Scalable Metal-Free Synthesis of Trifluoromethyl-Triazoles for Advanced Pharmaceutical Intermediate Production
Scalable Metal-Free Synthesis of Trifluoromethyl-Triazoles for Advanced Pharmaceutical Intermediate Production
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). Patent CN113105402B introduces a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, specifically targeting those bearing both trifluoromethyl and acyl groups. This innovation addresses a significant gap in current literature, where methods to simultaneously introduce these functionalities are scarce. The disclosed technology leverages a non-metallic iodine-promoted system in dimethyl sulfoxide (DMSO), offering a streamlined alternative to traditional transition-metal catalysis. For R&D directors and process chemists, this represents a pivotal shift towards more sustainable and cost-effective manufacturing protocols for high-value intermediates used in drugs like sitagliptin and maraviroc.
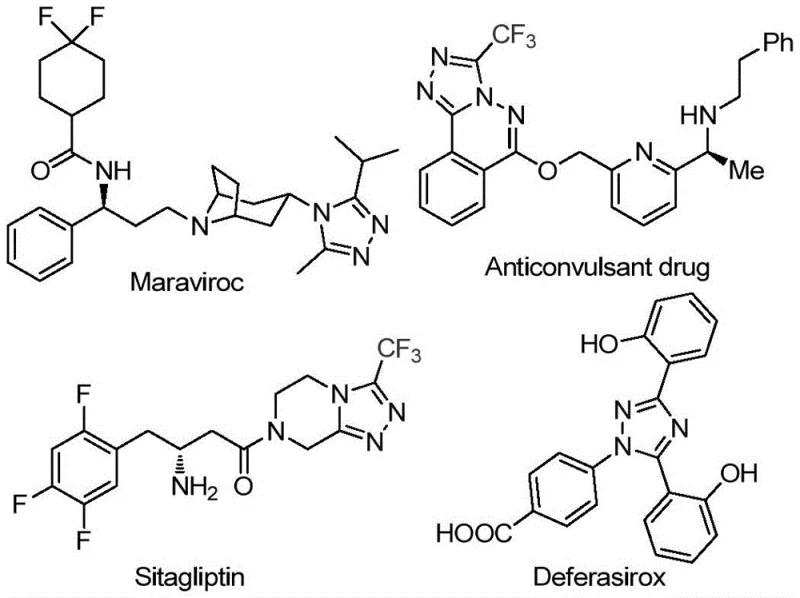
The strategic importance of this patent lies in its ability to simplify the supply chain for complex nitrogen-containing heterocycles. By utilizing cheap and readily available starting materials such as aryl ethanones and trifluoroethylimine hydrazides, the method drastically reduces raw material costs. Furthermore, the elimination of stringent anhydrous and oxygen-free requirements lowers the barrier for industrial adoption. This report analyzes the technical merits of this iodine-catalyzed cyclization, providing procurement managers and supply chain heads with a clear understanding of how this technology can enhance production reliability and reduce lead times for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles has relied heavily on transition metal catalysts or harsh reaction conditions that pose significant challenges for large-scale manufacturing. Conventional routes often require expensive palladium or copper complexes, which not only inflate the cost of goods sold (COGS) but also introduce complex purification steps to remove trace heavy metals to meet regulatory standards. Additionally, many existing protocols demand strictly anhydrous and inert atmospheres, necessitating specialized equipment and increasing operational complexity. The scarcity of methods capable of efficiently installing both trifluoromethyl and acyl groups simultaneously has further limited the structural diversity accessible to medicinal chemists, often forcing reliance on multi-step sequences with poor overall atom economy.
The Novel Approach
The methodology outlined in CN113105402B disrupts these traditional paradigms by employing elemental iodine as a benign and effective promoter. This novel approach utilizes a tandem sequence involving iodination and Kornblum oxidation of aryl ketones, followed by condensation and cyclization with trifluoroethylimine hydrazides. The reaction proceeds smoothly in DMSO at elevated temperatures (110-130°C) without the need for exotic ligands or sensitive catalysts. This metal-free strategy not only simplifies the workup procedure—often requiring only filtration and column chromatography—but also significantly broadens the functional group tolerance. The ability to access diverse 3,4,5-trisubstituted derivatives with high efficiency makes this a superior choice for generating libraries of bioactive molecules.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The core of this synthetic breakthrough involves a sophisticated yet operationally simple cascade reaction mechanism initiated by the interaction between iodine and dimethyl sulfoxide. Initially, the aryl ethyl ketone undergoes an iodination followed by a Kornblum oxidation to generate an aryl diketone intermediate in situ. This reactive species then condenses with the trifluoroethylimine hydrazide to form a hydrazone intermediate. Under the continued influence of iodine and the basic environment provided by sodium dihydrogen phosphate and pyridine, the hydrazone undergoes an intramolecular cyclization. This final step constructs the 1,2,4-triazole ring while preserving the critical trifluoromethyl and acyl functionalities. The precise control of reaction temperature and stoichiometry ensures high conversion rates and minimizes side reactions.
