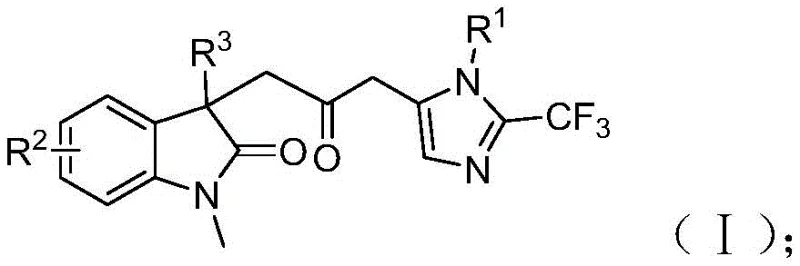
Advanced Palladium-Catalyzed Synthesis Pathway for High-Purity Carbonyl-Bridged Biheterocyclic Compounds at Commercial Scale
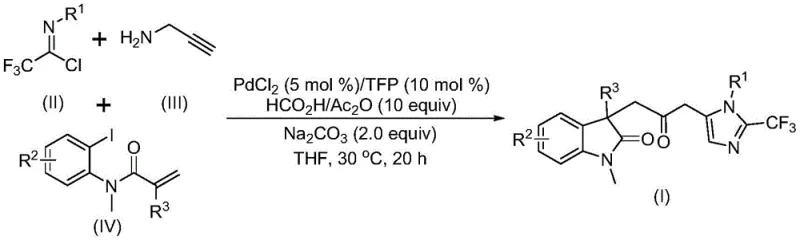
Our analysis of Chinese Patent CN115353511A reveals a groundbreaking multi-component synthesis method for carbonyl-bridged biheterocyclic compounds, which represent critical structural motifs in numerous bioactive pharmaceutical molecules including indolinone and imidazole derivatives with broad-spectrum biological activities. This innovative approach addresses longstanding challenges in heterocyclic chemistry by eliminating the need for toxic carbon monoxide gas while maintaining high reaction efficiency and substrate compatibility across diverse molecular frameworks. The patented methodology demonstrates exceptional potential for industrial scale-up, having been successfully validated at gram-scale with fifteen distinct substrate combinations yielding products between 40% and 92%. By leveraging readily available starting materials including trifluoroethylimidoyl chloride, propargylamine, and acrylamide under mild conditions (30°C), this process offers a significant advancement over conventional transition metal-catalyzed carbonylation techniques that require hazardous pressurized CO gas and specialized equipment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing biheterocyclic compounds typically rely on three primary methodologies: direct coupling of two heterocyclic substrates, oxidative cyclization using bifunctional nucleophiles with activated methyl-substituted heterocycles, or transition metal-catalyzed tandem cyclization reactions. These conventional methods often suffer from significant limitations including the requirement for toxic carbon monoxide gas under high-pressure conditions, narrow substrate scope with poor functional group tolerance, and complex multi-step procedures that increase production costs and reduce overall efficiency. The necessity for specialized equipment to handle pressurized CO presents substantial safety concerns and capital investment barriers for pharmaceutical manufacturers seeking scalable production pathways. Furthermore, many existing protocols demonstrate inconsistent yields across different substrate combinations and frequently require extensive purification processes to achieve pharmaceutical-grade purity specifications, resulting in higher waste generation and reduced process economics that undermine commercial viability for complex heterocyclic intermediate production.
The Novel Approach
The patented methodology presented in CN115353511A introduces a revolutionary palladium-catalyzed carbonylation cascade reaction that overcomes these critical limitations through an elegant multi-component strategy operating under ambient pressure conditions. By utilizing a formic acid/acetic anhydride mixture as a safe carbon monoxide surrogate at mild temperature (30°C), this process eliminates hazardous reagent handling while maintaining excellent reaction efficiency across diverse substrate combinations as demonstrated by fifteen successful implementations with yields ranging from 40% to 92%. The innovative catalytic system comprising palladium chloride (5 mol%) with trifuryl phosphine ligand (10 mol%) in tetrahydrofuran solvent enables high-yielding synthesis of structurally diverse carbonyl-bridged biheterocyclic compounds with exceptional functional group tolerance across various substitution patterns on R¹ (alkyl/aryl), R² (H/alkyl/halogen), and R³ (alkyl/phenyl/benzyl) positions. This one-pot methodology demonstrates remarkable versatility while featuring simplified workup procedures involving filtration and column chromatography that enhance practicality for industrial implementation in pharmaceutical manufacturing environments.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The reaction mechanism begins with oxidative addition of zero-valent palladium into the carbon-iodine bond of the iodinated precursor, followed by intramolecular Heck reaction to generate a key divalent alkyl palladium intermediate that serves as the foundation for subsequent transformations. This intermediate then undergoes carbonylation through carbon monoxide released from the formic acid/acetic anhydride mixture under mild conditions, forming an acyl palladium species that functions as the crucial electrophilic component for constructing the carbonyl bridge between heterocyclic systems. Simultaneously, base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine intermediate through intermolecular carbon-nitrogen bond formation, which subsequently undergoes isomerization to create the active nucleophilic species required for final cyclization.
