Advanced Metal-Free Synthesis of 1,2,3-Triazoles for Scalable Pharmaceutical Manufacturing
The landscape of heterocyclic chemistry is undergoing a significant transformation, driven by the urgent need for greener, safer, and more cost-effective synthetic routes for critical building blocks. Patent CN110015999B introduces a groundbreaking methodology for the synthesis of 1,2,3-triazole compounds, a structural motif ubiquitous in modern drug discovery and agrochemical development. Unlike traditional approaches that rely heavily on transition metal catalysis or harsh basic conditions, this invention leverages a simple yet highly effective acid-promoted one-pot reaction. By utilizing p-toluenesulfonyl azide, various amine compounds, and dicarbonyl substrates in the presence of acetic acid, the process achieves high yields while completely bypassing the need for expensive and toxic metal catalysts. This technological leap not only addresses the growing regulatory pressure regarding heavy metal residues in Active Pharmaceutical Ingredients (APIs) but also streamlines the manufacturing workflow, offering a compelling value proposition for both R&D teams focusing on purity and supply chain managers seeking operational efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,3-triazole ring has been dominated by the celebrated Copper(I)-catalyzed Azide-Alkyne Cycloaddition (CuAAC), often referred to as the "Click" reaction. While renowned for its high yield and regioselectivity, this method suffers from significant drawbacks when applied to the synthesis of complex pharmaceutical intermediates. The primary concern is the inevitable introduction of copper ions into the reaction matrix, which necessitates rigorous and costly purification steps to meet stringent regulatory limits for residual metals in drug substances. Furthermore, alternative methods involving Ruthenium catalysts or strong base-promoted cyclizations of enamines and azides present their own set of challenges, including limited substrate scope, harsh reaction conditions that compromise functional group tolerance, and the generation of hazardous waste streams. These conventional pathways often require inert atmosphere protection, such as nitrogen or argon, adding layers of complexity and equipment costs to the manufacturing process, thereby hindering their applicability in large-scale industrial settings where simplicity and robustness are paramount.
The Novel Approach
In stark contrast to these legacy technologies, the method disclosed in CN110015999B offers a paradigm shift by employing a metal-free, acid-catalyzed strategy that operates under remarkably mild conditions. The core innovation lies in the synergistic interaction between p-toluenesulfonyl azide, aniline derivatives, and beta-dicarbonyl compounds within a dichloromethane solvent system, facilitated by acetic acid. This approach eliminates the dependency on transition metals entirely, thereby removing the risk of metal contamination and the associated downstream purification burden. The reaction proceeds efficiently in a single reactor without the need for intermediate isolation or separation steps, characterizing it as a true one-pot synthesis. Moreover, the process does not require an oxygen-free environment or specialized inert gas protection, significantly reducing operational overhead. By optimizing parameters such as temperature (40-90°C) and reaction time (24-48 hours), this novel route delivers superior yields, often exceeding 90%, while maintaining a clean impurity profile that simplifies final product isolation.
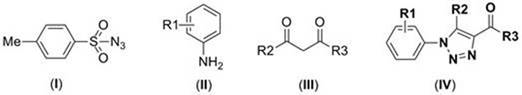
Mechanistic Insights into Acid-Promoted Triazole Cyclization
The mechanistic pathway of this acid-catalyzed cyclization represents a sophisticated orchestration of organic transformations that avoids the pitfalls of radical or organometallic mechanisms. The reaction initiates with the activation of the dicarbonyl component by the acidic additive, likely acetic acid, which enhances the electrophilicity of the carbonyl carbon and facilitates nucleophilic attack by the amine. This initial condensation forms an enamine or imine intermediate in situ, which subsequently reacts with the p-toluenesulfonyl azide. The sulfonyl azide acts as a masked source of nitrogen, undergoing decomposition or rearrangement to generate a reactive nitrene or diazo species that engages with the electron-rich enamine system. The presence of the acid is critical not only for activating the carbonyl but also for stabilizing charged intermediates throughout the cyclization cascade, driving the formation of the triazole ring with high regioselectivity. This mechanism ensures that the reaction proceeds smoothly without the need for external energy inputs like UV light or high-pressure conditions, making it inherently safer and more controllable than photochemical or high-energy alternatives.
From an impurity control perspective, the absence of metal catalysts fundamentally alters the impurity landscape of the final product. In metal-catalyzed reactions, side products often include metal-complexed species or oligomers formed via radical pathways initiated by the metal center. In this acid-mediated process, the primary byproducts are derived from simple hydrolysis or over-alkylation, which are generally easier to separate via standard chromatographic techniques. The use of p-toluenesulfonyl azide, while requiring careful handling due to its energetic nature, results in the formation of p-toluenesulfonic acid derivatives as leaving groups, which are water-soluble and easily removed during aqueous workup. This clean reaction profile minimizes the formation of difficult-to-remove trace impurities, ensuring that the resulting 1,2,3-triazole compounds meet the high-purity standards required for reliable pharmaceutical intermediate supplier specifications. The robustness of this mechanism across a wide range of substituents on the aniline and dicarbonyl components further underscores its versatility for generating diverse chemical libraries.
