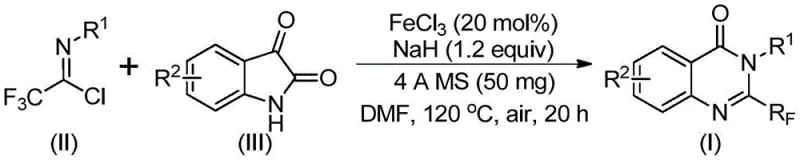
Scalable Production of 2-Trifluoromethyl Quinazolinones via Iron-Catalyzed Cyclization for Global Pharmaceutical Supply Chains
Scalable Production of 2-Trifluoromethyl Quinazolinones via Iron-Catalyzed Cyclization for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. A recent breakthrough detailed in patent CN111675662B introduces a highly efficient preparation method for 2-trifluoromethyl substituted quinazolinone compounds, a structural motif critical for developing next-generation anticancer and anti-inflammatory agents. This innovation addresses long-standing challenges in heterocyclic chemistry by leveraging earth-abundant iron catalysis to construct complex fused ring systems with exceptional precision. By shifting away from precious metal catalysts, this technology not only enhances the economic viability of producing these key intermediates but also aligns with modern green chemistry principles demanded by regulatory bodies worldwide. The method ensures high purity and structural integrity, making it an ideal candidate for integration into the supply chains of multinational pharmaceutical corporations seeking reliable sources for advanced drug scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted quinazolinones has relied heavily on cyclization reactions involving expensive and often hazardous synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate reacting with anthranilamides or isatoic anhydrides. These traditional pathways are frequently plagued by severe reaction conditions that require stringent anhydrous environments or cryogenic temperatures, leading to operational complexities and increased energy consumption. Furthermore, the substrate scope in conventional methods is often narrow, failing to accommodate sensitive functional groups which limits the structural diversity available for medicinal chemistry optimization. The reliance on costly reagents and the generation of significant chemical waste during purification processes further exacerbate the environmental footprint and overall production costs, creating bottlenecks for manufacturers aiming to scale these intermediates for commercial API production.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes readily available trifluoroethylimidoyl chloride and isatin derivatives as starting materials, catalyzed by inexpensive ferric chloride. This approach fundamentally simplifies the synthetic landscape by employing a tandem decarbonylation and cyclization strategy that proceeds efficiently under aerobic conditions. The use of sodium hydride as a base promoter facilitates the initial carbon-nitrogen bond formation, while the iron catalyst drives the subsequent ring closure with remarkable selectivity. This route eliminates the need for exotic reagents and allows for a broader range of substituents on the aromatic rings, thereby expanding the chemical space accessible to drug designers. The operational simplicity, combined with the use of common organic solvents like DMF, makes this process inherently more adaptable to existing manufacturing infrastructure without requiring specialized equipment upgrades.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the iron catalyst. The reaction initiates with an alkali-promoted nucleophilic attack where the nitrogen of the isatin derivative attacks the electrophilic carbon of the trifluoroethylimidoyl chloride, forming a transient trifluoroacetamidine intermediate. Subsequently, the ferric chloride species coordinates with the carbonyl oxygen, activating the system for a decisive decarbonylation step that releases carbon monoxide and drives the equilibrium toward the formation of the quinazolinone core. This iron-mediated cycle is crucial as it lowers the activation energy for the ring closure, allowing the reaction to proceed at moderate temperatures compared to thermal cyclizations. The presence of 4A molecular sieves in the reaction mixture plays a pivotal role in scavenging trace moisture, which protects the sensitive imidoyl chloride reactant from hydrolysis and ensures high conversion rates throughout the extended reaction period.
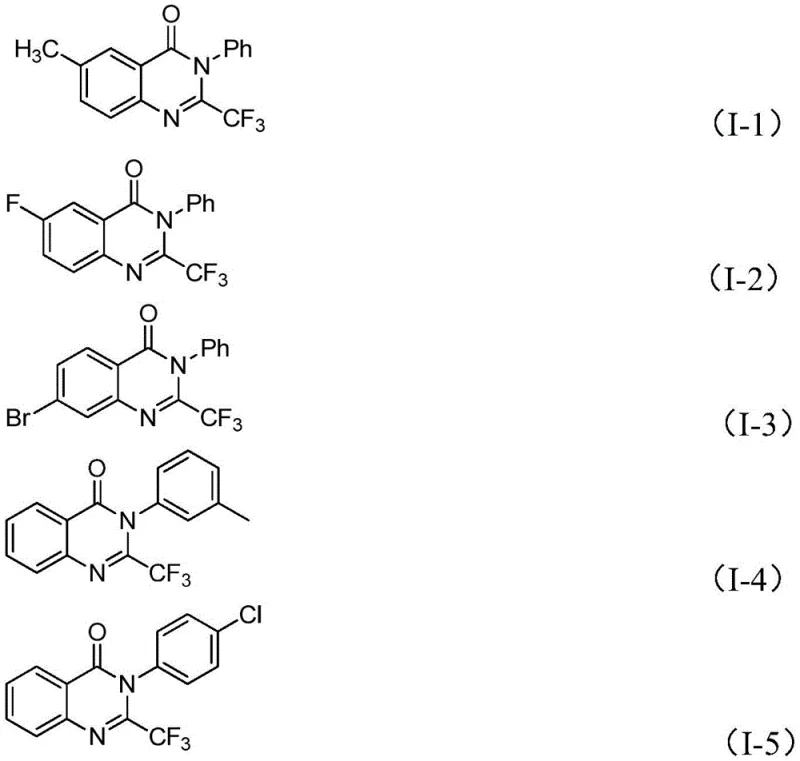
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions commonly associated with radical pathways or uncontrolled polymerization. The specific coordination geometry of the iron center directs the cyclization regioselectively, ensuring that the trifluoromethyl group is installed exclusively at the 2-position of the quinazolinone ring. This high regioselectivity is vital for pharmaceutical applications where isomeric impurities can complicate downstream purification and impact biological activity profiles. The tolerance for various substituents, including halogens like fluorine and bromine as well as alkyl groups, indicates that the catalytic cycle is robust against electronic variations in the substrate. This stability allows for the synthesis of a diverse library of analogs, such as those depicted in the structural examples, providing medicinal chemists with a versatile toolkit for structure-activity relationship studies.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of handling in standard laboratory and pilot plant settings. The process begins by combining the solid reagents, including the iron catalyst and base, with the liquid substrates in a polar aprotic solvent, creating a homogeneous reaction environment that maximizes molecular collisions. The two-stage temperature profile, starting at a lower temperature to form the intermediate and ramping up to drive the cyclization, is a critical parameter that balances reaction rate with product stability. Following the reaction completion, the workup procedure is straightforward, involving simple filtration to remove inorganic salts and molecular sieves, followed by standard chromatographic purification techniques that are familiar to process chemists. For a detailed breakdown of the specific molar ratios, solvent volumes, and temperature ramps optimized for different substrates, please refer to the standardized synthesis guide below.