This sophisticated cascade process efficiently constructs multiple chemical bonds in a single operation while maintaining excellent regioselectivity and stereochemical control through precise coordination chemistry at the palladium center. Impurity profile management is achieved through careful control of reaction parameters including temperature (30°C), stoichiometry (trifluoroethylimidoyl chloride:propargylamine:acrylamide:palladium chloride = 1:2:1.5:0.05), and solvent selection (THF), which collectively prevent side reactions while ensuring complete conversion of starting materials without degradation pathways that could compromise product quality.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
This patented methodology represents a significant advancement in the synthesis of complex biheterocyclic structures, offering pharmaceutical manufacturers a safer, more efficient alternative to traditional carbonylation approaches by eliminating hazardous reagents while maintaining high yields across diverse substrate combinations. The process demonstrates exceptional versatility through strategic modification of R-group substitutions on precursor molecules while preserving consistent reaction performance under standardized conditions. The simplified workup procedure involving filtration followed by column chromatography provides a reliable foundation for technology transfer to manufacturing facilities seeking robust production methods for complex heterocyclic intermediates required in drug discovery programs.
- Prepare reaction mixture with precise molar ratios: trifluoroethylimidoyl chloride (1 equiv), propargylamine (2 equiv), acrylamide (1.5 equiv), palladium chloride (0.05 equiv), trifuryl phosphine (0.1 equiv), sodium carbonate (2 equiv), and formic acid/acetic anhydride mixture (10 equiv) in THF solvent.
- Conduct reaction at controlled temperature of 30°C for 16 hours under standard atmospheric pressure without specialized equipment, monitoring progress through established analytical protocols to ensure complete conversion.
- Implement standardized workup procedure involving filtration through silica gel followed by column chromatography purification to achieve pharmaceutical-grade purity specifications while removing catalyst residues.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate supply chains by offering a more sustainable production pathway that enhances both operational reliability and environmental compliance without compromising product quality or yield consistency. The elimination of hazardous materials reduces regulatory burdens while improving workplace safety profiles across manufacturing facilities, creating significant value across procurement operations through simplified logistics and reduced risk management requirements.
- Cost Reduction in Manufacturing: The elimination of toxic carbon monoxide gas removes substantial safety infrastructure costs associated with specialized handling equipment while reducing regulatory compliance expenses related to hazardous material management. The use of inexpensive palladium chloride catalyst combined with commercially available starting materials significantly lowers raw material expenses compared to traditional methods requiring expensive transition metal complexes or specialized reagents.
- Enhanced Supply Chain Reliability: Reliance on readily available non-hazardous starting materials ensures consistent supply chain performance without vulnerabilities associated with regulated or specialized reagents that may face availability constraints. The robust reaction profile maintains high yields across diverse substrate combinations, providing manufacturing flexibility to accommodate changing production demands without requiring extensive process revalidation or equipment modifications.
- Scalability and Environmental Compliance: Mild reaction conditions (30°C) at atmospheric pressure enable straightforward scale-up using standard manufacturing equipment without costly modifications or specialized infrastructure investments. The elimination of hazardous reagents significantly reduces environmental impact while simplifying regulatory compliance pathways, aligning with industry sustainability goals through efficient atom economy that minimizes waste generation per unit of product produced.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations regarding implementation of this patented synthesis methodology for pharmaceutical intermediate production based on detailed analysis of patent specifications and experimental data provided in CN115353511A.
Q: How does this method eliminate safety concerns associated with traditional carbonylation?
A: The patented process replaces hazardous pressurized carbon monoxide gas with a safe formic acid/acetic anhydride mixture as CO surrogate, eliminating specialized high-pressure equipment requirements while maintaining excellent reaction efficiency at mild conditions (30°C). This fundamental redesign removes significant safety infrastructure costs and workplace hazards without compromising yield or product quality.
Q: What substrate flexibility does this methodology offer for pharmaceutical development?
A: The process demonstrates exceptional functional group tolerance across fifteen distinct substrate combinations with yields ranging from 40% to 92%, enabling synthesis of diverse trifluoromethyl-substituted biheterocyclic structures through strategic modification of R¹, R², and R³ positions on precursor molecules while maintaining consistent reaction performance.
Q: How does the process ensure consistent purity for pharmaceutical applications?
A: The mild reaction temperature prevents thermal decomposition pathways while optimized stoichiometry ensures complete conversion without side reactions. Standardized workup involving filtration followed by column chromatography effectively removes catalyst residues, delivering products meeting stringent pharmaceutical purity specifications through robust impurity control strategy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic intermediates, ensuring seamless technology transfer from laboratory to manufacturing scale through rigorous process development protocols. Our state-of-the-art facilities feature stringent purity specifications maintained through advanced analytical capabilities in dedicated QC labs that provide comprehensive characterization data meeting global regulatory requirements for pharmaceutical applications.
Request a Customized Cost-Saving Analysis today to understand how our patented methodology can enhance your supply chain efficiency through optimized production pathways tailored to your specific compound requirements. Contact our technical procurement team to receive specific COA data and route feasibility assessments that enable informed decision-making for your next pharmaceutical development program.