How to Synthesize 1,2,3-Triazole Compounds Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot-plant production. The protocol involves charging a reaction vessel with the three key starting materials—p-toluenesulfonyl azide, the chosen amine, and the dicarbonyl compound—along with the acetic acid additive in dichloromethane. The mixture is then heated to the optimized temperature range, typically around 90°C, and stirred for a period of 24 to 48 hours to ensure complete conversion. Following the reaction, the crude product can be isolated through standard extraction and purification methods, such as column chromatography, to afford the target triazole derivative in high purity. For a detailed breakdown of the specific molar ratios, solvent volumes, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Combine p-toluenesulfonyl azide, amine compounds, and dicarbonyl compounds in an organic solvent such as dichloromethane.
- Add acetic acid as a catalytic additive to the reaction mixture to promote cyclization under mild heating conditions.
- Heat the reaction mixture to 90°C for 24 hours, then purify the resulting 1,2,3-triazole product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route translates into tangible strategic advantages that extend beyond mere chemical elegance. The elimination of precious metal catalysts like copper or ruthenium removes a significant variable cost from the bill of materials, as these metals are subject to volatile market pricing and supply constraints. Furthermore, the removal of metal scavenging resins and the associated filtration steps drastically reduces the consumption of consumables and waste disposal costs. The simplified one-pot nature of the reaction means fewer unit operations are required, leading to reduced labor hours and increased throughput capacity in existing manufacturing facilities. This streamlined process flow enhances overall equipment effectiveness (OEE) and allows for faster turnaround times between batches, directly addressing the need for reducing lead time for high-purity pharmaceutical intermediates in a competitive market environment.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the complete eradication of expensive transition metal catalysts and the auxiliary reagents required to remove them. Traditional click chemistry often necessitates the use of specialized ligands and subsequent purification columns to strip residual copper to parts-per-million levels, a process that is both time-consuming and resource-intensive. By switching to an acetic acid-promoted system, manufacturers can bypass these costly downstream processing steps entirely. Additionally, the use of commodity chemicals such as anilines and beta-keto esters as starting materials ensures a stable and low-cost supply chain, shielding production budgets from the volatility associated with specialty catalytic reagents. This structural simplification of the synthetic route inherently lowers the cost of goods sold (COGS), providing a stronger margin profile for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the availability and stability of raw materials. The reagents utilized in this patented method, including p-toluenesulfonyl azide and various substituted anilines, are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. Unlike methods requiring air-sensitive catalysts that demand specialized storage and handling logistics, the reagents in this process are robust and can be managed with standard warehouse protocols. The reaction's tolerance to ambient conditions, specifically the lack of a requirement for nitrogen or argon blanketing, further simplifies the logistical footprint of the manufacturing site. This operational flexibility ensures consistent production schedules and mitigates the risk of delays caused by equipment failure or utility interruptions, thereby securing a steady flow of high-quality intermediates to downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden inefficiencies, but the one-pot design of this triazole synthesis is inherently scalable. The absence of exothermic metal-catalyzed steps reduces the thermal load on the reactor, minimizing the need for complex cooling systems and enhancing process safety. From an environmental standpoint, the avoidance of heavy metals aligns perfectly with green chemistry principles and increasingly strict environmental regulations regarding wastewater discharge. The solvent system, primarily dichloromethane, can be efficiently recovered and recycled, further minimizing the environmental footprint. This compliance-ready profile facilitates smoother regulatory approvals and reduces the administrative burden associated with environmental health and safety (EHS) reporting, making it an ideal candidate for sustainable manufacturing initiatives in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triazole synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on performance metrics and operational requirements. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production pipelines or for procurement specialists assessing the quality implications of the new method.
Q: Why is the metal-free synthesis of 1,2,3-triazoles preferred for pharmaceutical applications?
A: Traditional methods often utilize copper or ruthenium catalysts which can leave toxic metal residues in the final API. The method in CN110015999B eliminates metal catalysts entirely, ensuring higher purity and simplifying downstream purification processes.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies using dichloromethane as the solvent and acetic acid as the additive. The reaction proceeds efficiently at temperatures between 40-90°C, with 90°C for 24 hours yielding the best results.
Q: Can this process be scaled for industrial production?
A: Yes, the one-pot nature of the reaction, combined with the use of common solvents and the absence of sensitive metal catalysts or inert gas protection, makes it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,3-Triazole Compounds Supplier
The technological advancements detailed in CN110015999B represent a significant opportunity for the pharmaceutical and agrochemical industries to optimize their supply chains for critical heterocyclic building blocks. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage this metal-free synthesis route effectively, ensuring the delivery of high-quality intermediates that meet the rigorous demands of modern drug development. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, allowing us to seamlessly transition this innovative chemistry from the bench to the plant. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1,2,3-triazole compounds is free from metallic contaminants and adheres to the highest industry standards.
We invite potential partners to engage with our technical procurement team to discuss how this cost-effective and environmentally friendly synthesis method can be tailored to your specific project needs. By collaborating with us, you can access a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this metal-free protocol for your specific target molecules. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of innovation, reliability, and superior value.
