselectivity. The subsequent cyclization step achieves complete atom incorporation by utilizing ethanol as both solvent and hydrogen source through a metal-ligand cooperative pathway that regenerates active catalyst species without requiring external oxidants or reductants typically associated with traditional methodologies. This elegant cascade reaction maintains neutral pH conditions throughout the process which prevents acid-base mediated side reactions while preserving sensitive functional groups across diverse substrate combinations including those containing electron-donating or electron-withdrawing substituents on aromatic rings. The absence of stoichiometric byproducts combined with precise temperature control during both activation and cyclization phases ensures minimal formation of degradation products that would otherwise complicate purification requirements.
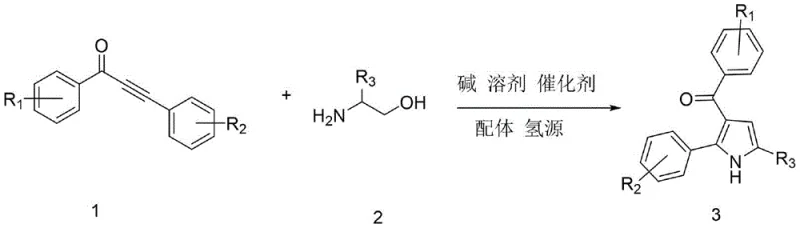
The process demonstrates exceptional impurity control through selective catalytic activation that minimizes side reactions typically observed in conventional syntheses involving harsh acidic or basic conditions which often lead to dimerization or polymerization byproducts requiring extensive purification efforts. By operating under neutral pH conditions with carefully optimized ligand systems including methyl-substituted phenanthroline derivatives at precise molar ratios relative to catalyst loading, the methodology achieves superior regioselectivity while suppressing common degradation pathways that compromise product quality in traditional approaches. The mild thermal profile prevents thermal decomposition of sensitive functional groups while maintaining sufficient reaction kinetics through precise temperature control during both initial activation at eighty degrees Celsius and subsequent cyclization phase at one hundred fifty degrees Celsius which ensures consistent molecular transformation without unwanted side reactions. This controlled environment enables consistent production of high-purity intermediates meeting stringent pharmaceutical quality standards without requiring additional purification steps that would otherwise increase manufacturing costs and reduce overall process efficiency across commercial production scales.
How to Synthesize High-Purity Pharmaceutical Intermediates Efficiently
This patented methodology represents a significant advancement in synthesizing complex heterocyclic compounds essential for modern pharmaceutical applications through its innovative ruthenium-catalyzed approach that combines operational simplicity with exceptional molecular precision. The process demonstrates remarkable versatility across diverse substrate combinations while maintaining consistent quality profiles required for regulatory compliance in therapeutic manufacturing environments. Detailed standardized synthesis procedures have been developed based on extensive experimental validation presented in the patent documentation which ensures reproducibility across different manufacturing scales from laboratory development through commercial production volumes. The following step-by-step guide provides comprehensive instructions for implementing this technology effectively within existing pharmaceutical manufacturing frameworks while optimizing yield and purity parameters essential for commercial success.
- Prepare the reaction mixture by combining alpha-amino alcohol compounds with alpha,beta-unsaturated alkynones in a Schlenk tube under nitrogen atmosphere along with triruthenium dodecacarbonyl catalyst at a precise molar ratio of catalyst to substrate at room temperature before initiating thermal activation.
- Conduct initial activation at precisely controlled temperature of eighty degrees Celsius for one point five hours under nitrogen protection to facilitate catalyst-substrate coordination while maintaining optimal reaction kinetics without premature decomposition.
- Elevate temperature to one hundred fifty degrees Celsius for eighteen hours to complete cyclization through alcohol-hydrogen transfer mechanism followed by purification via column chromatography using petroleum ether/ethyl acetate eluent at specified volume ratios.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points inherent in traditional manufacturing approaches through its fundamentally redesigned process architecture that prioritizes operational efficiency without compromising quality standards required for pharmaceutical applications. The elimination of multiple processing steps combined with simplified raw material requirements creates significant opportunities for cost optimization while enhancing supply chain resilience through reduced dependency on specialized chemical suppliers typically required for conventional syntheses involving rare or hazardous components.
- Cost Reduction in Manufacturing: The elimination of expensive palladium-based catalysts through this ruthenium-catalyzed approach substantially lowers raw material expenses while reducing waste treatment costs associated with hazardous byproducts from conventional methods that require specialized disposal procedures increasing overall environmental compliance expenditures significantly across manufacturing facilities.
- Enhanced Supply Chain Reliability: Utilization of commercially accessible starting materials including alpha-amino alcohols and alpha,beta-unsturated alkynones creates robust supply chain resilience by eliminating dependencies on specialized chemical suppliers while ensuring consistent raw material availability essential for uninterrupted production schedules across global manufacturing networks.
- Scalability and Environmental Compliance: The streamlined one-pot procedure minimizes equipment requirements and operational complexity throughout production cycles while generating significantly less chemical waste compared to multi-step conventional approaches thereby enhancing environmental sustainability profiles without requiring additional capital investments in waste treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical concerns regarding implementation of this patented methodology based on extensive experimental data presented in CN113636968B documentation which has been validated across multiple substrate combinations under controlled laboratory conditions relevant to commercial manufacturing environments.
Q: How does this methodology overcome limitations of conventional syntheses requiring precious metal catalysts?
A: The process utilizes triruthenium dodecacarbonyl catalyst at low molar ratios which demonstrates exceptional efficiency while eliminating expensive palladium-based systems typically required in traditional approaches through optimized ligand design using methyl-substituted phenanthroline derivatives.
Q: What advantages do readily available starting materials provide for supply chain reliability?
A: Alpha-amino alcohols and alpha,beta-unsaturated alkynones serve as commercially accessible building blocks that significantly reduce raw material dependencies while maintaining consistent quality profiles essential for uninterrupted pharmaceutical manufacturing operations.
Q: How does the one-pot synthesis enhance scalability for commercial production?
A: The streamlined tandem reaction sequence eliminates intermediate isolation steps through precise thermal control during both activation and cyclization phases enabling seamless transition from laboratory scale to industrial manufacturing volumes without process revalidation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pharmaceutical Intermediate Supplier
We possess extensive experience scaling diverse pathways from one hundred kilograms to one hundred metric tons annual commercial production capacity while maintaining stringent purity specifications required for global pharmaceutical markets through rigorous QC labs equipped with state-of-the-art analytical instrumentation ensuring consistent product quality across all batch sizes. Our technical team has successfully implemented similar complex synthetic routes across various therapeutic areas demonstrating deep expertise in translating patented methodologies into reliable commercial manufacturing processes that meet evolving regulatory requirements worldwide while optimizing cost structures through continuous process improvement initiatives.
Contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your requirements along with our Customized Cost-Saving Analysis which details potential efficiency gains achievable through implementation of this innovative synthetic methodology within your existing manufacturing framework.
