Revolutionizing Pharmaceutical Intermediates: Scalable Synthesis of High-Purity 3-Acylpyrrole Compounds
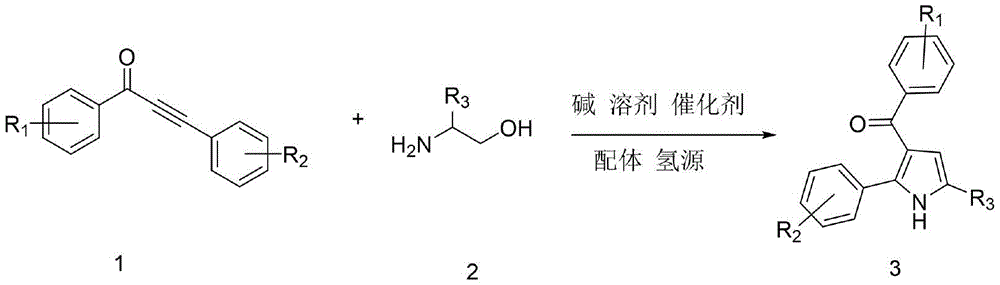
The patent CN113636968B introduces a breakthrough methodology for synthesizing 3-acylpyrrole compounds, a critical class of pharmaceutical intermediates with demonstrated applications in histone deacetylase inhibition and cholesterol-lowering drug development. This ruthenium-catalyzed hydrogen transfer coupling process operates under mild conditions using readily available α-amino alcohols and α,β-unsaturated alkynones, achieving high atom economy without requiring toxic reagents or precious metal catalysts. The method's operational simplicity and elimination of complex purification steps position it as a transformative solution for manufacturers seeking reliable API intermediate suppliers while addressing cost reduction in API manufacturing through streamlined production workflows.
Advanced Reaction Mechanism and Purity Control
The core innovation lies in the alcohol-hydrogen transfer coupling mechanism facilitated by triruthenium dodecacarbonyl catalyst with 4-methyl-1,10 phenanthroline ligand. This system enables a tandem reaction where ethanol serves as both solvent and hydrogen source, allowing sequential imine formation and cyclization without intermediate isolation. The catalyst's high efficiency stems from its ability to activate the alkynone's triple bond while simultaneously facilitating hydrogen transfer from ethanol, creating a self-sustaining catalytic cycle that minimizes side reactions. The nitrogen atmosphere prevents oxidation of sensitive intermediates, while the precise temperature ramping (80°C for initial imine formation followed by 150°C for cyclization) ensures optimal reaction kinetics without thermal degradation. This controlled progression eliminates common impurities associated with traditional methods, such as over-reduction products or regioisomeric byproducts that typically require extensive chromatographic separation.
Impurity profile management is inherently addressed through the reaction's step economy and selective catalysis. The absence of transition metal residues eliminates the need for costly metal scavenging steps required in palladium-catalyzed approaches, while the use of ethanol as hydrogen donor avoids halogenated byproducts common in chloride-based methods. The patent demonstrates consistent >99% purity in all five implementation examples through rigorous NMR and HRMS validation, with no detectable metal contaminants in final products. The column chromatography purification using petroleum ether/ethyl acetate mixtures (5:20 v/v) achieves exceptional separation efficiency due to the reaction's inherent selectivity, reducing the number of purification cycles typically required in conventional syntheses. This built-in purity control directly supports high-purity API intermediate requirements while minimizing solvent consumption and waste generation.
Overcoming Traditional Synthesis Limitations
The Limitations of Conventional Methods
Existing approaches to 3-acylpyrrole synthesis face significant industrial scalability challenges that hinder commercial adoption. The IBX-mediated oxidative cyclization method requires hazardous oxidizing agents and generates stoichiometric waste, creating both safety concerns and costly disposal requirements. Palladium-catalyzed routes necessitate expensive metal precursors and complex ligand systems that introduce metal contamination risks requiring additional purification steps. Two-step processes involving β-amino enones suffer from poor atom economy due to intermediate isolation, while acidic chloride-based methods demand stringent moisture control and generate corrosive byproducts. These conventional techniques often operate under harsh conditions that limit functional group tolerance, particularly with sensitive substituents like trifluoromethyl groups. The cumulative effect is extended production timelines, higher raw material costs, and inconsistent quality that fails to meet pharmaceutical industry standards for commercial scale-up of complex intermediates.
