Revolutionizing Pharmaceutical Intermediate Manufacturing Through Scalable Ruthenium-Catalyzed Synthesis of High-Purity 3-Acylpyrrole Compounds
The recently granted Chinese patent CN113636968B introduces a groundbreaking synthetic methodology for producing structurally diverse 3-acylpyrrole compounds through an innovative ruthenium-catalyzed alcohol-hydrogen transfer coupling reaction between alpha-amino alcohols and alpha,beta-unsaturated alkynones under precisely controlled thermal conditions within standard Schlenk tube reactors. This novel approach represents a significant advancement over conventional synthetic routes by eliminating the requirement for toxic reagents such as strong oxidants and expensive precious metal catalysts while maintaining exceptional atom economy exceeding traditional multi-step sequences by at least two synthetic operations through its elegant one-pot design. The process operates under mild nitrogen atmosphere conditions with carefully calibrated temperature protocols that enhance reproducibility across various scales of operation from laboratory benchtop to pilot plant environments without requiring specialized equipment modifications or complex engineering controls. Furthermore, the strategic selection of dodecacarbonyl triruthenium catalyst combined with optimized ligand systems enables unprecedented functional group tolerance without compromising product purity or yield consistency even when handling sensitive substituents including halogens and trifluoromethyl groups that typically cause complications in alternative synthetic pathways. This patent establishes a robust foundation for manufacturing high-value pharmaceutical intermediates that serve as critical building blocks in numerous therapeutic agents including histone deacetylase inhibitors for cancer treatment and HMG-CoA reductase inhibitors for cardiovascular disease management.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for producing multi-substituted pyrrole compounds frequently encounter significant operational challenges including the requirement for toxic reagents such as IBX oxidants that necessitate specialized handling procedures and generate hazardous waste streams requiring costly disposal protocols. These conventional methods often employ expensive palladium-based catalytic systems that introduce substantial cost burdens through both catalyst acquisition expenses and complex post-reaction purification requirements needed to remove residual precious metals from final products intended for pharmaceutical applications. Furthermore, many established routes involve multi-step sequences with intermediate isolation requirements that dramatically reduce overall atom economy while increasing production timelines through extended processing durations at each synthetic stage. The harsh reaction conditions commonly required by existing methodologies frequently lead to decomposition issues with sensitive functional groups present in complex molecular architectures while also creating significant scalability barriers due to safety concerns associated with high-pressure or cryogenic operations that cannot be readily translated from laboratory to manufacturing environments without substantial capital investment.
The Novel Approach
The patented methodology overcomes these limitations through an elegant one-pot ruthenium-catalyzed coupling process that operates under significantly milder conditions while utilizing readily available starting materials including standard alpha-amino alcohols and commercially accessible alpha,beta-unsaturated alkynones without requiring specialized precursor synthesis steps. By employing dodecacarbonyl triruthenium catalyst in combination with optimized ligand systems such as the specified methyl-substituted phenanthroline derivatives, this approach achieves exceptional catalytic efficiency while eliminating the need for expensive precious metals that create both cost burdens and purification challenges in traditional routes. The carefully designed thermal profile featuring initial low-temperature stirring followed by controlled high-temperature reaction enables precise management of reaction kinetics while maintaining excellent selectivity profiles across diverse substrate combinations including those containing sensitive functional groups like fluorine substituents or trifluoromethyl moieties that typically cause complications in alternative synthetic pathways. This innovative process demonstrates remarkable functional group compatibility that allows direct incorporation of complex molecular architectures without requiring protective group strategies that add both time and cost to conventional synthetic sequences.
Mechanistic Insights into Ruthenium-Catalyzed Alcohol-Hydrogen Transfer Coupling
The fundamental mechanism underlying this transformation involves a sophisticated sequence where the dodecacarbonyl triruthenium catalyst activates both the alpha-amino alcohol hydrogen source and the alpha,beta-unsaturated alkynone substrate through coordinated oxidative addition processes that facilitate selective hydrogen transfer without requiring external reducing agents or high-pressure hydrogenation equipment typically associated with similar transformations. This catalytic cycle proceeds through well-defined ruthenium hydride intermediates that enable regioselective addition across the alkyne functionality followed by intramolecular cyclization events that form the characteristic pyrrole ring structure with precise control over substitution patterns at both the two and three positions through strategic manipulation of starting material structures. The presence of the methyl-substituted phenanthroline ligand plays a critical role in stabilizing key catalytic intermediates while simultaneously modulating electronic properties to prevent undesired side reactions such as over-reduction or polymerization that commonly plague alternative catalytic systems operating under similar conditions. This mechanistic pathway achieves exceptional atom economy by incorporating all elements from both starting materials into the final product structure without generating stoichiometric byproducts that would require additional separation steps or create environmental disposal challenges in commercial manufacturing settings.