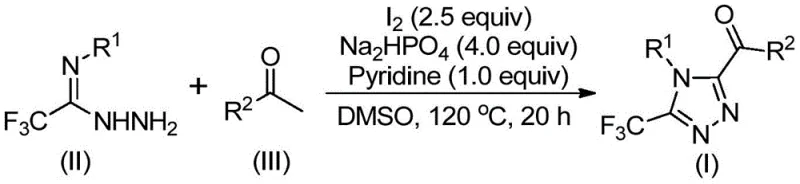
From an impurity control perspective, the mechanism offers distinct advantages due to the clean nature of the iodine-mediated transformations. The use of DMSO as both solvent and oxidant precursor streamlines the reaction profile, reducing the formation of complex byproducts often seen in metal-catalyzed cross-couplings. The stoichiometric balance, specifically the molar ratio of sodium dihydrogen phosphate to pyridine to iodine (4:1:2.5), is critical for driving the equilibrium towards the desired triazole product. Understanding this mechanistic pathway allows process engineers to fine-tune parameters for maximum yield, ensuring that the final API intermediate meets stringent purity specifications required for downstream pharmaceutical applications without extensive recrystallization.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
Implementing this synthesis requires careful attention to the two-stage heating protocol described in the patent to ensure complete conversion of the starting materials. The initial phase focuses on the activation of the ketone, while the second phase drives the cyclization to completion. Detailed standard operating procedures regarding reagent addition order and thermal profiles are essential for reproducibility. For a comprehensive guide on executing this transformation in a pilot or production setting, please refer to the standardized synthesis steps outlined below.
- Mix aryl ethyl ketone and iodine in DMSO, heating to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Add sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this iodine-promoted synthesis offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio. By relying on commodity chemicals like acetophenones and elemental iodine, manufacturers can mitigate supply risks associated with specialized catalysts that often face geopolitical or logistical bottlenecks. The robustness of the reaction conditions means that production can be scheduled with greater flexibility, as there is no need for extensive drying of solvents or purging of reactors with inert gases, thereby reducing utility costs and turnaround time between batches.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts such as palladium or rhodium directly translates to significant cost savings in raw material expenditure. Furthermore, the simplified post-treatment process, which avoids complex metal scavenging steps, reduces the consumption of silica gel and solvents during purification. This streamlined workflow lowers the overall operational expenditure (OPEX) per kilogram of produced intermediate, enhancing the margin potential for the final API. The use of inexpensive reagents ensures that the cost structure remains stable even during fluctuations in the global market for specialty chemicals.
- Enhanced Supply Chain Reliability: Sourcing aryl ethyl ketones and iodine is straightforward, as these are bulk chemicals available from multiple global suppliers, reducing dependency on single-source vendors. The tolerance of the reaction to ambient moisture and oxygen means that storage and handling requirements for intermediates are less stringent, minimizing the risk of batch failures due to environmental exposure. This resilience strengthens the supply chain against disruptions, ensuring consistent delivery schedules for downstream pharmaceutical clients who rely on just-in-time inventory models for their production lines.
- Scalability and Environmental Compliance: The protocol is inherently scalable, having been demonstrated to work efficiently from gram to multi-kilogram scales without loss of yield or selectivity. The absence of toxic heavy metals simplifies waste management and disposal, aligning with increasingly strict environmental regulations and green chemistry principles. This eco-friendly profile not only reduces the cost of waste treatment but also enhances the corporate sustainability metrics of the manufacturing facility, making it a more attractive partner for environmentally conscious pharmaceutical companies seeking compliant supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the method described in patent CN113105402B utilizes elemental iodine as a non-metal promoter, eliminating the need for costly palladium or copper catalysts and simplifying downstream purification.
Q: What are the optimal reaction conditions for scaling this process?
A: The process operates effectively in DMSO at temperatures between 110°C and 130°C without strict anhydrous or oxygen-free conditions, making it highly suitable for large-scale industrial reactors.
Q: Can this method tolerate diverse functional groups on the aromatic rings?
A: Yes, the protocol demonstrates excellent substrate scope, accommodating various substituents such as methyl, methoxy, chloro, and trifluoromethyl groups on both the hydrazide and ketone components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iodine-promoted synthesis described in CN113105402B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of this DMSO-based process, while our rigorous QC labs guarantee that every batch meets stringent purity specifications required for clinical and commercial use.
We invite you to collaborate with us to leverage this cost-effective and scalable technology for your next drug development program. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this metal-free route can optimize your budget. Please contact our technical procurement team today to request specific COA data for our triazole intermediates and discuss detailed route feasibility assessments for your target molecules.