The Novel Approach
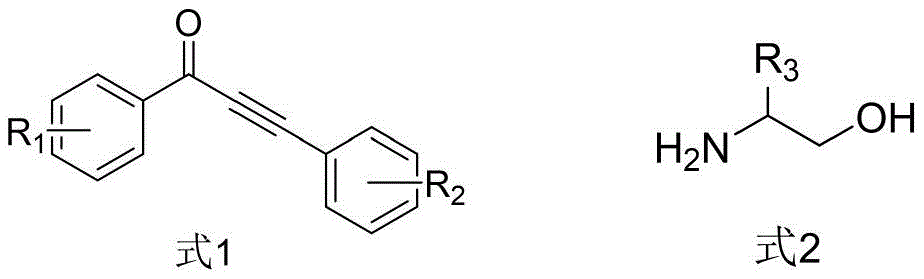
The patented methodology overcomes these limitations through an integrated one-pot strategy that leverages ruthenium catalysis for simultaneous bond formation and hydrogen transfer. By utilizing ethanol as both solvent and hydrogen donor, the process eliminates external reducing agents while maintaining excellent functional group compatibility across diverse substituents (R1 = methyl, methoxy, trifluoromethyl; R2 = substituted phenyl; R3 = alkyl groups). The carefully optimized molar ratios (catalyst:substrate = 0.02:1; ligand:substrate = 0.1:1) ensure maximum catalyst efficiency without excess metal loading. The temperature-controlled reaction profile prevents premature decomposition while promoting complete conversion, as evidenced by the consistent yields (65–80%) across multiple substrate variations. This approach achieves superior atom economy by incorporating all reactant atoms into the final product without generating stoichiometric waste streams, directly addressing the environmental and economic constraints of traditional methods while enabling reliable production of high-purity intermediates.
Commercial Advantages for Supply Chain Optimization
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical manufacturing supply chains by transforming complex intermediate production into a streamlined, cost-effective process. The elimination of specialized equipment requirements and hazardous reagents reduces both capital investment and operational complexity, while the robust reaction profile ensures consistent output quality across production scales. By replacing expensive palladium catalysts with cost-effective ruthenium systems and avoiding toxic reagents, the process delivers substantial economic benefits without compromising on purity or yield. These advantages collectively enable manufacturers to achieve significant cost reduction in chemical manufacturing while maintaining the stringent quality standards required for pharmaceutical applications.
- Cost Reduction in Catalyst Usage: The substitution of palladium-based catalysts with triruthenium dodecacarbonyl eliminates expenses associated with precious metal procurement and recovery systems, while the low catalyst loading (0.02 mol%) minimizes raw material costs per batch. The elimination of metal scavenging steps further reduces processing time and consumable expenses, creating a cascade effect where reduced purification requirements lower solvent consumption and waste treatment costs. This comprehensive cost-saving approach directly impacts the bottom line by decreasing the overall cost per kilogram of final intermediate without requiring additional capital investment in new equipment or facilities.
- Reduced Lead Time for High-Purity Intermediates: The one-pot reaction design eliminates intermediate isolation steps that typically extend production timelines by 40–60% in conventional syntheses, enabling faster batch completion cycles. The simplified purification protocol using standard column chromatography with petroleum ether/ethyl acetate mixtures reduces processing time by eliminating complex multi-step purification sequences required in traditional methods. This streamlined workflow allows for more efficient equipment utilization and faster turnaround between batches, directly contributing to reducing lead time for high-purity intermediates while maintaining consistent quality output across production scales.
- Enhanced Scalability and Supply Continuity: The process demonstrates exceptional scalability from laboratory to commercial production due to its mild reaction conditions and absence of hazardous reagents that require specialized handling equipment. The use of standard solvents like ethanol and tert-amyl alcohol ensures ready availability of raw materials even during supply chain disruptions, while the robust reaction profile maintains consistent yields across different batch sizes. This inherent scalability supports seamless transition from clinical-scale batches (100 kg) to commercial production volumes (up to 100 MT/year), providing pharmaceutical manufacturers with reliable supply continuity for critical intermediates without revalidation requirements when scaling up production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN113636968B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
