Scalable Synthesis of Bioactive Indole-Derived Piperidines for Advanced Oncology Drug Development
Scalable Synthesis of Bioactive Indole-Derived Piperidines for Advanced Oncology Drug Development
The pharmaceutical industry is constantly seeking novel scaffolds that combine structural complexity with potent biological activity, particularly in the realm of oncology. Patent CN115785122A introduces a groundbreaking methodology for the synthesis of indole-derived piperidine compounds, a class of molecules that merges two privileged structures known for their presence in anticancer agents and natural products. This innovation addresses a critical gap in medicinal chemistry by providing a robust, one-pot synthetic route to these complex heterocycles. The disclosed method leverages Lewis acid catalysis to facilitate a highly stereoselective cyclization, resulting in products that exhibit remarkable sensitivity and strong cytotoxic activity against human breast cancer MCF-7 cells. For research and development teams focused on expanding their oncology pipeline, this technology offers a direct pathway to accessing diverse chemical space with high efficiency.
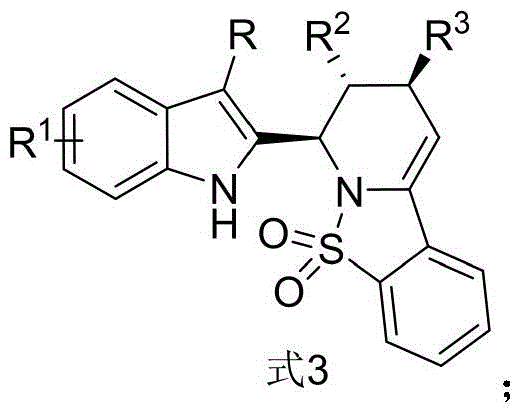
Traditionally, constructing fused or linked indole-piperidine systems often requires multi-step sequences involving harsh reagents, protecting group manipulations, and tedious purification processes that erode overall yield and increase production costs. Conventional approaches may suffer from poor stereocontrol, leading to difficult-to-separate diastereomeric mixtures that complicate downstream biological testing and regulatory filings. Furthermore, many existing methods rely on expensive transition metal catalysts that pose challenges regarding residual metal removal, a critical parameter for active pharmaceutical ingredients (APIs). These limitations create significant bottlenecks for procurement managers and supply chain heads who require reliable, cost-effective, and scalable sources of high-purity intermediates. The inability to efficiently access these scaffolds has historically restricted the exploration of their full therapeutic potential in drug discovery programs.
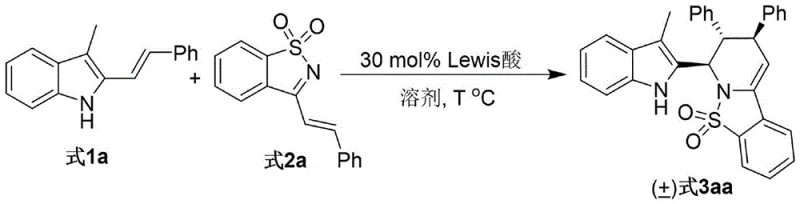
The novel approach detailed in the patent overcomes these historical barriers through an elegant Lewis acid-catalyzed cascade reaction. By reacting 3-alkyl-2-indole carbinols with alpha,beta-unsaturated N-sulfonyl imine derivatives, the method achieves the construction of the piperidine ring in a single operational step. The use of scandium triflate (Sc(OTf)3) as the catalyst of choice is particularly transformative, driving the reaction to completion with exceptional diastereoselectivity exceeding a 95:5 ratio. This high level of stereocontrol eliminates the need for complex chiral separations, significantly streamlining the manufacturing process. Moreover, the reaction proceeds under mild thermal conditions (50-90°C) in common organic solvents like acetonitrile, ensuring safety and ease of operation. This simplicity translates directly into reduced operational expenditure and a lower environmental footprint, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into Sc(OTf)3-Catalyzed Cyclization
The success of this synthesis hinges on the unique ability of the Sc(OTf)3 Lewis acid to activate the electrophilic species while maintaining compatibility with the nucleophilic indole moiety. Mechanistically, the scandium center coordinates with the nitrogen atom of the alpha,beta-unsaturated N-sulfonyl imine, increasing the electrophilicity of the beta-carbon. This activation facilitates a nucleophilic attack by the electron-rich C3 position of the indole ring (or potentially via an initial Friedel-Crafts type alkylation followed by cyclization depending on the specific substrate electronics). The subsequent intramolecular cyclization closes the piperidine ring, establishing multiple stereocenters in a highly controlled manner. The rigid transition state imposed by the catalyst-substrate complex ensures that the incoming groups approach from the preferred face, resulting in the observed high diastereoselectivity. Understanding this mechanism allows chemists to fine-tune substituent effects on both the indole and imine components to further optimize reaction outcomes.
Impurity control is another critical aspect where this mechanistic understanding provides value. The high selectivity of the Sc(OTf)3 catalyst minimizes the formation of regioisomers and oligomeric byproducts that often plague uncatalyzed or poorly catalyzed condensations. The reaction profile suggests that the formation of the desired piperidine ring is kinetically favored over competing polymerization pathways of the unsaturated imine. Additionally, the use of a 1:2 molar ratio of indole carbinol to imine derivative helps drive the reaction to completion while suppressing side reactions associated with excess nucleophile. The final purification via standard silica gel column chromatography effectively removes any trace catalyst or minor impurities, yielding a white solid product with high purity suitable for biological screening. This robustness in impurity profiling is essential for meeting the stringent quality standards required by regulatory bodies for pharmaceutical intermediates.