- Mix ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in DMF solvent.
- Stir the reaction mixture at 40°C for 10 hours, then heat to 120°C under air atmosphere for an additional 20 hours to complete cyclization.
- Filter the reaction mixture, adsorb onto silica gel, and purify via column chromatography to isolate the high-purity quinazolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed protocol represents a strategic opportunity to optimize the cost structure of API manufacturing while securing a more resilient supply of critical intermediates. The shift from precious metal catalysts to commodity iron salts drastically reduces the raw material expenditure per kilogram of product, directly impacting the bottom line without compromising quality. Additionally, the elimination of complex heavy metal scavenging steps simplifies the downstream processing workflow, reducing the consumption of specialized resins and solvents typically required for metal removal. This streamlined process not only shortens the overall production cycle time but also minimizes the generation of hazardous waste, aligning with increasingly strict environmental regulations and sustainability goals imposed by global regulatory agencies.
- Cost Reduction in Manufacturing: The substitution of expensive palladium or rhodium catalysts with ferric chloride results in a substantial decrease in catalyst costs, which is particularly significant when scaling to multi-ton production volumes. Furthermore, the use of commercially available isatins and imidoyl chlorides avoids the need for custom synthesis of exotic starting materials, leveraging existing supply chains to keep input costs low. The high yields reported across various substrates mean that less raw material is wasted, improving the overall atom economy and reducing the cost of goods sold for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: By utilizing reagents that are widely produced and stocked by chemical suppliers globally, this method mitigates the risk of supply disruptions often associated with specialized or single-source catalysts. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is less sensitive to minor fluctuations in temperature or atmospheric conditions compared to air-sensitive organometallic reactions. This reliability ensures consistent delivery timelines for downstream partners, fostering stronger long-term relationships between intermediate suppliers and pharmaceutical developers who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: The protocol has been demonstrated to be effective from gram-scale discovery chemistry up to larger batch sizes, indicating a smooth path for technology transfer to commercial manufacturing facilities. The use of DMF, a common industrial solvent, facilitates easy solvent recovery and recycling, further enhancing the environmental profile of the process. Moreover, the absence of toxic heavy metals in the final product streamlines the regulatory filing process, as extensive data on residual metal limits is not required, accelerating the time to market for new drug candidates utilizing this scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology in industrial settings. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential licensees and manufacturing partners. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing production lines or for assessing the quality attributes of the resulting intermediates.
Q: What are the primary advantages of this iron-catalyzed method over traditional palladium catalysis?
A: This method utilizes inexpensive ferric chloride instead of precious metals like palladium, significantly reducing raw material costs and eliminating the need for complex heavy metal removal steps required in GMP manufacturing.
Q: Does this synthesis tolerate diverse functional groups on the aromatic ring?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully accommodating electron-donating groups like methyl and methoxy, as well as electron-withdrawing halogens such as fluorine, chlorine, and bromine without compromising yield.
Q: Is this process suitable for large-scale industrial production?
A: The process is designed for scalability, utilizing common solvents like DMF and robust reaction conditions that have been validated from gram-scale experiments up to potential multi-kilogram commercial batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed synthesis in delivering high-quality pharmaceutical intermediates to the global market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the laboratory bench to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone delivered meets the highest standards required for clinical and commercial applications. We are committed to supporting your R&D efforts with reliable supply and technical expertise that drives innovation forward.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next drug development program. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, and let us help you optimize your supply chain for success.