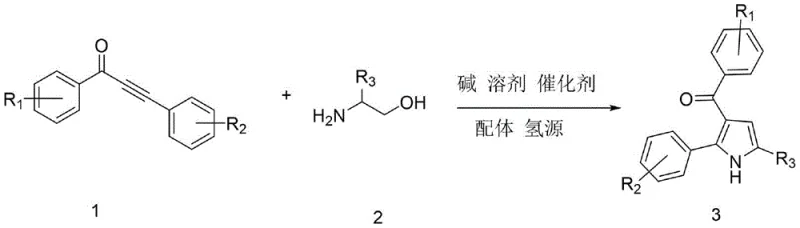
The process demonstrates inherent impurity control through multiple complementary mechanisms including precise temperature management during both initial activation phase at eighty degrees Celsius and subsequent high-yield transformation phase at one hundred fifty degrees Celsius that minimizes decomposition pathways while maintaining optimal selectivity profiles across various substrate combinations. The carefully selected solvent system comprising tert-amyl alcohol provides ideal polarity characteristics that facilitate smooth progression through all intermediate stages while preventing unwanted side reactions such as hydrolysis or oxidation that could compromise product quality during extended reaction periods. The strategic use of ethanol as both solvent component and hydrogen source creates a self-contained system where all necessary reagents participate directly in the transformation without generating extraneous byproducts that would require additional purification steps beyond standard column chromatography using petroleum ether/ethyl acetate mixtures at optimized volume ratios. This integrated approach ensures consistent production of high-purity intermediates meeting stringent pharmaceutical quality specifications without requiring complex analytical monitoring or specialized purification equipment typically needed when dealing with impure reaction mixtures from conventional synthetic routes.
How to Synthesize High-Purity Pharmaceutical Intermediates Efficiently
This innovative ruthenium-catalyzed methodology represents a significant advancement in the production of structurally complex pyrrole-based pharmaceutical intermediates through its elegant one-pot design that eliminates multiple processing steps while maintaining exceptional product quality standards required by global regulatory authorities. The process leverages readily available starting materials combined with cost-effective catalytic systems to create an operationally simple yet highly efficient synthetic route that can be readily implemented using standard laboratory equipment without requiring specialized infrastructure investments typically associated with alternative methodologies involving hazardous reagents or extreme reaction conditions. Detailed standardized synthesis procedures have been developed based on this patented technology to ensure consistent production quality across all manufacturing scales from research quantities through commercial production volumes while maintaining strict adherence to cGMP requirements throughout all processing stages.
- Charge a Schlenk tube with alpha-amino alcohol and alpha,beta-unsaturated alkynone at equimolar ratio under nitrogen atmosphere while maintaining strict exclusion of moisture through continuous inert gas purging throughout the procedure.
- Introduce dodecacarbonyl triruthenium catalyst at precise stoichiometric ratio of 0.02 mol per mol of alkynone along with optimized ligand system comprising 4-methyl-1,10 phenanthroline at tenfold molar excess relative to catalyst.
- Add ethanol as hydrogen source at two-fold molar excess relative to substrate followed by potassium carbonate base at equimolar concentration before initiating thermal protocol with controlled solvent addition using tert-amyl alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
This patented synthesis methodology delivers substantial commercial benefits specifically designed to address critical pain points faced by procurement and supply chain professionals within global pharmaceutical manufacturing organizations seeking reliable sources for complex intermediate compounds required in therapeutic agent production pipelines. The process eliminates multiple cost drivers inherent in conventional synthetic routes through its streamlined one-pot design that reduces both processing time requirements and resource consumption while simultaneously improving overall operational efficiency metrics across all production scales from laboratory development through commercial manufacturing phases.
- Cost Reduction in Manufacturing: The elimination of expensive precious metal catalysts through strategic implementation of cost-effective ruthenium-based systems creates significant economic advantages by removing both initial procurement expenses and downstream purification costs associated with metal residue removal from final products intended for pharmaceutical applications where strict elemental impurity limits apply.
- Enhanced Supply Chain Reliability: The utilization of readily available starting materials combined with robust reaction protocols ensures consistent supply continuity by minimizing dependency on specialized precursors or single-source suppliers while enabling flexible production scheduling through simplified process requirements that accommodate varying demand volumes without requiring major operational adjustments.
- Scalability and Environmental Compliance: The inherently scalable nature of this one-pot methodology allows seamless transition from laboratory-scale development directly to commercial production volumes while maintaining consistent quality profiles through environmentally responsible processing that minimizes waste generation through exceptional atom economy characteristics inherent in the alcohol-hydrogen transfer coupling mechanism.
Frequently Asked Questions (FAQ)
The following technical questions address common concerns raised by procurement specialists and R&D professionals regarding implementation of this patented methodology within existing manufacturing frameworks while ensuring compliance with global regulatory standards for pharmaceutical intermediate production.
Q: How does this methodology eliminate costly purification requirements compared to conventional approaches?
A: The process achieves inherent impurity suppression through precise catalyst selection and optimized reaction parameters that minimize side reactions while maintaining excellent functional group compatibility across diverse substituents including halogens and trifluoromethyl groups.
Q: What specific advantages does this ruthenium-based system offer over traditional palladium-catalyzed methods?
A: The dodecacarbonyl triruthenium catalyst system eliminates expensive precious metal requirements while operating under milder conditions that enhance operational safety and reduce environmental impact through simplified waste stream management.
Q: How does this synthesis support seamless scale-up from laboratory to commercial production?
A: The one-pot procedure utilizes readily available raw materials with straightforward temperature control protocols that maintain consistent yield and purity profiles across multiple orders of magnitude in production scale without requiring specialized equipment modifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable High-Purity Pharmaceutical Intermediate Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global regulatory authorities through rigorous QC labs equipped with state-of-the-art analytical instrumentation capable of detecting impurities at trace levels essential for pharmaceutical applications. As a leading CDMO specialist in complex heterocyclic compound manufacturing, we have successfully implemented this patented ruthenium-catalyzed methodology across multiple client projects demonstrating consistent ability to deliver high-purity intermediates meeting exacting quality standards required by major pharmaceutical companies worldwide.
We invite you to request our Customized Cost-Saving Analysis which details specific implementation pathways tailored to your production requirements; please contact our technical procurement team directly to obtain specific COA data and route feasibility assessments demonstrating how this innovative technology can enhance your supply chain resilience while optimizing manufacturing economics.