How to Synthesize Indole-Derived Piperidine Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators should follow the optimized protocol derived from the patent examples. The process begins with the precise weighing of 3-alkyl-2-indole carbinol and the alpha,beta-unsaturated N-sulfonyl imine derivative, maintaining a molar ratio of approximately 1:2 to ensure complete consumption of the limiting reagent. These solids are dissolved in anhydrous acetonitrile, which has been identified as the optimal solvent for balancing solubility and reaction rate. The addition of 30 mol% Sc(OTf)3 initiates the catalytic cycle, and the mixture is heated to 70°C with continuous stirring. Reaction progress is monitored via thin-layer chromatography (TLC) until the starting materials are fully consumed, typically within 8 hours. Upon completion, the reaction mixture is filtered to remove any insoluble particulates, concentrated under reduced pressure, and the residue is purified by flash column chromatography using a petroleum ether and ethyl acetate mixture. Detailed standardized synthesis steps are provided in the guide below.
- Combine 3-alkyl-2-indole carbinol and alpha,beta-unsaturated N-sulfonyl imine derivative in acetonitrile solvent.
- Add 30 mol% Sc(OTf)3 Lewis acid catalyst and stir the mixture at 70°C until TLC indicates completion.
- Filter the reaction mixture, concentrate the filtrate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly correlates to cost reduction in pharmaceutical intermediate manufacturing. By consolidating what would traditionally be a multi-step sequence into a single pot, the process eliminates the need for intermediate isolation, drying, and multiple reactor setups. This reduction in unit operations significantly lowers labor costs, energy consumption, and solvent usage. Furthermore, the avoidance of precious metal catalysts like palladium or rhodium, replacing them with the more abundant and recoverable scandium triflate, mitigates the risk of supply chain disruptions associated with critical raw materials. The economic model is further strengthened by the high atom economy of the reaction, ensuring that a maximum proportion of the input mass ends up in the final product.
- Cost Reduction in Manufacturing: The elimination of complex protection-deprotection strategies and the use of a single catalytic step significantly lowers the overall cost of goods sold (COGS). Since the reaction does not require cryogenic conditions or ultra-high pressures, the capital expenditure for specialized equipment is minimized, allowing production to occur in standard glass-lined or stainless steel reactors. The high yield and selectivity mean less raw material is wasted on off-spec products, maximizing the return on investment for every kilogram of starting material purchased. Additionally, the simple workup procedure reduces the volume of hazardous waste generated, lowering disposal costs and environmental compliance burdens.
- Enhanced Supply Chain Reliability: The starting materials, specifically 3-alkyl-2-indole carbinols and N-sulfonyl imines, are structurally simple and can be sourced from a wide range of global chemical suppliers or synthesized via well-established commodity routes. This diversity of supply sources reduces the risk of single-vendor dependency and ensures business continuity even during market fluctuations. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental factors, leading to consistent batch-to-batch reproducibility. This reliability is crucial for maintaining steady production schedules and meeting the just-in-time delivery requirements of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial large-scale production in mind from the outset. The use of acetonitrile, a solvent with well-understood recycling protocols, facilitates closed-loop solvent recovery systems that minimize VOC emissions. The absence of heavy metal contaminants in the final product simplifies the regulatory approval process for clinical trial materials, accelerating time-to-market. The mild reaction temperatures enhance operational safety, reducing the risk of thermal runaways and ensuring a safer working environment for plant personnel. These factors collectively position this technology as a sustainable and compliant solution for long-term commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this indole-piperidine synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the fit of this technology within their existing R&D and manufacturing frameworks. The focus is on practical applicability, quality assurance, and the strategic advantages offered by this novel chemical transformation.
Q: What is the primary advantage of using Sc(OTf)3 in this synthesis?
A: Sc(OTf)3 provides superior diastereoselectivity (>95:5 dr) and high yields under mild conditions compared to other Lewis acids like Cu(OTf)2 or ZnCl2, ensuring consistent product quality for pharmaceutical applications.
Q: Is this synthesis method suitable for large-scale manufacturing?
A: Yes, the process utilizes conventional reaction conditions, simple workup procedures involving filtration and concentration, and commercially available raw materials, making it highly amenable to industrial scale-up.
Q: What biological activity do these compounds exhibit?
A: The synthesized indole-derived piperidine compounds demonstrate significant cytotoxic activity against human breast cancer MCF-7 cells, with IC50 values indicating strong potential for oncology drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole-Derived Piperidine Supplier
As the demand for sophisticated oncology intermediates grows, partnering with an experienced CDMO becomes essential for translating patent innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific requirements of Lewis acid-catalyzed reactions, including rigorous moisture control and efficient solvent recovery systems. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of indole-derived piperidine meets the highest international standards for pharmaceutical applications. Our commitment to quality ensures that your drug development timeline remains on track without interruptions due to supply chain inconsistencies.
We invite you to leverage our technical expertise to optimize your specific derivative synthesis. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project volume. We are prepared to provide specific COA data for reference standards and comprehensive route feasibility assessments to demonstrate how we can support your transition from bench-scale discovery to full-scale commercial manufacturing. Let us be your strategic partner in bringing these promising anticancer candidates to the patients who need them most.
